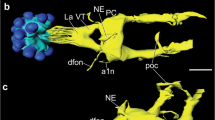
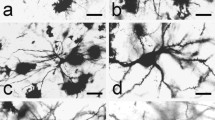
Summary
The gross structure as well as the neuronal and non-neuronal components of the lamina ganglionaris of the locust Schistocerca gregaria are described on the basis of light- and electron-microscopical preparations of Golgj (selective silver) and ordinary histological preparations. The array of optic cartridges within the lamina neuropile — their order and arrangement — and the composition of the cartridges are described. There are six types of monopolar neurons: three whose branches reach to other cartridges and three whose branches are confined to their own cartridges. Retinula axons terminate either in the lamina or the medulla neuropiles. There are three types of centrifugal neurons, two types of horizontal neuron, as well as glia and trachea in the lamina neuropile. The development of the lamina neuropile is described in terms of developing monopolar and centrifugal axons, growing retinula fibres, and composition of the developing optic cartridges.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Anderson H (1976) Postembryonic development of the insect visual system. PhD thesis, University of Leicester
Anderson H (1978a) Postembryonic development of the visual system of the locust, Schistocerca gregaria. I. Pattern of growth and developmental interactions in the retina and optic lobe. J Embryol Exp Morphol 45:55–83
Anderson H (1978b) Postembryonic development of the visual system of the locust, Schistocerca gregaria. II. An experimental investigation of the formation of the retina-lamina projection. J Embryol Exp Morphol 46:147–170
Bennett R, Tunstall J, Horridge GA (1967) Spectral sensitivity of single retinula cells of the locust. Z vergl Physiol 55:195–206
Braitenberg V (1970) Ordnung und Orientierung der Elemente im Sehsystem der Fliege. Kybernetik 7:235–242
Burtt ET, Catton WT (1966) Perception by locusts of rotated patterns. Science 151:224
Butler R (1973) The anatomy of the compound eye of Periplaneta americana L. I. General features. J Comp Physiol 83:223–238
Cajal SR (1909) Nota sobre la retina de la mosca (M. vomitoria L). Trab Lab Invest Biol Univ Madr 7:217–257
Cajal SR, Sánchez D (1915) Contribucion al conocimiento de los centros nerviosos de los insectos. Trab Lab Invest Biol Univ Madr 13:1–168
Dunn GA (1971) Mutual contact inhibition of extension of chick sensory nerve fibres in vitro. J Comp Neurol 143:491–508
Eley S, Shelton PMJ (1976) Cell junctions in the developing compound eye of the desert locust Schistocerca gregaria. J Embryol Exp Morphol 36:409–423
Goetz KG (1968) Flight control in Drosophila by visual perception of motion. Kybernetik 4:199–208
Goetz KG (1972) Processing of cues from the moving environment in the Drosophila navigation system. In: Wehner R (ed) Information Processing in the Visual Systems of Arthropods. Springer, Berlin Heidelberg New York, p 255–264
Hayat MA (1970) Principles and techniques of electron microscopy: Biological application. Vol I. The Nostrand Reinhold Company, New York London, p 342–343
Horridge GA (1966) The retina of the locust. In: Proceedings of the International Symposium on the functional organization of the compound eye. Pergamon Press, Oxford New York, p 513–541
Horridge GA, Barnard PBT (1965) Movement of palisade in locust retinula cells when illuminated. Quart J Micr Sci 106:131–135
Horridge GA, Scholes JH, Shaw S, Tunstall J (1965) Extracellular recordings from single neurones in the optic lobe and brain of the locust. In: Treherne JE, Beament JWL (eds) The physiology of the insect central nervous system. Academic Press, London New York, p 165–202
Hunt RK, Jacobson M (1974) Neuronal specificity revisited. Curr Top Devl Biol 8:203–259
Karnovsky MJ (1965) A formaldehyde-glutaraldehyde fixative of high osmolality for use in electron microscopy. J Cell Biol 27:137A
Kien (1975) Motion detection in locusts and grasshoppers. In: Horridge GA (ed) Compound eye and vision of insects. Clarendon Press, Oxford, p 411–422
LoPresti V, Macagno ER, Levinthal C (1973) Structure and development of neuronal connections in isogenic organisms: cellular interactions in the development of the optic lamina of Daphnia. Proc Natl Acad Sci USA 70:433–437
McCann GD, Dill JC (1969) Fundamental properties of intensity, form and motion perception in the visual nervous systems of Calliphora phoenicia and Musca domestica. J Gen Physiol 53:385–413
McCann GD, Foster SF (1971) Binocular interactions of motion detection fibres in the optic lobes of flies. Kybernetik 8:193–203
Meinertzhagen IA (1973) Development of the compound eye and optic lobe of insects. In: Young D (ed) Developmental neurobiology of arthropods. Cambridge University Press, London, p 51–104
Meinertzhagen IA (1976) The organization of perpendicular fibre pathways in the insect optic lobe. Phil Trans R Soc Lond B 274:555–596
Mouze M (1978a) Contribution a l'étude du développement postembryonnaire de l'appareil visuel des Odonates Anisoptères (Insectes). PhD thesis, Université des Sciences et Techniques de Lille
Mouze M (1978b) Role des fibres post-rétiniennes dans la croissance du lobe optique de la larve d'Aeshna cyanea Müll. (Insecte, Odonate). Wilhelm Roux's Arch 184:325–350
Nordlander RH, Edwards JS (1969) Postembryonic brain development in the Monarch butterfly Danaus plexippus plexippus L. II. The optic lobes. Wilhelm Roux Arch Entw Mech Org 163:197–220
Palka J (1965) Diffraction and visual acuity of insects. Science 149:551–553
Palka J, Pinter RB (1975) Theoretical and experimental analysis of visual acuity in insects. In: Horridge GA (ed) The compound eye and vision of insects. Clarendon Press, Oxford, p 321–337
Pantin CFA (1969) Notes on microscopical technique for zoologists. Cambridge University Press, London
Pease DC (1964) Histological techniques for electron microscopy. Academic Press, New York
Reynolds EW (1963) The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol 17:208–212
Sánchez D (1919) Sobre el desarollo de los elementos nerviosos en la retina del Pieris brassicae L. (Continuación). Trab Lab Invest Biol Univ Madr 17:1–63
Shaw SR (1967) Simultaneous recording from two cells in locust retina. Z vergl Physiol 55:183–194
Shelton PMJ (1976) The development of the insect compound eye. In: Lawrence PA (ed) Insect development. Symp Roy ent Soc 8, Blackwell Scientific Publications, Oxford London
Shelton PMJ, Nowel MS (1981) A polarity marker in the locust retina — non-specific establishment of neuronal connections confirmed. In preparation
Sperry RW (1963) Chemoaffinity in the orderly growth of nerve fibre patterns and connections. Proc Natl Acad Sci USA 50:703–710
Strausfeld NJ (1976) Atlas of an insect brain. Springer Verlag, Heidelberg
Strausfeld NJ, Blest AD (1970) Golgi studies on insects. Part I. The optic lobes of Lepidoptera. Phil Trans Roy Soc Lond B 258:81–223
Thorson J (1964) Dynamics of motion perception in the desert locust. Science 145:69–71
Trujillo-Cenóz O (1965) Some aspects of the structural organization of the intermediate retina of dipterans. J Ultrastruct Res 13:1–33
Tunstall J, Horridge GA (1967) Electrophysiological investigation of the optics of the locust retina. Z vergl Physiol 55:167–182
Wallace GK (1959) Visual scanning in the desert locust Schistocerca gregaria Forskål. J Exp Biol 36:512–525
West RW (1972) Superficial warming of epoxy blocks for cutting of 25–150 μm sections to be resectioned in the 40–90 nm range. Stain Technology 47:201–204
Wilson M, Gerrard P, McGinness S (1978) The unit structure of the locust compound eye. Cell Tissue Res 195:205–226
Author information
Authors and Affiliations
Additional information
MSN was supported in part by a Fulbrights-Hays Scholarsship. We are grateful to the Science Research Council for its grant to PMJS.
Rights and permissions
About this article
Cite this article
Nowel, M.S., Shelton, P.M.J. A Golgi-electron-microscopical study of the structure and development of the lamina ganglionaris of the locust optic lobe. Cell Tissue Res. 216, 377–401 (1981). https://doi.org/10.1007/BF00233626
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00233626