Abstract
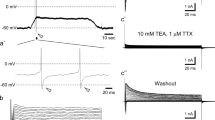
Voltage-dependent sodium currents were analyzed in detail from dissociated mammalian taste receptor cells using the whole-cell patch clamp technique. Approximately 50–75% of all taste receptor cells expressed sodium currents. These currents activated close to −50 mV (holding potential = −80 mV) with maximal currents most often occurring at −10 mV. The distribution of maximal inward currents across all cells appeared to display two peaks, at −254 pA and −477 pA, possibly due to differences in sodium channel density. Inward currents were eliminated by replacing 90% of external sodium with N-methyl-D-glucamine. The currentvoltage relationship of the activated current, as measured by a tail current analysis, was linear, suggesting an ohmic nature of the open channel conductance. The relationship between the time to the peak activated current and the step potential was well fit by a double exponential curve (τ1 = 6.18, τ2 = 37.8 msec). Development of inactivation of the sodium current was dependent upon both voltage- and temporal-parameters. The voltage dependence of the time constant (τ) obtained from removal of inactivation, development of inactivation, and decay of the sodium current displayed a bell-shaped curve with a maximum of 55 msec at −70 mV. In addition to fast inactivation (half maximal at −50 mV), these currents also displayed a slow inactivation (half maximal at −65 mV). Voltage-dependent sodium currents were reversibly inhibited by nanomolar concentrations of tetrodotoxin (Kd = 10−8 m). There was no evidence of a TTX-insensitive sodium current. This description broadens our understanding of gustatory transduction mechanisms with a particular relevance to the physiological role of receptor cell action potentials.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Akabas, M.H., Dodd, J., Al-Awqati, Q. 1990. Identification of electrophysiologically distinct subpopulations of rat taste cells. J. Membrane Biol. 114:71–78
Akisata, T., Oda, M. 1978. Taste buds in the vallate papillae of the rat studied with freeze-fracture preparation. Arch. Histol. Jpn. 41:87–98
Almers, W. 1978. Gating currents and charge movements in excitable membranes. Rev. Physiol. Biochem. Pharmacol. 82:96–190
Aimers, W., Roberts, W.M., Ruff, R.L. 1984. Voltage clamp of rat and human skeletal muscle: Measurements with an improved loose-patch technique. J. Physiol. 347:751–768
Avenet, P., Lindemann, B. 1987. Patch-clamp study of isolated taste receptor cells of the frog. J. Membrane Biol. 97:223–240
Avenet, P., Lindemann, B. 1991. Noninvasive recording of receptor cell action potentials and sustained currents from single taste buds maintained in the tongue: The response to mucosal NaCl and amiloride. J. Membrane Biol. 124:33–41
Baer, M., Best, P.M., Reuter, H. 1976. Voltage-dependent action of tetrodotoxin in mammalian cardiac muscle. Nature 263:344–345
Barres, B.A., Chun, L.L.Y., Corey, D.P. 1989. Glial and neuronal forms of the voltage-dependent sodium channel: Characteristics and cell-type distribution. Neuron 2:1375–1388
Béhé, P., DeSimone, J.A., Avenet, P., Lindemann, B. 1990. Membrane currents in taste cells of the rat fungiform papilla. Evidence for two types of Ca currents and inhibition of K currents by saccharin. J. Gen. Physiol. 96:1061–1084
Beidler, L.M., Smallman, R.L. 1965. Renewal of cells within taste buds. J. Cell Biol. 27:263–272
Benoit, E., Corbier, A., Dubois, J.M. 1985. Evidence for two transient sodium currents in the frog node of Ranvier. J. Physiol. 361:339–360
Cohen, C.J., Beam, B.P., Colatsky, T.J., Tsien, R.W. 1981. Tetrodotoxin block of sodium channels in rabbit Purkinje fibers: Interactions between toxin binding and channel gating. J. Gen. Physiol. 78:383–411
Frelin, C., Vigne, P., Lazdunski, M. 1983. Na+ channels with high and low affinity tetrodotoxin binding sites in the mammalian skeletal muscle cell. Difference in functional properties and sequential appearance during rat skeletal myogenesis. J. Biol. Chem. 258:7256–7259
Gilly, W.F., Armstrong, C.M. 1984. Threshold channels—novel type of sodium channel in squid giant axon. Nature 309:448–50
Gonoi, T., Sherman, S.J., Catterall, W.A. 1985. Voltage clamp analysis of tetrodotoxin-sensitive and-insensitive sodium channels in rat muscle cells developing in vitro. J. Neurosci. 5:2559–2564
Haimovich, B., Tanaka, J.C., Barchi, R.L. 1986. Developmental appearance of sodium channel subtypes in rat skeletal muscle cultures. J. Neurochem. 47:1148–1152
Henderson, R., Ritchie, J.M., Strichartz, G.R. 1974. Evidence that tetrodotoxin and saxitoxin act at a metal cation binding site in the sodium channels of nerve membrane. Proc. Nat. Acad. Sci. USA 71:3936–3940
Herness, M.S. 1989. A dissociation procedure for mammalian taste cells. Neurosci. Lett. 106:60–64
Hille, B. 1992. Ionic channels in excitable membranes. Sinauer, Sunderland, MA
Holland, V.F., Zampighi, G.A., Simon, S.A. 1989. Morphology of fungiform papillae in canine lingual epithelium: Location of intercellular junctions in the epithelium. J. Comp. Neurol. 279:13–27
Howe, J.R., Ritchie, J.M. 1990. Sodium currents in Schwann cells from myelinated and non-myelinated nerves of neonatal and adult rabbits. J. Physiol. 425:169–210
Jones, S.W. 1987. Sodium currents in dissociated bull-frog sympathetic neurones. J. Physiol. 389:605–627
Kashiwayanagi, M., Miyake, M., Kurihara, K. 1983. Voltagedependent Ca2+ channel and Na+ channel in frog taste cells. Am. J. Physiol. 244:C82–88.
Keynes, R., Rojas, E. 1976. The temporal and steady-state relationships between activation of the sodium conductance and movement of the gating particles in the squid giant axon. J. Physiol. 255:157–189
Kinnamon, J.C. 1986. Organization and Innervation of Taste Buds. In: Neurobiology of Taste and Smell, Chap. 12, T.E. Finger and W.L. Silver, editors, pp. 277–297. John Wiley & Sons, New York
Kinnamon, S.C., Cummings, T.A. 1992. Chemosensory transduction mechanisms in taste. Ann. Rev. Physiol. 54:715–731
Kinnamon, S.C., Roper, S.D. 1988. Membrane properties of isolated mudpuppy taste cells. J. Gen. Physiol. 91:351–371
Marty, A., Neher, E. 1983. Tight-seal whole-cell recording. In: Single-Channel Recording, B. Sakmann, & E. Neher, editors, pp. 107–122. Plenum, New York
Matteson, D.R., Armstrong, C.M. 1982. Evidence for a population of sleepy sodium channels in squid axon at low temperature. J. Gen. Physiol. 79:739–758
Miller, I.J. 1971. Peripheral interactions among single papilla inputs to gustatory nerve fibers. J. Gen. Physiol. 57:1–25
Miller, I.J. 1974. Branched chorda tympani neurons and interactions among taste receptors. J. Comp. Neurol. 158:155–156
Murray, R.G. 1986. The mammalian taste bud type III cell: a critical analysis. J. Ultrastrct. Molec. Struct. Res. 95:175–188
Pappone, P.A. 1980. Voltage-clamp experiments in normal and denervated mammalian skeletal muscle fibers. J. Physiol. 306:377–410
Patlak, J., Ortizm, M. 1985. Slow currents through single sodium channels of the adult rat heart. J. Gen. Physiol. 86:89–104
Reutter, K., Witt, M. 1993. Morphology of vertebrate taste organs and their nerve supply. In: Mechanisms of Taste Transduction, Chap. 2, S.A. Simon and S.D. Roper, editors, pp. 29–82. CRC, Boca Raton
Roper, S.D. 1983. Regenerative impulses in taste cells. Science 220:1311–1312
Smith, D.V., Bealer, S.L. 1975. Sensitivity of the rat gustatory system to the rate of stimulus onset. Physiol. & Beh. 15:303–314
Spielman, A.I., Mody, I., Brand, J.G., Whitney, G., MacDonald, J.F., Salter, M.W. 1989. A method for isolating and patch clamping single mammalian taste receptor cells. Brain Res. 503:326–329
Stühmer, W., Methfessel, C., Sakmann, B., Noda, M., Numa, S. 1987. Patch-clamp characterization of sodium channels expressed from rat brain cDNA. Euro. Biophys. J. 14:131–138
Teeter, J. 1985. Dye-coupling in catfish taste buds. In: Proceedings of the 19th Japanese Symposium on Taste and Smell, K. Ueda, editor, pp. 29–33. JASTS, Osaka, Japan
Yang, J., Roper, S. 1987. Dye-coupling in taste buds in the mudpuppy Necturus maculosus. J. Neurosci. 7:3561–3565
Author information
Authors and Affiliations
Additional information
This work was supported by National Institutes of Health grant NIDCD DC00401.
Rights and permissions
About this article
Cite this article
Herness, M.S., Sun, XD. Voltage-dependent sodium currents recorded from dissociated rat taste cells. J. Membarin Biol. 146, 73–84 (1995). https://doi.org/10.1007/BF00232681
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00232681