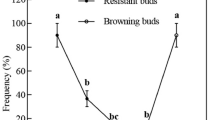
Summary
Transformation of cucumber cv. Endeavor was attempted using three Agrobacterium tumefaciens strains (a supervirulent leucinopine type, an octopine type and a nopaline type), each harbouring one of three binary vectors which contained an acidic chitinase gene from petunia, and basic chitinase genes from tobacco and bean, respectively, driven by the CaMV 35S promoter. Petiole explants were inoculated with a bacterial suspension (108 cells·ml−1), cocultivated for 48–96 h and placed on Murashige and Skoog (MS) medium with 5.0 μM each of 2,4-D and BA, 50 mg·l−1 kanamycin and 500 mg·l−1 carbenicillin. The frequency of embryogenic callus formation ranged from 0 to 12%, depending on strains/vectors used and length of cocultivation, with the highest being obtained using the leucinopine strain with petunia acidic chitinase gene. The kanamycin-resistant embryogenic calli were used to initiate suspension cultures (in liquid MS medium with 1.0/1.0 μM 2,4-D/BA, 50 mg·l−1 kanamycin) for multiplication of embryogenic cell aggregates. Upon plating of cell aggregates onto solid MS medium with 1.0/1.0 μM NAA/BA and 50 mg·l−1 kanamycin, calli continued to grow and later differentiated into plantlets. Transformation by the leucinopine strain and all three vectors was confirmed by PCR amplification of the NPT II gene in transgenic calli and plants, in addition to Southern analysis. Expression of the acidic chitinase gene (from petunia) and both basic chitinase genes (from tobacco and bean) in different transgenic cucumber lines was confirmed by Western analyses.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- 2,4-D:
-
2,4-dichlorophenoxyacetic acid
- BA:
-
6-benzyl-aminopurine
- CaMV:
-
cauliflower mosaic virus
- NAA:
-
naphthaleneacetic acid
- NPT II:
-
neomycin phosphotransferase II
- PCR:
-
polymerase chain reaction
References
An G (1987) Methods in Enzymol. 153:292–305
Boller T, Gehri A, Mauch F, Vogeli U (1983) Planta 157:22–31
Broglie K, Chet I, Holliday M, Chessman R, Biddle P, Knowlton S, Mauvais CJ, Brogue R (1991) Science (Wash.) 254:1194–1197
Broglie R, Brogue K (1993) Phil. Trans. R. Soc. (London) B 342:265–270
Cade RM, Wehner TC, Blazich FA (1990) J. Amer. Soc. Hort. Sci. 115:691–696
Chee P (1990a) Plant Cell Rpt. 9:245–248
Chee PP (1990b) HortSci. 52:792–793
Chee PP, Slightom JL (1991) J. Amer. Soc. Hort. Sci. 116:1098–1102
Chee PP, Tricoli DM (1988) Plant Cell Rpt. 7:274–277
Gambley RL, Dodd WA (1990) Plant Cell Tissue Organ Cult. 20:177–183
Legrand M, Kauffmann S, Geoffrey P, Fritig B (1987) Proc. Nat. Acad. Sci. USA 84:6750–6754
Linthorst HJM, Van Loon LC, Van Rossum CM, Mayer A, Bol JF, Van Roekel JSC, Meulenhoff EJS, Cornelissen BJ (1990) Mol. Plant-Microbe Interact. 3:252–258
Metraux JP (1986) Physiol. Mol. Plant Pathol. 28:161–169
Mettler U (1987) Plant Mol. Biol. Rept. 5:346–349
Murashige T, Skoog F (1962) Physiol. Plant. 15:473–497
Orczyk W, Nadolska-Orczyk A, Malepzy S (1988) Acta. Soc. Bot. Poloniae 57:195–200
Punja ZK, Abbas N, Sarmento GG, Tang FA (1990) Plant Cell Tissue Organ Cult. 21:93–102
Punja ZK, Zhang YY (1993) J. Nematol. 25:526–540
Raharjo SHT, Punja ZK (1994) In Vitro Cell. Dev. Biol. 30P:16–20
Sarmento GG, Alpert K, Tang FA, Punja ZK (1992) Plant Cell Tissue Organ Cult. 31:185–193
Trulson AJ, Shahin EA (1986) Plant Sci. 47:35–43
Turk SCHJ, Melchers LS, den Dulk-Ras H, Regensgurg-Tuink AJG, Hoykaas PJJ (1991) Plant Mol. Biol. 16:1051–1059
Van den Elzen PJM, Jongedijk E, Melchers LS, Cornelissen BJC (1994) Phil. Trans. R. Soc. Lond. B 342:271–278
Zhang YY, Punja ZK (1994) Plant Sci. 99:141–150
Author information
Authors and Affiliations
Additional information
Communicated by S. Gleddie
Rights and permissions
About this article
Cite this article
Raharjo, S.H.T., Hernandez, M.O., Zhang, Y.Y. et al. Transformation of pickling cucumber with chitinase-encoding genes using Agrobacterium tumefaciens . Plant Cell Reports 15, 591–596 (1996). https://doi.org/10.1007/BF00232459
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00232459