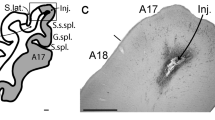
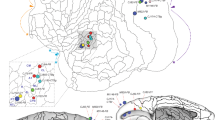
Summary
In the common marmoset (Callithrix jacchus), the cortical projection from the pulvinar and other diencephalic structures into the striate and prestriate cortex was investigated with various fluorescent retrograde tracers. Single cortical injections as well as multiple injections at distances of 1–2 mm with one tracer into an extended but coherent cortical region were applied. Fields with multiple injections were placed so that they touched each other (minimal distances 2 to 3 mm). Retrogradely labelled cells in the LGN and/or the pulvinar were arranged in coherent columns, volumes or slabs, but cell volumes resulting from neighbouring cortical injections overlapped at their border (for details of the thalamo-cortical topography see the companion paper Dick et al. (1991)). Double labelled cells (dl) were only found in the zones of overlap of the cell volumes labelled by the respective tracers. The relative number of dl-cells in these overlap zones was 6.2 ± 3.1%. The dl-frequency was the same in the various nuclei of the pulvinar and the LGN. In the main layers of LGN, dl-cells were found only in the overlap zone of two injection fields into area 17, but a few dl-cells were found in interlaminar cells after injections into area 17 and 18. Maximal cortical distances between injection fields which produced dl in the pulvinar, were 3 to exceptionally 4 mm but dl was highest at injection distances ≤2.5 mm and decreased sharply at wider distances. Such overlap zones were concerned with identical or overlapping regions of visual field representation in the cortex and probably also in the pulvinar. Although in individual experiments up to four different tracers were injected into different striate/prestriate regions, often embracing the same visual field representation, individual cells in the pulvinar showed dl from maximally only two tracers injected into neighbouring cortical regions. We conclude that dl in the posterior thalamic projection nuclei is determined essentially by cortical distance and thus reflects the local domain of branching of thalamo-cortical afferents. Pruning of such branches during development may further restrict bifurcating axons to identical visual field representations, but representation of identical visual field regions in different visual areas is not, per se, a sufficient condition for dl. It is not found if such regions are further apart from each other than the typical local domain of 2–3 mm, exceptionally up to 4 mm in one experiment after injections into area 17 and MT. Dl in the intralaminar nucleus CeL (5.0 ± 4.6%), the claustrum (5.4 ± 3.6%) and in the amygdala (5.7 ± 1.9%) was of the same order as in the pulvinar and LGN. In the hypothalamus around 10% and in the Nucleus basalis Meynert 15.8% of the cells labelled by visual cortical injections were double labelled. In all these extrathalamic regions dl was also restricted to overlap zones, but overlap of labelled fields in these nuclei was much wider and included the whole striate/prestriate cortex except for some topographical separation of striate and prestriate projection zones in the claustrum. Only in the Nucl. basalis Meynert and the hypothalamus some cells were labelled by three tracers.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Allmann JM, Lane RH, Miezin FM (1972) A representation of the visual field in the inferior nucleus of the pulvinar in the owl monkey (Aotus trivirgatus). Brain Res. 40:291–302
Allmann JM, Kaas JH (1975) The dorsomedial cortical visual area: a third tier area in the occipital lobe of the owl monkey (Aotus trivigatus). Brain Res 100:473–487
Aschoff A, Holländer H (1982) Fluorescent compounds as retrograde tracers compared with horseradish peroxydase (HRP). I. A parametric study in the central visual system of the albino rat. JNS Meth 6:179–197
Bender DB (1981) Retinotopic organization of macaque pulvinar. J Neurophysiol 46:672–693
Benedek G, Norita M, Creutzfeldt O (1983) Electrophysiological and anatomical demonstration of an overlapping striate and tectal projection to the lateral posterior-pulvinar complex of cat. Exp Brain Res 52:157–169
Benevento LA, Rezak M (1976) The cortical projection of the inferior pulvinar and adjacent lateral pulvinar in the rhesus monkey (Macaca mulatta): an autoradiographic study. Brain Res 108:1–24
Birnbacher D, Albus K (1987) Divergence of single axons in afferent projections to the cat's visual cortical areas 17, 18 and 19: parametric study. J Comp Neurol 261:543–561
Brandner S, Redies H (1990) The projection from medial geniculate to field A1 in cat: organization in the isofrequency dimension. J Neurosci 10:50–61
Brodmann K (1909) Vergleichende Lokalisationslehre der Großhirnrinde. JA Barth, Leipzig
Brysch I, Creutzfeldt O, Hayes NL, Schlingensiepen KH (1984) The second intralaminar thalamo-cortical projection system. Anat Embryol 169:111–118
Bullier JH, Kennedy H, Salinger W (1984) Bifurcation of subcortical afferents to visual areas 17, 18 and 19 in the cat cortex. J Comp Neurol 228:309–328
Bullier JH, Kennedy, H (1987) Axonal bifurcation in the visual system. TINS (10) 5:205–210
Burton H, Jones EG (1976) The posterior thalamic region and its cortical projection in New World and Old World monkeys. J Comp Neurol 169:249–302
Creutzfeldt OD (1985) Comparative aspects of representation in the visual system. Exp Brain Res Suppl 11:53–81
Dick A, Kaske A, Creutzfeldt OD (1991) Topographical and topological organization of the thalamo-cortical projection to striate and prestriate cortex in the marmoset, Callithrix jacchus. Exp Brain Res 84:233–253
Doty W (1983) Nongeniculate afferents to striate cortex in macaques. J Comp Neurol 218:159–173
Ferster D, LeVay S (1978) The axonal arborizations of lateral geniculate neurons in the striate cortex of cat. J Comp Neurol 182:923–944
Gallyas F (1979) Silver staining of myelin by means of physical development. Neurol Res 1:203–209
Gilbert CD, Wiesel TN (1983) Clustered intrinsic connections in cat visual cortex. J Neurosci 3:1116–1133
Graham J, Lin CS, Kaas JH (1979) Subcortical projections of six visual cortical areas in the owl monkey, Aotus trivirgatus. J Comp Neurol 187:557–580
Graybiel AM, Berson DM (1980) Histochemical identification and afferent connections of subdivisions in the lateralis posteriorpulvinar complex and related thalamic nuclei in the cat. Neuroscience 5:1175–1238
Hubel DH, Wiesel TN, LeVay S (1977) Plasticity of ocular dominance columns in monkey striate cortex. Philos Trans R Soc Lond B. 278:377–409
Jones EG (1985) The thalamus. Plenum Press, New York London
Innocenti GM (1981) Growth and reshaping of axons in the establishment of visual callosal connections. Science 212:824–827
Katz LC, Burkhalter W, Dreyer J (1984) Fluorescent latex microspheres as a retrograd neuronal marker for in vivo and in vitro studies of visual cortex. Nature 310:498–500
Keizer K, Kuypers HGJM, Huisman AM, Dann O (1983) Diamidino yellow dihydrochloride (DY:2HCI): a fluorescent retrograde neuronal tracer which migrates only very slowly out of the cell and can be used in combination with TB and FB in double-labelling experiments. Exp Brain Res 51:179–191
Kennedy H, Bullier J (1985) A double labeling investigation of the afferent connectivity to cortical areas V1 and V2 of the macaque monkey. J Neurosci 10:2815–2830
Kuypers HGJM, Bentivoglio B, Van der Kooy D, Catsman-Berrevoets CE (1979) Retrograde transport of bisbenzimide and propidium iodide through axons to their cell bodies. Neurosci Lett 12:1–7
LeVay S, Sherk H (1981) The visual claustrum of the cat. II. The visual field map. J Neurosci 9:981–992
Lysakowski A, Standage GP, Benevento LA (1988) An investigation of collateral projections of the dorsal lateral geniculate nucleus and other subcortical structures to cortical areas V1 and V4 in the macaque monkey: a double-lable retrograde tracer study. Exp Brain Res 69:651–661
Martin KAC (1986) Neuronal circuits in cat striate cortex. In: Jones EG, Peters A (eds) Cerebral cortex, Vol 2. Plenum Press, New York, pp 241–267
Mason R (1978) Functional organization in the pulvinar complex. Exp Brain Res 31:51–66
Mason R (1981) Differential responsiveness of cells in the visual zones of the cats LP-pulvinar complex to visual stimuli. Exp Brain Res 43:25–33
Parnavelas JG (1986) Physiological properties of identified neurons. In: Jones EG, Peters A (eds) The cerebral cortex, Vol 2. Plenum Press, New York, pp 205–236
Petersen SE, Robinson DL, Keys W (1985) Pulvinar nuclei of the behaving rhesus monkey: visual responses and their modulation. J Neurophysiol 54:867–886
Raczkowski D, Diamond IT (1980) Cortical connections of the pulvinar nucleus in Galago. J Comp Neurol 193:1–40
Raczkowski D, Rosenquist AC (1981) Retinotopic organization in the lateral posterior-pulvinar complex. Brain Res 221:185–191
Redies H, Brandner S, Creutzfeldt OD (1989) Anatomy of the auditory thalamocortical system of the guinea pig. J Comp Neurol 282:489–511
Schmued LC, Fallon JH (1986) Fluoro-gold: a new fluorescent retrograde axonal tracer with numerous unique properties. Brain Res 377:147–154
Spatz WB (1977) Der visuelle Bereich der Großhirnrinde: experimentell-anatomische Untersuchungen zu seiner Gliederung und der ipsilateralen Assoziationsverbindungen bei Callithrix jacchus. Habilitationsschrift. Med. Fakultät, Johann-Wolfgang- Goethe Universität, Frankfurt/Main
Storm-Mathisen J (1970) Quantitative histochemistry of acetylcholineesterase in rat hippocampal region correlated to histochemical staining. J Neurochem 17:739–750
Tanaka M, Lindsley E, Lausmann S, Creutzfeldt OD (1990) Afferent connections of the prelunate visual association cortex (areas V4 and DP). Anat Embryol 181:19–30
Weller RE, Kaas JH (1981) Cortical and subcortical connections of visual cortex in primates. In: Woolsey CN (eds) Cortical sensory organization. 2. Multiple visual areas. Humana Press, Clifton NJ, pp 121–155
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kaske, A., Dick, A. & Creutzfeldt, O.D. The local domain for divergence of subcortical afferents to the striate and extrastriate visual cortex in the common marmoset (Callithrix jacchus): a multiple labelling study. Exp Brain Res 84, 254–265 (1991). https://doi.org/10.1007/BF00231445
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00231445