Abstract
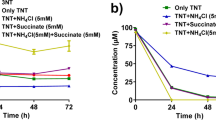
A sulfate-reducing bacterium, Desulfovibrio sp. (B strain) isolated from an anaerobic reactor treating furfural-containing waste-water was studied for its ability to metabolize trinitrotoluene (TNT). The result showed that this isolate could transform 100 ppm TNT within 7 to 10 days of incubation at 37°C, when grown with 30 mm pyruvate as the primary carbon source and 20 mm sulfate as electron acceptor. Under these conditions, the main intermediate produced was 2,4-diamino-6-nitrotoluene. Under culture conditions where TNT served as the sole source of nitrogen for growth with pyruvate as electron donor and sulfate as electron acceptor, TNT was first converted to 2,4-diamino-6-nitrotoluene within 10 days of incubation. This intermediate was further converted to toluene by a reductive deamination process via triaminotoluene. Apart from pyruvate, various other carbon sources such as ethanol, lactate, formate and H2 + CO2 were also studied as potential electron donors for TNT metabolism. The rate of TNT biotransformation by Desulfovibrio sp. (B strain) was compared with other sulfate-reducing bacteria and the results were evaluated. This new strain may be useful in decontaminating TNT-contaminated soil and water under anaerobic conditions in conjunction with toluene-degrading denitrifiers (Pseudomonas spp.) or toluene-degrading sulfate reducers in a mixed culture system.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bak F, Widdel F (1986) Anaerobic degradation of indolic compounds by sulfate reducing enrichment cultures, and description of Desulfobacterium indolicum gen. nov., sp. nov. Arch Microbiol 146:170–176
Balch WE, Wolfe RS (1976) New approach to the cultivation of methanogenic bacteria: 2-mercaptoethane sulfonic acid (HS-CoM) dependent growth of Methanobacterium ruminantium in a pressurized atmosphere. Appl Environ Microbiol 32:781–789
Boopathy R, Daniels L (1991) Isolation and characterization of a furfural degrading sulfate-reducing bacterium from an anaerobic digester. Curr Microbiol 23:327–332
Bradford MM (1977) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Bridge JE, Swanston C, Lane RE, Davis TP (1942) Trinitotoluene poisoning. Proc R Soc Med 35:553–560
Bruhn C, Lenke H, Knackmuss HJ (1987) Nitrosubstituted aromatic compounds as nitrogen source for bacteria. Appl Environ Microbiol 53:208–210
Carpenter DF, McCormick NG, Cornell JH, Kaplan AM (1978) Microbial transformation of 14C-labeled 2,4,6-trinitrotoluene in activated sludge system. Appl Environ Microbiol 35:949–954
Channon HJ, Mills GT, Williams RT (1944) The metabolism of 2,4,6-trinitrotoluene (TNT). Biochem J 38:70–85
Dale JJ (1921) The fate of TNT in the animal body. Med Res Counc (GB) Spec Rep Ser 58:53–61
Daniels L, Belay N, Rajagopal BS (1986) Assimilatory reduction of sulfate and sulfite by methanogenic bacteria. Appl Environ Microbiol 51:703–709
Dickel O, Knackmuss HJ (1991) Catabolism of 1,3-dinitrobenzene by Rhodococcus sp. QT-1. Arch Microbiol 157:76–79
Fernando T, Bumpus JA, Aust SD (1990) Biodegradation of TNT (2,4,6-trinitrotoluene) by Phanerochaete chrysosporium. Appl Environ Microbiol 56:1666–1671
Fewson CA (1981) Biodegradation of aromatics with industrial relevance. In: Leisenger T, Cook AM, Huttler R, Nuesch J (eds) Microbial degradation of xenobiotics and recalcitrant compounds. Academic Press, London, pp 141–179
Haigler BE, Spain JC (1991) Biotransformation of nitrobenzene by bacteria containing toluene degradative pathways. Appl Environ Microbiol 57:3156–3162
Hallas L, Alexander M (1983) Microbial transformation of nitroaromatic compounds in sewage effluents. Appl Environ Microbiol 45:1234–1241
Hamilton A (1921) Trinitrotoluene as an industrial poison. J Ind Hyg 3:102–116
Harvey SD, Fellows RJ, Cataldo DC, Bean RM (1990) Analysis of 2,4,6-trinitrotoluene and its transformation products in soils and plant tissues by high performance liquid chromatography. J Chromatogr 518:361–374
Kaplan DL, Kaplan AM (1982a) Mutagenecity of 2,4,6-trinitrotoluene surfactant complexes. Bull Environ Contam Toxicol 28:33–38
Kaplan DL, Kaplan AM (1982b) Thermophilic biotransformations of 2,4,6-trinitrotoluene under simulated composting conditions. Appl Environ Microbiol 44:757–760
Keither LH, Telliard WA (1979) Priority pollutants. I. A perspective view. Environ Sci Technol 13:416–423
Klausmeier RE, Osmon JL, Walls DR (1974) The effect of trinitrotoluene on microorganisms. Dev Ind Microbiol 15:309–317
Lemberg R, Callaghan JP (1944) Metabolism of symmetrical trinotrotoluene. Nature 154:768
McCormick NG, Feeherry FE, Levinson HS (1976) Microbial transformation of 2,4,6-trinitrotoluene and other nitroaromatic compounds. Appl Environ Microbiol 31:949–958
Naumova RP, Selivanovskay SY, Mingatina FA (1986) Possibility of deep bacterial destruction of 2,4,6-trinitrotoluene. Mikrobiologiya 57:218–222
Nay MW Jr, Randall CW, King PH (1974) Biological treatability of trinitrotoluene manufacturing waste water. J Water Pollut Control Fed 46:485–497
Osmon JL, Klausmeier RE (1972) The microbial degradation of explosives. Dev Ind Microbiol 14:247–252
Pereira WE, Short DL, Manigold DB, Roscio PJ (1979) Isolation and characterization of TNT and its metabolites in ground water by gas chromatograph-mass spectrometer-computer techniques. Bull Environ Contam Toxicol 21:554–562
Sax NI (1963) Dangerous properties of industrial materials, 2nd edn. Reinhold Publishing, New York
Saz AK, Slie RB (1954) The inhibition of organic nitro reductase by aureomycin in cell free extracts. II. Cofactor requirements for the nitro reductase enzyme complex. Arch Biochem Biophys 51:5–16
Schackmann A, Muller R (1991) Reduction of nitroaromatic compounds by different Pseudomonas species under aerobic conditions. Appl Microbiol Biotechnol 34:809–813
Schnell S, Schink B (1991) Anaerobic aniline degradation via reductive deamination of 4-aminobenzoyl-CoA in Desulfobacterium anilini. Arch Microbiol 155:183–190
Schocher RJ, Seyfried B, Vazquez F, Zeyer J (1991) Anaerobic degradation of toluene by pure cultures of denitrifying bacteria. Arch Microbiol 157:7–12
Simpson JR, Evans WC (1953) The metabolism of nitrophenols by certain bacteria. Biochem J 55:24
Spanggord RJ, Spain JC, Nishino SF, Mortelmans KE (1991) Biodegradation of 2,4-dinitrotoluene by a Pseudomonas sp. Appl Environ Microbiol 57:3200–3205
Szewzyk U, Schink B (1989) Degradation of hydroquinone, gentisate and benzoate by a fermenting bacterium in pure or defined mixed culture. Arch Microbiol 151:541–545
Traxler RW, Wood E, Delaney JM (1974) Bacterial degradation of alpha-TNT. Dev Ind Microbiol 16:71–76
Tschech A, Schink B (1988) Methanogenic degradation of anthranilate (2-aminobenzoate). Syst Appl Microbiol 11:9–12
Valli K, Brock BJ, Joshi DK, Gold MH (1992) Degradation of 2.4-dinitrotoluene by the lignin-degrading fungus Phanerochaete chrysosporium. Appl Environ Microbiol 58:221–228
Voegtlin C, Hooper CW, Johnson JM (1919) Trinitrotoluene poisoning. US Public Health Rep. 34:1307–1313
Widdel F (1988) Microbiology and ecology of sulfate or sulfur reducing bacteria. In: Zehnder AJB (ed) Biology of anaerobic microorganisms. Wiley, New York, pp 469–585
Won WD, Heckly RJ, Glover DJ, Hoffsommer JC (1974) Metabolic disposition of 2,4,6-trinitrotoluene. Appl Environ Microbiol 27:513–516
Won WD, Disalvo LH, Ng J (1976) Toxicity and mutagenicity of 2,4,6-trinitrotoluene and its microbial metabolites. Appl Environ Microbiol 31:576–580
Author information
Authors and Affiliations
Additional information
Correspondence to: R. Boopathy
Rights and permissions
About this article
Cite this article
Boopathy, R., Kulpa, C.F. & Wilson, M. Metabolism of 2,4,6-trinitrotoluene (TNT) by Desulfovibrio sp. (B strain). Appl Microbiol Biotechnol 39, 270–275 (1993). https://doi.org/10.1007/BF00228618
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00228618