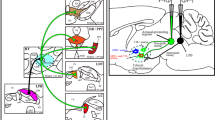
Abstract
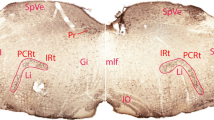
In addition to giving primary projections to the parabrachial and periaqueductal gray regions, ascending lamina I projections course through and terminate in brainstem regions known to contain catecholaminergic cells. For this reason, double-labeling experiments were designed for analysis with light and electron microscopy. The lamina I projections in the Cynomolgus monkey were anterogradely labeled with Phaseolus vulgaris leucoagglutinin (PHA-L) and catecholamine-containing neurons were labeled immunocytochemically for tyrosine hydroxylase (TH). Light level double-labeling experiments revealed that the terminations of the lamina I ascending projections through the medulla and pons strongly overlap with the localization of catecholamine cells in: the entire rostrocaudal extent of the ventrolateral medulla (A1 caudally, C1 rostrally); the solitary nucleus and the dorsomedial medullary reticular formation (A2 caudally, C2 rostrally); the ventrolateral pons (A5); the locus coeruleus (A6); and the subcoerulear region, the Kölliker-Fuse nucleus, and the medial and lateral parabrachial nuclei (A7). At the light microscopic level, close appositions between PHA-L-labeled lamina I terminal varicosities and TH-positive dendrites and somata were observed, particularly in the A1, A5 and the A7 cell groups on the contralateral side. At the electron microscopic level, examples of lamina I terminals were found synapsing on cells of the ventrolateral catecholamine cell groups in preliminary studies. The afferent input relayed by these lamina I projections could provide information about pain, temperature, and metabolic state as described previously. Lamina I input could impact interactions of the catecholamine system with higher brain centers modulating complex autonomic, endocrine, sensory, motor, limbic and cortical functions such as memory and learning. Nociceptive lamina I input to catecholamine cell regions with projections back to the spinal cord could form a feedback loop for control of spinal sensory, autonomic and motor activity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aston-Jones G, Bloom FE (1981) Norepinephrine-containing locus coeruleus neurons in behaving rats exhibit pronounced responses to non-noxious environmental stimuli. J Neurosci 1: 887–900
Aston-Jones G, Ennis M, Peiribone VA, Nickeil WT, Shipley MT (1986) The brain nucleus locus coeruleus: restricted afferent control of a broad efferent network. Science 234: 734–737
Beal JA, Penny JE, Bicknell HR (1981) Structural diversity of marginal (lamina I) neurons in the adult monkey (Macacca mulatto) lumbosacral spinal cord: a golgi study. J Comp Neurol 202: 237–254
Bernard J-F, Dallel R, Raboisson P, Villanueva L, Le Bars D (1995) Organization of the efferent projections from the spinal cervical enlargement to the parabrachial area and periaqueductal gray: a PHA-L study in the rat. J Comp Neurol 353: 480–505
Carlton SM, Honda CN, Denoroy L (1989) Distribution of phenylethanolamine N-methyltransferase cell bodies, axons, and terminals in monkey brainstem: an immunohistochemical mapping study. J Comp Neurol 287: 273–285
Carlton SM, Honda CN, Willcockson WS, Lacrampe M, Zhang D, Denoroy L, Chung JM, Willis WD (1991) Descending adrenergic input to the primate spinal cord and its possible role in modulation of spinothalamic cells. Brain Res 543: 77–90
Cechetto DF, Standaert DG, Saper CB (1985) Spinal and trigeminal dorsal horn projections to the parabrachial nucleus in the rat. J Comp Neurol 240: 153–160
Ciriello J, Caverson MM, Park DH (1986) Immunohistochemical identification of noradrenaline- and adrenaline-synthesizing neurons in the cat ventrolateral medulla. J Comp Neurol 253: 216–230
Clark FM, Proudfit HK (1991) The projection of noradrenergic neurons in the A7 catecholamine cell group to the spinal cord in the rat demonstrated by anterograde tracing combined with immunocytochemistry. Brain Res 547: 279–288
Craig AD (1992) Spinal and trigeminal lamina I input to the locus coeruleus anterogradely labeled with Phaseolus vulgaris leucoagglutinin (PHA-L) in the cat and the monkey. Brain Res 584: 325–328
Craig AD (1995) Distribution of brainstem projections from spinal lamina I neurons in the cat and the monkey. J Comp Neurol 361: 225–248
Craig AD, Kniffki K-D (1985) Spinothalamic lumbosacral lamina I cells responsive to skin and muscle stimulation in the cat. J Physiol (Lond) 365: 197–221
Dahlström A, Fuxe K (1965) Evidence for the existence of monoamine neurons in the central nervous system. I. Demonstration of monoamines in the cell bodies of brain stem neurons. Acta Physiol Scand 62: 1–36
Doyle CA, Maxwell DJ (1991) Ultrastructural analysis of noradrenergic nerve terminals in the cat lumbosacral spinal dorsal horn: a dopamine-β-hydroxylase immunocytochemical study. Brain Res 563: 329–333
Duggan AW, North RA (1984) Electrophysiology of opioids. Pharmacol Rev 35: 219–281
Fallon JH, Koziel DA, Moore RY (1978) Catecholamine innervation of the basal forebrain. II. Amygdala, suprarhinal cortex and entorhinal cortex. J Comp Neurol 180: 509–532
Feil K, Herbert H (1995) Topographic organization of spinal and trigeminal somatosensory pathways to the rat parabrachial and Kölliker-Fuse nuclei. J Comp Neurol 353: 506–528
Felten DL, Sladek JR Jr (1983) Monoamine distribution in primate brain. V. Monoaminergic nuclei: anatomy, pathways and local organization. Brain Res Bull 10: 171–284
Fleetwood-Walker SM, Hope PJ, Iggo A, Mitchell R, Molony V (1983) The effects of iontophoretically applied noradrenaline on the cutaneous sensory responses of identified dorsal horn neurones in the cat. J Physiol (Lond) 342: 63–64
Foote SL, Bloom FE, Aston-Jones G (1983) Nucleus locus coeruleus: new evidence of anatomical and physiological specificity. Physiol Rev 63: 844–914
Foote SL, Berridge CW, Adams LM, Pineda JA (1991) Electrophysiological evidence for the involvement of the locus coeruleus in alerting, orienting, and attending. Prog Brain Res 88: 521–532
Fung SJ, Manzoni D, Chan JYH, Pompeiano O, Barnes CD (1991) Eocus coeruleus control of spinal motor output. Prog Brain Res 88: 395–409
Ginsberg SD, Hof PR, Young WG, Morrison JH (1993) Noradrenergic innervation of the hypothalamus of rhesus monkeys: immunoreactive fibers and quantitative analysis of varicosities in the paraventricular nucleus. J Comp Neurol 327: 597–611
Hancock MB (1986) Two-color immunoperoxidase staining: visualization of anatomic relationships between immunoreactive neural elements. Am J Anat 175: 343–352
Headley PM, Duggan AW, Griersmith BT (1978) Selective reduction by noradrenaline and 5-hydroxytryptamine of nociceptive responses of cat dorsal horn neurones. Brain Res 145: 185–189
Hodge CJ, Apkarian AV, Stevens RT (1986) Inhibition of dorsalhorn cell responses by stimulation of the Kölliker-Fuse nucleus. J Neurosurg 65: 825–833
Hökfelt T, Johansson O, Goldstein M(1984) Central catecholamine neurons as revealed by immunohistochemistry with special reference to adrenaline neurons In: Björklund A, Hökfelt T (eds) Handbook of chemical neuroanatomy, vol 2. Elsevier, New York, pp 157–276
Holstege G, Kuypers HGJM (1982) The anatomy of brain stem pathways to the spinal cord in cat: a labeled amino acid tracing study. Prog Brain Res 57: 145–175
Hudson ME, Fuxe K, Goldstein M, Kalia M (1986) Spinal projections of noradrenergic and adrenergic neurons in the medulla oblongata: new evidence in central cardiovascular control. Soc Neurosci Abstr 12: 535
Hylden JLK, Hayashi H, Bennett GJ (1986) Lamina I spinomesencephalic neurons in the cat ascend via the dorsolateral funiculi. Somatosensory Res 4: 31–41
Jones SL, Gebhart GF (1988) Inhibition of spinal nociceptive transmission from the midbrain, pons and medulla in the rat: activation of descending inhibition by morphine, glutamate and electrical stimulation. Brain Res 460: 281–296
Jouvet M (1972) The role of monoamines and acetylcholine-containing neurons in the regulation of the sleep-waking cycle. Ergeb Physiol 64: 166–307
Kitamura T, Yamada J, Sato H, Yamashita K (1993) Cells of origin of the spinoparabrachial fibers in the rat: a study with fast blue and WGA-HRP. J Comp Neurol 328: 449–461
Kuraishi Y, Hirota N, Sato Y, Kaneko S, Satoh M, Takagi H (1985) Noradrenergic inhibition of the release of substance P from the primary afferents in the rabbit spinal dorsal horn. Brain Res 359: 177–182
Lackner KJ (1980) Mapping of monoamine neurones and fibres in the cat lower brainstem and spinal cord. Anat Embryol 161: 169–195
Lima D, Coimbra A (1986) A golgi study of the neuronal population of the marginal zone (lamina I) of the rat spinal cord. J Comp Neurol 244: 53–71
Lima D, Mendes-Ribeiro JA, Coimbra A (1991) The spino-lateroreticular system of the rat: projections from the superficial dorsal horn and structural characterization of marginal neurons involved. Neuroscience 45: 137–152
Loewy AD, Spyer KM (1990) Central regulation of autonomic functions. Oxford University Press, New York
Maeda T, Pin C, Salvert D, Ligier M, Jouvet M (1973) Les neurones contenant des catecholamines du tegmentum pontique et leurs voies de projection chez le chat. Brain Res 57: 119–152
Mehler WR, Feferman ME, Nauta WJH (1960) Ascending axon degeneration following anterolateral cordotomy: an experimental study in the monkey. Brain 83: 718–750
Menétrey D, Basbaum AI (1987) Spinal and trigeminal projections to the nucleus of the solitary tract: a possible substrate for somatovisceral and viscerovisceral reflex activation. J Comp Neurol 255: 439–450
Panksepp J, Jaloweic JE, Morgane PJ, Zolovick AJ, Stern WC (1973) Noradrenergic pathways and sleep-waking states in cats. Exp Neurol 41: 233–245
Petrov T, Krukoff TL, Jhamandas JH (1993) Branching projections of catecholaminergic brainstem neurons to the paraventricular hypothalamic nucleus and the central nucleus of the amygdala in the rat. Brain Res 609: 81–92
Plotsky PM, Cunningham ET, Widmaier EP (1989) Catecholaminergic modulation of corticotropin-releasing factor role in adrenocorticotropin secretion. Endocr Rev 10: 437–458
Proudfit HK (1992) The behavioral pharmacology of the noradrenergic descending system. In: Besson J-M, Guilbaud G (eds) Towards the use of noradrenergic agonists for the treatment of pain. Elsevier, New York, pp 119–136
Rajaofetra N, Ridet J-L, Poulat P, Marlier L, Sandillon F, Geffard M, Privat A (1992) Immunocytochemical mapping of noradrenergic projections to the rat spinal cord with an antiserum against noradrenaline. J Neurocytol 21: 481–494
Ross CA, Ruggiero DA, Joh TH, Park DH, Reis DJ (1984) Rostral ventrolateral medulla: selective projections to the thoracic autonomic cell column from the region containing C1 adrenaline neurons. J Comp Neurol 228: 168–185
Saper CB, Levisohn D (1983) Afferent connections of the median preoptic nucleus in the rat: anatomical evidence for a cardiovascular integrative mechanism in the anteroventral third ventricular. Brain Res 288: 21–31
Sawchenko PE, Swanson LW (1982) The organization of noradrenergic pathways from the brainstem to the paraventricular and supraoptic nuclei in the rat. Brain Res 257: 275–325
Schachner M, Hedley-Whyte ET, Hsu DW, Schoonmaker G, Bignami A (1977) Ultrastructural localization of glial fibrillary acidic protein in mouse cerebellum by immunoperoxidase labeling. J Cell Biol 75: 67–73
Schofield SPM, Everitt BJ (1981) The organisation of catecholamine-containing neurons in the brain of the rhesus monkey (Macaca mulatta). J Anat 132: 391–418
Segal M, Markram H, Richter-Levin G (1991) Actions of norepinephrine in the rat hippocampus. Prog Brain Res 88: 323–330
Slugg RM, Light AR (1994) Spinal cord and trigeminal projections to the pontine parabrachial region in the rat as demonstrated with Phaseolus vulgaris leucoagglutinin. J Comp Neurol 339: 49–61
Standaert DG, Watson SJ, Houghten RA, Saper CB (1986) Opioid peptide immunoreactivity in spinal and trigeminal dorsal horn neurons projecting to the parabrachial nucleus in the rat. J Neurosci 6: 1220–1226
Stevens RT, Hodge CJ Jr, Apkarian AV (1982) Kölliker-Fuse nucleus: the principal source of pontine catecholaminergic cells projecting to the lumbar spinal cord of cat. Brain Res 239: 589–594
Tetal MJ, Blaustein JD (1991) Immunocytochemical evidence for noradrenergic regulation of estrogen receptor concentrations in the guinea pig hypothalamus. Brain Res 565: 321–329
Tucker DC, Saper CB, Ruggiero DA, Reis DJ (1987) Organization of central adrenergic pathways: I. Relationships of ventrolateral medullary projections to the hypothalamus and spinal cord. J Comp Neurol 259: 591–603
Ungerstedt U (1971) Stereotaxic mapping of the monoamine pathways in the rat brain. Acta Physiol Scand Suppl 367: 1–48
Weinberg RJ, Van Eyck SL (1991) A tetramethylbenzidine/tungstate reaction for horseradish peroxidase histochemistry. J Histochem Cytochem 39: 1143–1148
Westlund KN, Coulter JD (1980) Descending projection of the locus coeruleus and subcoeruleus/medial parabrachial nuclei in monkey: axonal transport studies and dopamine-beta-hydroxylase immunochemistry. Brain Res Rev 2: 235–264
Westlund KN, Craig AD (1993) Spinal cord lamina I terminations in brainstem catecholamine cell groups. Soc Neurosci Abstr 19: 517
Westlund KN, Bowker RM, Ziegler MG, Coulter JD (1983) Noradrenergic projections to the spinal cord of the rat. Brain Res 263: 15–31
Westlund KN, Bowker RM, Ziegler MG, Coulter JD (1984) Origins and terminations of descending noradrenergic projections to the spinal cord of monkey. Brain Res 292: 1–16
Westlund KN, Carlton SM, Zhang D, Willis WD (1990) Direct catecholaminergic innervation of primate spinothalamic tract neurons. J Comp Neurol 299: 178–186
Wiberg M, Westman J, Blomqvist A (1987) Somatosensory projection to the mesencephalon: an anatomical study in the monkey. J Comp Neurol 264: 92–117
Willcockson WS, Chung JM, Hori Y, Lee KH, Willis WD (1984) Effects of iontophoretically released amino acids and amines on primate spinothalamic tract cells. J Neurosci 4: 732–740
Yaksh TL (1986) The effects of intrathecally administered opioid and adrenergic agents on spinal function. In Yaksh TL (ed) Spinal afferent processing. Plenum Press, New York, pp 505–540
Yeomans DC, Proudfit HK (1992) Antinociception induced by microinjection of substance P into the A7 catecholamine cell group in the rat. Neuroscience 49: 681–691
Zhang E-T, Han Z-S, Craig AD (1993) Morphological types of retrogradely labeled lamina I spinothalamic neurons in the cat. Soc Neurosci Abstr 19: 1571
Zhao Z-Q, Duggan AW (1988) Idazoxan blocks the action of noradrenaline but not spinal inhibition from electrical stimulation of the locus coeruleus and nucleus Kölliker-Fuse of the cat. Neuroscience 25: 997–1005
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Westlund, K.N., Craig, A.D. Association of spinal lamina I projections with brainstem catecholamine neurons in the monkey. Exp Brain Res 110, 151–162 (1996). https://doi.org/10.1007/BF00228547
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00228547