Summary
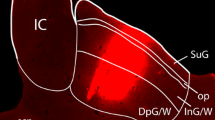
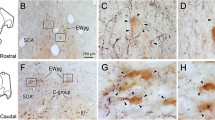
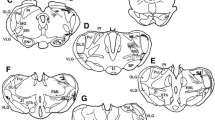
Electrical stimulation of the optic tectum in many vertebrate species elicits eye, head or body orienting movements in the direction of the receptive field location recorded at the site of stimulation; in the barn owl, tectal stimulation produces short latency saccadic head movements (du Lac and Knudsen 1990). However, the barn owl, like other avians, lacks a direct projection from the tectum to the spinal cord, implying that less direct connections underlie tectally mediated head movements. In order to determine the pathways by which the tectum gains access to spinal cord circuitry, we searched for overlap regions between tectal efferent projections and the locations of cells afferent to the spinal cord. Tectal efferent pathways and terminal fields were revealed by anterograde labeling using horseradish peroxidase (HRP) or tritiated amino acids injected into the optic tectum. Cells afferent to the spinal cord were identified by means of retrograde labeling using HRP, rhodamine, or rhodamine-coupled latex beads injected into the cervical spinal cord. A comparison of results from the anterograde and retrograde labeling experiments demonstrated several areas of overlap. All of the cell groups that both received heavy tectal input and contained a high proportion of cells projecting to the spinal cord were located in the medial half of the midbrain and rhombencephalic tegmentum, and included the red nucleus, the interstitial nucleus of Cajal, the medial reticular formation, the nucleus reticularis pontis gigantocellularis, and the nucleus reticularis pontis oralis. All of these cell groups receive their tectal input from the medial efferent pathway, one of three major output pathways from the tectum. The other two output pathways (the rostral and the caudal) project to regions containing no more than a few scattered cells that are afferent to the spinal cord. Based on these data and on the functions of homologous cell groups in other vertebrates, we hypothesize that the medial efferent pathway and its brainstem target nuclei are primarily responsible for tectally mediated orienting head movements in the barn owl.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ariens-Kappers C, Huber GC, Crosby EC (1936) The comparative anatomy of the nervous system of vertebrates including man. Macmillan, New York
Benowitz LI, Karten H (1976) Organization of tectofugal pathways in the pigeon: a retrograde transport study. J Comp Neurol 167:503–520
Butler AB, Northcutt RG (1980) Projections of the optic tectum in the longnose gar, Lepisosteus osseus. Brain Res 190:333–346
Büttner U, Büttner-Ennever JA, Henn V (1977) Vertical eye movement-related activity in the rostral mesencephalic reticular formation of the alert monkey. Brain Res 130:239–252
Cabot JB, Reiner A, Bogan N (1982) Avian bulbospinal pathways: anterograde and retrograde studies of cells of origin, funicular trajectories, and laminar terminations. Prog Brain Res 57:79–107
Craigie EH (1928) Observations on the brain of the humming bird (Chrysolampis mosquitus Linn and Chlorostilbon caribaeus Lawr) J Comp Neurol 45:377–481
Dacey DM, Ulinski PS (1986) Optic tectum of the eastern garter snake, Thamnophis sirtalis. I. Efferent pathways. J Comp Neurol 245:1–28
Dean P, Redgrave P, Sahibzada N, Tsuji K (1986) Head and body movements produced by electrical stimulation of superior colliculus in rats: effects of interruption of crossed tectoreticulospinal pathway. Neuroscience 19:367–380
Ebbesson SOE, Vanegas H (1976) Projections of the optic tectum in two teleost species. J Comp Neurol 165:161–180
Fuchs AF, Kaneko CRS, Scudder CA (1985) Brainstem control of saccadic eye movements. Ann Rev Neurosci 8:307–338
Grantyn A, Berthoz A (1987) Reticulo-spinal neurons participating in the control of synergic eye and head movements during orienting in the cat. I. Behavioral properties. Exp Brain Res66:339–354
Grantyn A, Grantyn R (1982) Axonal patterns and sites of termination of cat superior colliculus neurons projecting in the tectobulbar spinal tract. Exp Brain Res 46:243–256
Grantyn A, Grantyn R, Berthoz A, Ribas J (1982) Tectal control of vertical eye movements: a search for the underlying circuits in the mesecephalon. In: Roucoux A, Crommelinck M (eds): Physiological and pathological aspects of eye movements. Junk, The Hague, pp 337–344
Graybiel AM (1978) A satellite system of the superior colliculus: the parabigeminal nucleus and its projections to the superficial collicular layers. Brain Res 145:365–374
Grobstein P (1988) Between the retinotectal projection and directed movement: topography of a sensorimotor interface. Brain Behav Evol 31:34–48
Grobstein P (1989) Organization in the sensorimotor interface: a case study with increased resolution. In: Ewert J-P, Arbib MA (eds) Visuomotor coordination: amphibians, comparisons, models and robots. Plenum, New York, 537–563
Grobstein P (1990) Strategies for analyzing complex organization in the nervous system. I. Lesion experiments, the old rediscovered. In: E Schwartz (ed) Computational Neuroscience MIT Press, Cambridge, pp 245–255
Grobstein P, Comer C (1983) The nucleus isthmi as an intertectal relay for the ipsilateral oculotectal projection in the frog, Rana pipiens. J Comp Neurol 217:54–74
Harting JK (1977) Descending pathways from the superior colliculus: an autoradiographic analysis in the rhesus monkey (Macaca, mulatta). J Comp Neurol 173:583–612
Hinsey JC, Ransom SW, Dixon HH (1930) Responses elicited by stimulation of the mesenephalic tegmentum in the cat. Arch Neurol Psychiat 24:966–977
Holcombe V, Hall WC (1981b) The laminar organization and distribution of the crossed tectoreticular pathways. J Neurosci 1:1103–1112
Holstege G, Crowie RJ (1989) Projections from the rostral mesencephalic reticular formation to the spinal cord: an HRP and autoradiographic tracing study in the cat. Exp Brain Res 75:265–279
Huerta MF, Harting JK (1982) Tectal control of spinal cord activity: neuroanatomical demonstration of pathways connecting the superior colliculus with the cervical spinal cord grey. Prog Brain Res 57:293–328
Hunt SP, Kunzle H (1976) Observation on the projections and intrinsic organization of the pigeon optic tectum: an autoradiographic study based on anterograde and retrograde, axonal and dendritic flow. J Comp Neurol 170:153–172
Ingram WR, Ransom SW, Hannet FI, Zeiss FR, Terwilliger EH (1932) Results of stimulation of the tegmentum with the Horsely-Clarke stereotaxic apparatus. Arch Neurol Psychiat 28:513–541
Jungherr E (1945) Certain nuclear groups in the avian mesencephalon. J Comp Neurol 82:55–75
Karten HJ, Hodos W (1967) A stereotaxic atlas of the brain of pigeon (Columbia livia). Johns Hopkins Press, Baltimore
du Lac S, Knudsen EI (1990) Neural maps of head movement vector and speed in the optic tectum of the barn owl. J Neurophysiol 63:136–146
Lane JK (1978) A protocol for horseradish peroxidase histochemistry as practised in the lab of E. G. Jones In: Neuroanatomical techniques. (Society of Neuroscience Monograph)
Lawrence DG, Kuypers HG (1968) The functional organization of the motor system n the monkey. II. The effects of lesions of the descending brain-stem pathways. Brain 91:15–36
Masino T (1992) Brainstem control of orienting movements: Intrinsic coordinate systems and underlying circuitry. Brain Behav Evol 40:98–111
Masino T, Knudsen EI (1990) Distinct neural circuits control horizontal and vertical components of head movement in the barn owl. Nature 345:434–437
Masino T, Knudsen EI (1992) Orienting head movements resulting from electrical microstimulation of the brainstem tegmentum in the barn owl. J Neurosci (in press)
Masino T, Grobstein P (1989a) The organization of descending tectofugal pathways in frog, Rana pipiens. 1. Lateralization, parcellation and an intermediate spatial representation. Exp Brain Res 75:227–244
Masino T, Grobstein P (1989b) The organization of the descending tectofugal pathways in the frog, Rana pipiens. 2. Evidence for the involvement of a tecto-tegmento-spinal pathway. Exp Brain Res 75:245–264
Masino T, Grobstein P (1989c) Tectal connectivity in the frog, Rana pipiens: tectotegmental projections and a general analysis of topographic organization. J Comp Neurol 291:103–127
Mesulum MM (1975) The blue reaction product in horseradish peroxidase neurohistochemistry: incubation parameters and visibility. J Histochem Cytochem 24:1273–1280
Moschovakis AK, Karabalas AB (1985) Observations on the somatodendritic morphology and axonal trajectory of intracellularly HRP labelled efferent neurons located in the deepest layers of the superior colliculus of the cat. J Comp Neurol 239:276–308
Ramon y Cajal S (1972) Histologie du system nerveaux de l'homme et des vertebrés, 2nd edn, vol II. Instituto Ramon y Cajal, Madrid, pp. 196–212
Raphan T, Cohen B (1978) Brainstem mechanisms for rapid and slow eye movements. J Neurophysiol 34:920–936
Reiner A, Karten HJ (1982) Laminar distribution of the cells of origin of the descending pathways in the pigeon (Columba livia). J Comp Neurol 201:165–187
Robinson DA (1972) Eye movements evoked by collicular stimulation in the alert monkey. Vision Res 12:1795–1808
Roucoux A, Guitton D, Crommelink M (1980) Stimulation of the superior colliculus in alert cat. II. Eye and head movements evoked when the head is unrestrained. Exp Brain Res 39:75–85
Takahashi TT, Konishi M (1988) Projections of nucleus angularis and nucleus laminaris to the lateral lemniscal nuclear complex of the barn owl. J Comp Neurol 274:212–238
Tehovnik EJ, Yeomans JS (1986) Two converging brainstem pathways mediating circling behavior. Brain Res 385:329–342
Webster DMS, Steeves JD (1988) Origins of brainstem-spinal projection in the duck and goose. J Comp Neurol 273:573–583
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Masino, T., Knudsen, E.I. Anatomical pathways from the optic tectum to the spinal cord subserving orienting movements in the barn owl. Exp Brain Res 92, 194–208 (1992). https://doi.org/10.1007/BF00227965
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00227965