Abstract
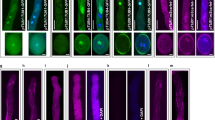
The behavior and role of the microtubule (MT) and actin-myosin components of the cytoskeleton during pollen tube growth in two species of Pinus were studied using anti-α-tubulin, rhodamine-phalloidin, anti-myosin, and the appropriate inhibitors. Within germinated pollen tubes MTs were arranged obliquely or transversely, but in elongated tubes they were arranged along the tube's long axis. MTs were localized in the tube tip region, excluding the basal part. Altered growth was found in pollen tubes treated with colchicine; the tips of many pollen tubes incubated in the liquid medium were branched and/or rounded, and those in the agar medium were divided into many branches. Both the branching and the rounding were considered to be caused by the disturbance of polarizing growth of the tube due to MT disorganization with colchicine treatment. Actin filaments (F-actin) were found in the major parts of many pollen tubes along their long axis, excluding the tip region. In a few tubes, however, F-actin was distributed throughout the tube. The areas in the pollen tube containing F-actin were filled with abundant cytoplasmic granules, but the areas without F-actin had very few granules. The tube nucleus, which migrated from the grain area into the tube, was closely associated with F-actin. Germination of pollen grains treated with cytochalasin B was little affected, but further tube elongation was inhibited. Myosin was identified on cytoplasmic granules and to a lesser extent on the tube nucleus in the pollen tubes. Several granules were attached to the nuclear envelope. Tube growth was completely inhibited by N-ethylmaleimide treatment. In generative cells that were retained in the pollen grain, both MT and F-actin networks were observed. Myosin was localized on the cytoplasmic granules but not on the cell surface. In conclusion, it was shown that actin-myosin and MTs were present in gymnospermous Pinus pollen tubes and it is suggested that the former contributed to outgrowth of the tubes and the latter contributed to polarized growth. Several differences in the behavior of cytoskeletal elements in generative cells compared to angiosperms were revealed and are discussed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Åström H, Virtanen I, Raudaskoski M (1991) Cold-stability in the pollen tube cytoskeleton. Protoplasma 160:99–107
Brewbaker J, Kwack BH (1963) The essential role of calcium ion in pollen germination and pollen tube growth. Am J Bot 50:859–865
Cai G, Bartalesi A, Moscatelli A, Casino CD, Tiezzi A, Cresti M (1992) Microtubular motors in the pollen tube of Nicotiana tabacum. In: Ottaviano E, Mulcahy DL, Gorla MS, Mulcahy GB (eds) Angiosperm pollen and ovules. Springer, New York Berlin Heidelberg, pp 218–223
Derksen J, Traas JA (1984) Growth of tobacco pollen tubes in vitro: effects of drug interference with the cytoskeleton. In: Willemse MTM, Van Went JL (eds) Proceedings of the 8th International Symposium on Sexual Reproduction in Seed Plants, Ferns and Mosses, 22–24 August 1984. Pudoc, Wageningen, The Netherlands, pp 64–70
Doonan JH, Cove DJ, Lloyd CW (1988) Microtubules and microfilaments in tip growth: evidence that micro tubules impose polarity on protonemal growth in Physcomitrella patens. J Cell Sci 89:533–540
Franke WW, Herth W, Van der Woude WJ, Morré DJ (1972) Tubular and filamentous structures in pollen tubes: possible involvement as guide elements in protoplasmic streaming and vectorial migration of secretory vesicles. Planta 105:317–341
Heslop-Harrison J, Heslop-Harrison Y (1989) Myosin associated with the surfaces of organelles, vegetative nuclei and generative cells in angiosperm pollen grains and tubes. J Cell Sci 94:319–325
Heslop-Harrison J, Heslop-Harrison Y, Cresti M, Tiezzi A, Moscatelli A (1988) Cytoskeletal elements, cell shaping and movement in the angiosperm pollen tube. J Cell Sci 91:49–60
Kohno T, Shimmen T (1987) Ca2+-induced fragmentation of actin filaments in pollen tubes. Protoplasma 141:177–179
Kohno T, Shimmen T (1988) Accelerated sliding of pollen tube organelles along Characeae actin bundles regulated by Ca2+. J Cell Biol 106:1539–1543
Kohno T, Chaen S, Shimmen T (1990) Characterization of the translocator associated with pollen tube organelles. Protoplasma 154:179–183
Kohno T, Okagaki T, Kohama K, Shimmen T (1991) Pollen tube extract supports the movement of actin filaments in vitro. Protoplasma 161:75–77
Lancelle SA, Cresti M, Hepler PK (1987) Ultrastructure of the cytoskeleton in freeze-substituted pollen tubes of Nicotiana alata. Protoplasma 140:141–150
Mascarenhas JP, Lafountain J (1972) Protoplasmic streaming, cytochalasin B, and growth of the pollen tube. Tissue Cell 4:11–14
Moscatelli A, Tiezzi A, Vignani R, Cai G, Bartalesi A, Cresti M (1988) Presence of kinesin in tobacco pollen tube. In: Cresti M, Gori P, Pacini E (eds) Sexual reproduction in higher plants. Springer, Berlin Heidelberg New York, pp 205–209
Palevitz BA, Cresti M (1989) Cytoskeletal changes during generative cell division and sperm formation in Tradescantia virginiana. Protoplasma 150:54–71
Palevitz BA, Liu B (1992) Microfilaments (F-actin) in generative cells and sperm: an evaluation. Sex Plant Reprod 5:89–100
Perdue TD, Parthasarathy MV (1985) In situ localization of F-actin in pollen tubes. Eur J Cell Biol 39:13–20
Picton JM, Steer MW (1983) Evidence for the role of Ca2+ ions in tip extension in pollen tubes. Protoplasma 115:11–17
Pierson ES, Derksen J, Traas JA (1986) Organization of microfilaments and microtubules in pollen tubes grown in vitro or in vivo in various angiosperms. Eur J Cell Biol 41:14–18
Pierson ES, Kengen HMP, Derksen J (1989) Microtubules and actin filaments co-localize in pollen tubes of Nicotiana tabacum L. and Lilium longiflorum Thunb. Protoplasma 150:75–77
Rosen WG, Gawlik SR, Dashek WV, Siegesmund KA (1964) Fine structure and cytochemistry of Lilium pollen tubes. Am J Bot 51:61–71
Tang X, Hepler PK, Scordilis SP (1989) Immunochemical and immunocytochemical identification of a myosin heavy chain polypeptide in Nicotiana pollen tubes. J Cell Sci 92:569–574
Terasaka O, Niitsu T (1990) Unequal cell division and chromatin differentiation in pollen grain cells. II. Microtubule dynamics associated with the unequal cell division. Bot Mag Tokyo 103:133–142
Terasaka O, Niitsu T (1992) Actin dynamics during pollen ontogeny of Tradescantia paludosa. Jpn J Palynol 38:37–49
Tiezzi A, Moscatelli A, Milanesi C, Ciampolini F, Cresti M (1987) Taxol-induced structures derived from cytoskeletal elements of the Nicotiana pollen tube. J Cell Sci 88:657–661
Tiwari SC, Polito VS (1988) Organization of the cytoskeleton in pollen tubes of Pyrus communis: a study employing conventional and freeze-substitution electron microscopy, immunofluorescence, and rhodamine-phalloidin. Protoplasma 147:100–112
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Terasaka, O., Niitsu, T. Differential roles of microtubule and actin-myosin cytoskeleton in the growth of Pinus pollen tubes. Sexual Plant Reprod 7, 264–272 (1994). https://doi.org/10.1007/BF00227708
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00227708