Abstract
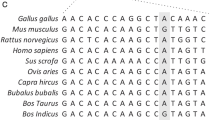
For the detection of polymorphisms within the 5′-flanking region of the β-lactoglobulin (-LG) -encoding gene a nucleotide sequence containing 795 bp of the promoter and 59 bp of exon I was cloned and sequenced. After comparing the sequence from the DNA of 11 diverse cows (different breeds and milk-protein yields), 14 singlebp substitutions were identified within the 5′-flanking region and two in the 5′-untranslated region (5′-UTR) of exon I. Some of the variants are located in potential binding sites for trans-acting factors or in the 5′-UTR. A PCR-based RFLP analysis was performed, and the genotypes of an additional 60 cows were identified at five variable 5′-flanking sites. The results reveal three frequent combinations between the A and B alleles of the protein-coding region and the novel 5′-flanking DNA variants. This finding may explain the differences of the protein-variant-dependent β-LG synthesis (A>B) observed in vivo. A sequence comparison of the bovine and ovine promoters reveals an homology of 92.8% and shows a higher degree of conservation between positions -600 and -300.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adan RAH, Cox JJ, van Kats JP, Burbach JPH (1992) Thyroid hormone regulates the oxytocin gene. J Biol Chem 267:3771–3777
Aleandri R, Buttazzoni LG, Schneider JC, Caroli A, Davoli R (1990) The effects of milk protein polymorphisms on milk components and cheese-producing ability. J Dairy Sci 73:241–255
Alexander LJ, Hayes G, Pearse MJ, Beattie CW, Stewart AF, Willis IM, Mackinlay AG (1989) Complete sequence of bovine β-lactoglobulin cDNA. Nucleic Acids Res 17:6739
Bailly A, Le Page C, Rauch M, Milgrom E (1986) Sequence-specific DNA binding of the progesterone receptor to the uteroglobin gene: effects of hormone, antihormone and receptor phosphorylation. EMBO J 5:3235–3241
Beato M (1989) Gene regulation by steroid hormones. Cell 56:335–344
Bech A-M, Kristiansen KR (1990) Milk protein polymorphism in Danish dairy cattle and the influence of genetic variants on milk yield. J Dairy Res 57:53–62
Bleck GT, Bremel RD (1993) Sequence and single-base polymorphisms of the bovine α-lactalbumin 5′-flanking region. Gene 126:213–218
Chiu R, Imagawa M, Imbra RJ, Bockoven JR, Karin M (1987) Multiple cis- and trans-acting elements mediate the transcriptional response to phorbol esters. Nature 329:648–651
Dean C, Brian J, Riser ME, O'Malley BW (1983) A 5′-flanking sequence essential for progesterone regulation of an ovalbumin fusion gene. Nature 305:551–554
De Lange DJ, Meyer EHH, van Rensburg L, Swanepoel A (1990) The polymorphic protein system in cow's milk. I. The association with the production of milk protein and fat during lactation. S Afr J Dairy Sci 22:67–71
Eigel WN, Butler JE, Ernstrom CA, Farrell jr HM, Harwalkar VR, Jenness R, Whitney R Mcl (1984) Nomenclature of proteins of cow's milk: fifth revision. J Dairy Sci 67:1599–1631
Graml R, Weiss G, Buchberger J, Pirchner F (1989) Different rates of synthesis of whey protein and casein by alleles of the β-lactoglobulin and α S1-casein locus in cattle. Genet Sel Evol 21:547–554
Grant DJ, Maeda N (1993) A base substitution in the promoter associated with the human haptoglobin 2-I-modified phenotype decreases transcriptional activity and responsiveness to interleukin-6 in human hepatoma cells. Am J Hum Genet 52:974–980
Groenen MAM, Dijkhof RJM, van der Poel JJ, van Diggelen R, Verstege E (1992) Multiple octamer binding sites in the promoter region of the bovine α S2-casein gene. Nucleic Acids Res 20:4311–4318
Harris S, Ali S, Anderson S, Archibald AL, Clark AJ (1988) Complete nucleotide sequence of the genomic ovine β-lactoglobulin gene. Nucleic Acids Res 16:10379–10380
Hayashi S, Watanabe J, Kawajiri K (1991) Genetic polymorphisms in the 5′-flanking region change transcriptional regulation of the human cytochrome P450IIEI gene. J Biochem 110:559–565
Karin M, Haslinger A, Holtgreve H, Richards RI, Krauter P, Westsphal HM, Beato M (1984) Characterization of DNA sequences through which cadmium and glucocorticoid hormones induce the human metallothionein-IIA gene. Nature 308:513–519
Kozak M (1986) Influences of mRNA secondary structure on initiation by eukaryotic ribosomes. Proc Natl Acad Sci USA 83:2850–2854
Kozak M (1989) Circumstances and mechanisms of inhibition of translation by secondary structure in eukaryotic mRNAs. Mol Cell Biol 9:5134–5142
Lee CS, Oka T (1992) A pregnancy-specific mammary nuclear factor involved in the repression of the mouse β-casein gene transcription by progesterone. J Biol Chem 267:5797–5801
Lin CY, McAllister AJ, Ng-Kwai-Hang KF, Hayes JF, Batra TR, Lee AJ, Roy GL, Vesely JA, Wauthy JM, Winter KA (1989) Relationships of milk protein types to lifetime performance. J Dairy Sci 72:3085–3090
Mao IL, Buttazzoni LG, Aleandri R (1992) Effects of polymorphic milk protein genes on milk yield and composition traits in Hoistein cattle. Acta Agric Scand, Section A, Anim Sci 42:1–7
McLean DM, Graham ERB, Ponzoni RW, McKenzie HA (1984) Effects of milk protein genetic variants on milk yield and composition. J Dairy Res 51:531–546
Mermod N, Williams TJ, Tjian R (1988) Enhancer binding factors AP-4 and AP-1 act in concert to activate SV40 late transcription in vitro. Nature 332:557–561
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16:1215
Mink S, Härtig E, Jennewein P, Doppler W, Cato ACB (1992) A mammary cell-specific enhancer in mouse mammary tumor virus DNA is composed of multiple regulatory elements including binding sites for CTF/NF1 and a novel transcription factor, mammary cell-activating factor. Mol Cell Biol 12:4906–4918
Mitchell PJ, Wang C, Tjian R (1987) Positive and negative regulation of transcription in vitro: enhancer-binding protein AP-2 is inhibited by SV40 T Antigen. Cell 50:847–861
Näär AM, Boutin J-M, Lipkin SM, Yu VC, Holloway JM, Glass CK, Rosenfeld MG (1991) The orientation and spacing of core DNA-binding motifs dictate selective transcriptional responses to three nuclear receptors. Cell 65:1267–1279
Ng-Kwai-Hang KF, Hayes JF, Moxley JE, Monardes HG (1986) Relationships between milk protein polymorphisms and major milk constituents in Holstein-Friesian cows. J Dairy Sci 69:22–26
Ng-Kwai-Hang KF, Monardes HG, Hayes JF (1990) Association between genetic polymorphism of milk proteins and production traits during three lactations. J Dairy Sci 73:3414–3420
Papiz MZ, Saywer L, Eliopoulos EE, North ACT, Findlay JBC, Sivaprasadarao R, Jones TA, Newcomer ME, Kraulis PJ (1986) The structure of β-lactoglobulin and its similarity to plasma retinolbinding protein. Nature 324:383–385
Pelletier J, Sonenberg N (1985) Insertion mutagenesis to increase secondary structure within the 5′ noncoding region of a eukaryotic mRNA reduces translational efficiency. Cell 40:515–526
Rosen JM, O'Neal DL, McHugh JE, Comstock JP (1978) Progesterone-mediated inhibition of casein mRNA and polysomal casein synthesis in the rat mammary gland during pregnancy. Biochem 17:290–297
Said HM, Ong DE, Shingleton JL (1989) Intestinal uptake of retinol: enhancement by bovine milk β-lactoglobulin. Am J Clin Nutr 49:690–694
Saiki RK, Gelfand DH, Stoffel S, Scharf SJ, Higuchi R, Horn GT, Mullis KB, Erlich HA (1988) Primer-directed enzymatic amplification of DNA with a thermostable DNA Polymerase. Science 239:487–491
Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA 74:5463–5467
Schmitt-Ney M, Doppler W, Ball RK, Groner B (1991) β-casein gene promoter activity is regulated by the hormone-mediated relief of transcriptional repression and a mammary gland-specific nuclear factor. Mol Cell Biol 11:3745–3755
Topper YJ, Freeman CS (1980) Multiple hormone interactions in the developmental biology of the mammary gland. Physiol Rev 60:1049–1106
Umesono K, Murakami KK, Thompson CC, Evans RM (1991) Direct repeats as selective response elements for the thyroid hormone, retinoic acid, and the vitamin D3 receptor. Cell 65:1255–1266
Watson CJ, Gordon KE, Robertson M, Clark AJ (1991) Interaction of DNA-binding proteins with a milk protein gene promoter in vitro: identification of a mammary gland-specific factor. Nucleic Acids Res 19:6603–6610
Whitelaw CBA, Harris S, McClenaghan M, Simons JP, Clark AJ (1992) Position-independent expression of the ovine β-lactoglobulin gene in transgenic mice. Biochem J 286:31–39
Yanish-Perron C, Vieira J, Messing J (1985) Improved M13 phage cloning vectors and host strains: nucleotide sequences of M13mp18 and pUC19 vectors. Gene 33:103–119
Ziska SE, Bhattacharjee M, Herber RL, Qasba PK, Vonderhaar BK (1988) Thyroid hormone regulation of α-lactalbumin: differential glycosylation and messenger ribonucleic acid synthesis in mouse mammary glands. Endocrinology 123:2242–2248
Author information
Authors and Affiliations
Additional information
Communicated by E. J. Eisen
Rights and permissions
About this article
Cite this article
Wagner, V.A., Schild, T.A. & Geldermann, H. DNA variants within the 5′-flanking region of milk-protein-encoding genes II. The β-lactoglobulin-encoding gene. Theoret. Appl. Genetics 89, 121–126 (1994). https://doi.org/10.1007/BF00226993
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00226993