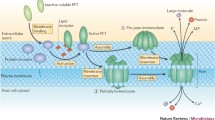
Summary and Conclusions
Depending on the size of the pores one wishes to produce in plasma membranes, the choice will probably fall on one of the three toxins discussed above. S. aureus α-toxin should be tried first when pores of 1–1.5 nm diameter are required. This is generally the case when Ca2+ and nucleotide dependence of a given process is being studied. If α-toxin does not work, this is probably due to the fact that the toxin either does not produce pores, or that the pores are too small. In this case, high concentrations of α-toxin should be tried. If this still does not work, we recommend the use of HlyA. When very large pores are to be created, e.g. for introduction of antibodies into the cells, SLO or another member of this toxin family are the agents of choice. SLO preparations need to be checked for presence of protease contaminants. Tetanolysin currently offers advantages since it is protease-free, and the size of the pores can probably be controlled by varying the toxin dose. Methods for assessing the size of pores created by such agents have been published in the recent literature, and the appropriate papers can be consulted whenever the need arises.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ahnert-Hilger G, Bhakdi S, Gratzl M (1985) Minimal requirements for exocytosis: a study using PC 12 cell permeabilized with staphylococcal α-toxin. J Biol Chem 260: 12730–12734
Ahnert-Hilger G, Bader MF, Bhakdi S, Gratzl M (1988) Introduction of macromolecules into bovine adrenal medullary chromaffin cells and rat pheochromocytoma cells (PC 12) by permeabilization with streptolysin-O: inhibitory effect of tetanus toxin on catecholamine secretion. J Neurochem 52: 1751–1758
Bader MF, Thierse D, Aunis D, Ahnert-Hilger G, Gratzl M (1986) Characterization of hormone and protein release from α-toxin permeabilized chromaffin cells in primary culture. J Biol Chem 261: 5777–5783
Belmonte G, Cescatti L, Ferrari B, Nicolussi T, Ropele M, Menestrina G (1987) Pore-formation by Staphylococcus aureus α-toxin in lipid bilayers: temperature and toxin concentration dependence. Eur Biophys J 14: 349–358
Bhakdi S, Greulich S, Muhly M, Eberspächer B, Becke H, Thiel A, Hugo F (1989) Potent leukocidal action of Escherichia coli hemolysin mediated by permeabilization of target cell membranes. J Exp Med 169: 737–754
Bhakdi S, Mackman N, Nicaud JM, Holland IB (1986) Escherichia coli hemolysin may damage target cell membranes by generating transmembrane pores. Infect Immun 52: 63–69
Bhakdi S, Martin E (1991) Superoxide generation by human neutrophils induced by low doses of Escherichia coli hemolysin. Infect Immun 59: 2955–2962
Bhakdi S, Muhly M, Korom S, Schmidt G (1990) Effects of Escherichia coli hemolysin on human monocytes. Cytocidal action and stimulation of interleukin 1 release. J Clin Invest 85: 1746–1753
Bhakdi S, Tranum-Jensen J (1988) Damage to cell membranes by pore-forming bacterial cytolysins. Prog Allergy 40: 1–43
Bhakdi S, Muhly M, Korom S, Hugo F (1989) Release of interleukin-1β associated with potent cytocidal action of staphylococcal α-toxin on human monocytes. Infect Immun 57: 3512–3519
Bhakdi S, Muhly M, Mannhardt U, Hugo F, Klapettek K, Müller-Eckhardt C, Roka L (1988) Staphylococcal α-toxin promotes blood coagulation via attack on human platelets. J Exp Med 168: 527–542
Bhakdi S, Roth M, Sziegoleit A, Tranum-Jensen J (1984) Isolation and identification of two hemolytic forms of streptolysin-O. Infect Immun 46: 394–400
Bhakdi S, Tranum-Jensen J, Sziegoleit A (1985) Mechanism of membrane damage by streptolysin-O. Infect Immun 47: 52–60
Bhakdi S, Tranum-Jensen J (1988) Damage to cell membranes by pore-forming bacterial cytolysins. Prog Allergy 40: 1–43
Bhakdi S, Tranum-Jensen J (1991) S. aureus α-toxin. Microbiol Rev 55: 733–751
Boehm DF, Welch RA, Snyder IS (1990) Domains of Escherichia coli hemolysin (HlyA) involved in binding of calcium and erythrocyte membranes. Infect Immun 58: 1959–1964
Cassidy P, Harshman S (1976) Biochemical studies on the binding of staphylococcal 125I-labeled α-toxin to rabbit erythrocytes. Biochemistry 15: 2348–2355
Cavalieri SJ, Bohach GA, Snyder IS (1984) Escherichia coli α-hemolysin: characteristics and probable role in pathogenicity. Microbiol Rev 48: 326–343
Cavalieri SJ, Snyder IS (1982) Effect of Escherichia coli alpha-hemolysin on human peripheral leukocyte function in vitro. Infect Immun 37: 966–974
Forti S, Menestrina G (1989) Staphylococcal α-toxin increases the permeability of lipid vesicles by cholesterol and pH-dependent assembly of oligomeric channels. Eur J Biochem 181: 767–773
Füssle R, Bhakdi S, Sziegoleit A, Tranum-Jensen J, Kranz T, Wellensiek HL (1981) On the mechanism of membrane damage by S. aureus α-toxin. J Cell Biol 91: 83–94
Gadeberg OV, Orskov I (1984) In vitro cytotoxin effect of alpha-hemolytic Escherichia coli on human blood granulocytes. Infect Immun 45: 255–259
Geoffroy C, Mengaud J, Alouf JE, Cossart P (1990) Alveolysin, the thiol-activated toxin of Bacillus alvei, is homologous to listeriolysin-O, perfringolysin-O, and streptolysin-O and contains a single cysteine. J Bacteriol 172: 7301–7305
Grimminger F, Sibelius U, Bhakdi S, Suttorp N, Seeger W (1991) Escherichia coli hemolysin is a potent inductor of phosphoinositide hydrolysis and related metabolic responses in human neutrophils. J Clin Invest 88: 1531–1539
Hildebrand A, Roth M, Bhakdi S (1991) Staphylococcus aureus α-toxin: dual mechanisms of binding to target cells. J Biol Chem 266: 17195–17200
Ikigae H, Nakae T (1987) Interaction of the α-toxin of S. aureus with the liposome membrane. J Biol Chem 262: 2150–2155
Issartel J-P, Koronakis V, Hughes C (1991) Activation of Escherichia coli prohaemolysin to the mature toxin by acyl carrier protein-dependent fatty acylation. Nature 351: 759–761
Keane WF, Welch R, Gekker G, Peterson PK (1987) Mechanism of Escherichia coli alphahemolysin-induced injury to isolated renal tubular cells. Am J Pathol 126: 350–357
Kehoe MA, Miller L, Walker JA, Boulnois GJ (1987) Nucleotide sequence of the streptolysin-O (SLO) gene: Structural homologies between SLO and other membrane damaging, thiol-activated toxins. Infect Immun 55: 3228–3232
Ludwig A, Jarchau T, Benz R, Goebel W (1988) The repeat domain of E. coli hemolysin is responsible for its Ca-dependent binding to erythrocytes. Mol Gen Genet 214: 553–561
Mackman N, Holland IB (1984) Functional characterization of a cloned hemolysin determinant from E. coli of human origin, encoding information for the secretion of a 107 K polypeptide. Mol Gen Genet 196: 129–134
Menestrina G, Mackman N, Holland IB, Bhakdi S (1987) Escherichia coli hemolysin forms voltage dependent ion channels in lipid membranes. Biochim Biophys Acta 905: 109–117
Ni J, Watson JV, Cox H, Karpas A (1993) Multiparameter flow cytometric analysis of a novel cytotoxin (Factor 2) induced tumor cell membrane permeability. Cytometry 14: 281–286
Pinkney M, Beachey E, Kehoe M (1989) The thiol-activated toxin streptolysin O does not require a thiol group for cytolytic activity. Infect Immun 57: 2553–2558
Saunders FK, Mitchell TJ, Walker JA, Andrew PW, Boulnois GJ (1989) Pneumolysin, the thiol-activated toxin of Streptococcus pneumoniae, does not require a thiol group for in vitro activity. Infect Immun 57: 2547–2552
Suttorp N, Flöer B, Schnittler H, Seeger W, Bhakdi S (1990) Effects of Escherichia coli hemolysin on endothelial cell function. Infect Immun 58: 3796–3801
Tobkes N, Wallace BA, Bayley H (1985) Secondary structure and assembly mechanism of an oligomeric channel protein. Biochemistry 24: 1915–1920
Tweten R (1988) Nucleotide sequence of the gene for perfringolysin O (theta toxin) from Clostridium perfringens: significant homology with the genes for streptolysin and pneumolysin. Infect Immun 56: 3235–3240
Walker JA, Allen RA, Falmagne P, Johnson MK, Boulnois GJ (1987) Molecular cloning, characterization and complete nucleotide sequence of the gene for pneumolysin, the sulfhydryl-activated toxin of Streptococcus pneumoniae. Infect Immun 55: 1184–1189
Walker B, Krishnasastry M, Zorn L, Bayley H (1992) Assembly of the oligomeric membrane pore formed by staphylococcal α-hemolysin examined by truncation mutagenesis. J Biol Chem 267: 21782–21786
Welch RA (1991) Pore-forming cytolysins of gram-negative bacteria. Mol Microbiol 5: 521–529
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bhakdi, S., Weller, U., Walev, I. et al. A guide to the use of pore-forming toxins for controlled permeabilization of cell membranes. Med Microbiol Immunol 182, 167–175 (1993). https://doi.org/10.1007/BF00219946
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00219946