Abstract
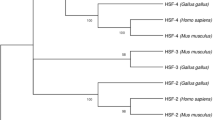
Three genes encoding members of the M r 70 000 heat shock protein family (HSP70) are known to lie in the class III region of the human major histocompatibility complex. IN order to determine whether these genes or their protein products exhibit any polymorphism the three genes have been specifically amplified from genomic DNA and sequenced. The HSP70-1 and HSP70-2 genes encode the major heat-inducible HSP70. A comparison of the nucleotide sequences of these genes from B8, SC01, DR3, B18, F1C30, DR3, and B7, SC30, DR2 haplotypes has revelad only very limited sequence variation which is not associated with any amino acid polymorphism. The HSP70-Hom gene encodes a protein that is highly related to HSP70-1, but which is not heat-inducible. Nucleotide sequence analysis of this gene from different haplotypes has revealed a Met → Thr amino acid substitution at residue 493 in a number of the haplotypes tested. This variable amino acid lies in the proposed peptide-binding site of the HSP70-Hom protein.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bjorkman, P. J., Saper, M. A., Samraoui, B., Bennett, W. S., Strominger, J. L., and Wiley, D. C.: Structure of the human class I histocompatibility antigen HLA-A2. Nature 239: 506–512, 1987
Chappell, T. G., Konforti, B. B., Schmid, S. L., and Rothman, J. E.: The ATPase core of a clathrin uncoating protein. J Biol Chem 262: 746–751, 1987
Chiang, H. L., Terlecky, S. R., Plant, C. P., and Dice, J. F.: A role for a 70-kilodalton heat shock protein in lysosomal degradation of intracellular proteins. Science 246: 382–385, 1989
Dice, J. F.: Peptide sequences that cytosolic proteins for lysosomal proteolysis. Trends Biochem 15: 305–309, 1990
Ennis, P. D., Zemmour, J., Salter, R. D., and Parham, P.: Rapid cloning of HLA-A, −B cDNA by using the polymerase chain reaction: frequency and nature of errors produced in amplification. Proc Natl Acad Sci USA 87: 2833–2837, 1990
Flajnik, M. F., Canel, C., Kramer, J., and Kasahara, M.: Which came first, MHC class I or class II? Immunogenetics 33: 295–300, 1991
Flynn, G. C., Chappell, T. G., and Rothman, J. E.: Peptide binding and release by proteins implicated as catalysts of protein assembly. Science 245: 385–390, 1989
Fujimoto, H., Ito, Y., Ando, A., Matsumoto, M., Fujio K., Miura, K., Shirai, M., and Inoko, H.: The human HSP70 homolog gene located in the HLA class III is expressed in the testicular germ cells. In K. Tsuji, M. Aizawa, and T. Sasazuki (eds.): HLA 1991, Proceedings of the 11th International Histocompatibility Workshop, Oxford University Press, in press, 1992
Goate, A. M., Cooper, D. N., Hall, C., Leung, T. K. C., Solomon, E., and Lim, L.: Localisation of the human heat-shock HSP70 gene sequence to chromosome 6 and detection of two other loci by somatic-cell hybrid and restriction fragment length polymorphism analysis. Somatic Cell Mol Genet 13: 119–130, 1987
Hunt, C. and Morimoto, R. I.: Conserved features of eukaryotic HSP70 genes revealed by comparison with the nucleotide sequence of human HSP70. Proc Natl Acad Sci USA 82: 6455–6459, 1985
Langer, T. and Neupert, W.: Heat shock proteins hsp60 and hsp70: their roles in folding assembly and membrane translocation of proteins. Curr Top Microbiol Immunol 167: 3–30, 1991
Madden, D. R., Gorga, J. C., Strominger, J. L., and Wiley, D. C.: The structure of HLA-B27 reveals nonamer self-peptides bound in an extended conformation. Nature 353: 321–325, 1991
Matsumoto, M. and Fujimoto, H.: Cloning of a hsp70-related gene expressed in mouse spermatids. Biochem Biophys Res Comm 166: 43–49, 1990
Milner, C. M. and Campbell, R. D.: Structure and expression of the MHC-linked HSP70 genes. Immunogenetics 32: 242–251, 1990
Morimoto, R. I. and Milarski, K. L.: Expression and function of vertebrate HSP70 genes. In R. I. Morimoto, A. Tissieres, and C. Georgopoulos (eds.): Stress Proteins in Biology and Medicine, pp. 322–359, Cold Spring Harbour Laboratory Press, New York, 1990
Morimoto, R. I., Tissieres, A., and Georgopoulos, C.: The stress response, function of the proteins, and perspectives. In R. I. Morimoto, A. Tissieres, and C. Georgopoulos (eds.): Stress Proteins in Biology and Medicine, pp. 1–36, Cold Springer Harbour Laboratory Press, New York,1990
Parham, P., Lomen, C. E., Lawlor, D. A., Ways, J. P., Holmes, N., Coppin, H. L., Salter, R. D., Wan, A. M., and Ennis, P. D.: Nature of polymorphism in HLA-A, −B and −C molecules. Proc Natl Acad Sci USA 85: 4005–4009, 1988
Pierce, S. K., DeNagel, D. C., and VanBuskirk, A. M.: A role for heat shock proteins in antigen processing and presentation. Curr Top Microbiol Immunol 167: 83–92, 1991
Rippmann, F., Taylor, W. R., Rothbard, J. B., and Green, N. M.: A hypothetical model for the peptide binding domain of hsp70 based on the peptide binding domain of HLA. EMBO J 10: 1053–1059, 1991
Saper, M. A., Bjorkman, P. J., and Wiley, D. C.: Refined structure of the human histocompatibility antigen HLA-A2 at 2.6 Å resolution. J Mol Biol 219: 277–319, 1991
Sargent, C. A., Dunham, I., Trowsdale, J., and Campbell, R. D.: Human major histocompatibility complex contains genes for the major heat shock protein HSP70. Proc Natl Acad Sci USA 86: 1968–1972, 1989
Snoek, M., Groot, P. C., Spies, T., Campbell, R. D., and Demant, P.: Fine mapping of the crossover sites in the C4-H-2D region of H-2 recombinants mouse strains. Immunogenetics 34: 409–412, 1991
Teuscher, C., Gasser, D. L., Woodward, S. R., and Hickey, W. F.: Experimental allergic orchitis in mice. VI. Recombinations within the H-2S/H-2D interval define the map postion of the H-2-associated locus controlling disease susceptibility. Immungenetics 32: 337–344, 1990
VanBuskirk, A. M., DeNage, D. C., Guagliardi, L. E., Brodsky, F. M., and Pierce, S. K.: Cellular and subcellular distribution of PBP72/74, a peptide-binding protein that plays a role in antigen processing. J Immunol 146: 500–506, 1991
Winfield, J. B. and Jarjour, W. N.: Stress proteins, autoimmunity and autoimmune disease. Curr Top Microbiol Immunol 167: 161–189, 1991
Young, D. B., Mehlert, A., and Smith, D. F.: Stress proteins and infectious diseases. In R. I. Morimoto, A. Tissieres, and C. Georgopoulos (eds.): Stress Proteins in Biology and Medicine, pp. 131–165, Cold Spring Harbour Laboratory Press, New York, 1990
Author information
Authors and Affiliations
Additional information
Address correspondence and offprint requests to: R. D. Campbell.
Rights and permissions
About this article
Cite this article
Milner, C.M., Campbell, R.D. Polymorphic analysis of the three MHC-linked HSP70 genes. Immunogenetics 36, 357–362 (1992). https://doi.org/10.1007/BF00218042
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00218042