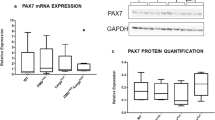
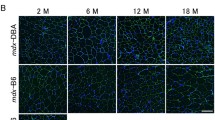
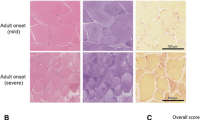
Summary
A mutant mouse with a hereditary myotonia, ‘arrested development of righting response’, ADR, was investigated with respect to mononucleated cell populations in skeletal muscle. Upon enzymatic dissociation of different muscles from mice aged between 15 and 120 days, a 3-to 5-fold higher yield of mononucleated cells per muscle fresh weight was obtained from mice with the ADR syndrome than from control mice. Clonal cell culture showed that the absolute number of cells with myogenic potential was increased and that mutant clones had shorter generation times than wild-type controls. Morphological differentiation of ADR myotubes was indistinguishable from that of the controls. Light microscopy confirmed the presence of increased numbers of mononucleated cells per muscle volume. At the ultrastructural level, there were 3.3 times as many satellite cells (the myogenic stem cells of mature muscle) per myofibre nucleus in ADR than in controls. Because no fibre degeneration was observed in the ADR mutant, we conclude that the enlarged mutant satellite cell pool is not a result of compensatory proliferation but is a consequence of fibre-type transformation and/or delayed maturation of the myotonic muscle.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Blau HM, Webster C (1981) Isolation and characterization of human muscle cells. Proc Natl Acad Sci USA 78:5623–5627
Bradley WG (1979) Muscle fiber splitting. In: Mauro A (ed) Muscle regeneration. Raven Press, New York, pp 215–232
Bulfield G, Siller WG, Wight PAL, Moore KJ (1984) X chromosome-linked muscular dystrophy (mdx) in the mouse. Proc Natl Acad Sci USA 81:1189–1192
Campion DR (1985) The muscle satellite cell: a review. Int Rev Cytol 87:225–251
Cullen MJ (1986) Ultrastructure of myofibres in Duchenne muscular dystrophy (DMD). J Muscle Res Cell Motil 7(1):81
Cullen MJ, Watkins SC (1981) The role of satellite cells in regeneration in diseased muscle. Adv Physiol Sci 24:341–349
Dangain J, Vrbová G (1984) Muscle development in mdx mutant mice. Muscle Nerve 7:700–704
Engel WK, Cunningham GG (1963) Rapid examination of muscle tissues. An improved trichrome method. Neurology 13:919–923
Füchtbauer EM, Reininghaus J, Jockusch H (1985) Fibre histochemistry and contractile properties of mouse mutant skeletal muscles. J Muscle Res Cell Motil 6:83
Gibson MC, Schultz E (1982) The distribution of satellite cells and their relationship to specific fiber types in soleus and extensor digitorum longus muscle. Anat Rec 202:329–337
Gibson MC, Schultz E (1983) Age-related differences in absolute numbers of skeletal muscle satellite cells. Muscle Nerve 6:574–580
Heller AH, Eicher EM, Hallett M, Sidman RL (1982) Myotonia, a new inherited muscle disease in mice. J Neurosci 2:924–933
Jockusch H, Bertram K (1986) “Arrested development of righting response” (adr) and “myotonia” (mto) are allelic. Mouse News Letter (75):19
Jockusch H, Burkart W, Burger MM (1980) Hereditary motor endplate disease (med) of the mouse: Observations on dissociated myogenic cells and their development in culture. Cell Tissue Res 207:241–248
Konigsberg IR (1971) Diffusion-mediated control of myoblast fusion. Dev Biol 26:133–152
Laarse WJ van der, Crusio WE, Maslam S, Abeelen JHF van (1984) Genetic architecture of numbers of fast and slow muscle fibres in the mouse soleus. Heredity 53:643–647
Lojda Z, Gossrau R, Schiebler TH (1976) Enzymhistochemische Methoden. Springer, Berlin Heidelberg New York, pp 64–78
Maier A, Gambke B, Pette D (1986) Degeneration-regeneration as a mechanism contributing to the fast to slow conversion of chronically stimulated fast-twitch rabbit muscle. Cell Tissue Res 244:635–643
Mauro A (1961) Satellite cell of skeletal muscle fibers. J Biophys Biochem Cytol 9:493–495
Mehrke G (1985) Physiologische und genetische Faktoren bei der Nerv-Muskel-Interaktion in Zellkultur. PhD thesis, University of Bielefeld
Mehrke G, Reininghaus J, Jockusch H (1986a) Unusual contractile properties of mutant (ADR) mouse muscle are caused by repetitive action potentials. J Muscle Res Cell Motil 7:85
Miike T (1983) Maturational defect of regenerating muscle fibers in cases with Duchenne and congenital muscular dystrophies. Muscle Nerve 6:545–552
Peters J (1986) Mouse gene list. Mouse News Letter 74:3–55
Schmalbruch H (1985) Skeletal Muscle. In: Handbuch der mikroskopischen Anatomie des Menschen. Oksche A, Vollrath L (eds) Vol. II/6 Springer, Berlin Heidelberg New York
Schmalbruch H, Hellhammer U (1976) The number of satellite cells in normal human muscle. Anat Rec 185:279–288
Schultz E (1974) A quantitative study of the satellite cell population in postnatal mouse lumbrical muscle. Anat Rec 180:589–596
Snow MH (1977) The effects of aging on satellite cells in skeletal muscles of mice and rats. Cell Tissue Res 185:399–408
Stuhlfauth I, Reininghaus J, Jockusch H, Heizmann CW (1984) Calcium-binding protein, parvalbumin, is reduced in mutant mammalian muscle with abnormal contractile properties. Proc Natl Acad Sci USA 81:4814–4818
Terasawa K (1986) Muscle regeneration and satellite cells in Fukuyama type congenital muscular dystrophy. Muscle Nerve 9:465–470
Wakayama Y, Schotland DL (1979) Muscle satellite cells in Duchenne dystrophy. In: Mauro A (ed) Muscle regeneration. Raven Press, New York, pp 121–129
Watkins WJ, Watts DC (1984) Biological features of the new A2G-adr mouse mutant with abnormal muscle function. Lab Anim 18:1–6
Watts RL, Watkins J, Watts DC (1978) A new mouse mutant with abnormal muscle function: comparison with the Re-dy mouse. In: Lunt GG, Marchbanks RM (eds) The biochemistry of myasthenia gravis and muscular dystrophy. Academic Press, London, pp 331–334
Yasin R, Kundu D, Thompson EJ (1981) Growth of adult human cells in culture at clonal densities. Cell Differ 10:131–137
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schimmelpfeng, J., Jockusch, H. & Heimann, P. Increased density of satellite cells in the absence of fibre degeneration in muscle of myotonic mice. Cell Tissue Res. 249, 351–357 (1987). https://doi.org/10.1007/BF00215519
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00215519