Abstract
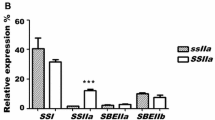
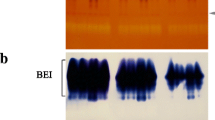
The effects of waxy mutations on starch-granule-bound starch synthases (EC 2.4.1.18) in the developing endosperm of barley (Hordeum vulgare L.) and maize (Zea mays L.) have been investigated. Three granule-bound starch synthases in barley endosperm were identified by use of antibodies to known starch synthases, by reconstitution and assay of individual proteins from sodium dodecyl sulphate-polyacrylamide gels of granule-bound proteins, and by partial purification of proteins released by enzymic digestion of starch. These are proteins of 60, 77 and 90 kDa. Use of antibodies to known starch synthases and partial purification of proteins released by enzymic digestion of starch indicated that there may be at least four granule-bound starch synthases in maize endosperm: proteins of 59, 74, 77 and 83 kDa. Mutations at the waxy loci of both species affected only the 60- (barley) and 59-(maize) kDa isoforms. No evidence was found that other putative isoforms are altered in abundance or activity by the mutations. The contribution of our results to understanding of the starch synthase activity of intact starch granules and the mechanism of amylose synthesis is discussed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ainsworth C, Clark J, Balsdon J (1993) Expression, organisation and structure of the genes encoding the waxy protein (granule-bound starch synthase) in wheat. Plant Mol Biol 22: 67–82
Denyer K, Barber LM, Burton R, Hedley CL, Hylton CM, Johnson S, Jones DA, Marshall J, Smith AM, Tatge H, Tomlinson K, Wang TL (1995a) The isolation and characterisation of novel low-amylose mutants of Pisum sativum L. Plant Cell Environ 18: 1019–1026
Denyer K, Hylton CM, Jenner CF, Smith AM (1995b) Identification of multiple isoforms of soluble and granule-bound starch synthase in developing wheat endosperm. Planta 196: 256–265
Denyer K, Sidebottom C, Hylton CM, Smith AM (1993) Soluble isoforms of starch synthase and starch-branching enzyme also occur within starch granules in developing pea embryos. Plant J 4: 191–198
Dry I, Smith AM, Edwards EA, Bhattacharyya M, Dunn P, Martin C (1992) Characterisation of cDNAs encoding two isoforms of granule-bound starch synthase which show differential expression in developing storage organs. Plant J 2: 193–202
Echt CS, Schwart D (1981) Evidence for the inclusion of controlling elements within the structural gene at the waxy locus in maize. Genetics 99: 275–284
Edwards A, Marshall J, Sidebottom C, Visser RGF, Smith AM, Martin C (1995) Biochemical and molecular characterisation of a starch synthase from potato tubers which is both soluble and granule-bound. Plant J 8: 283–294
Frydman RB, Cardini CE (1967) Studies on the biosynthesis of starch. II. Some properties of the adenosine diphosphate glucose: starch glucosyltransferase bound to the starch granule. J Biol Chem 242: 312–317
Hager DA, Burgess RR (1980) Elution of proteins from SDS-polyacrylamide gels, removal of SDS, and renaturation of enzymatic activity: results with sigma subunit of E. coli RNA polymerase, wheat germ DNA topoisomerase, and other enzymes. Anal Biochem 109: 76–86
Jenner CF, Denyer K, Hawker JS (1994) Caution on the use of the generally accepted methanol precipitation technique for the assay of soluble starch synthase in crude extracts of plant tissues. Aust J Plant Physiol 21: 17–22
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685
Macdonald FD, Preiss J (1983) Solubilization of starch-granule-bound starch synthase of normal maize kernels. Plant Physiol 73: 175–178
Macdonald FD, Preiss J (1985) Partial purification and characterisation of granule-bound starch synthases from normal and waxy maize. Plant Physiol 78: 849–852
McDonald ALM, Stark JR, Morrison WR, Ellis RP (1991) The composition of starch granules from developing barley genotypes. J Cereal Sci 13: 93–112
Morrison WR, Scott DC, Karklas J (1986) Variation in the composition and physical properties of barley starches. Stärke 38: 374–379
Mu C, Harn C, Ko Y-T, Singletary GW, Keeling BP, Wasserman BP (1994) Association of a 76 kDa polypeptide with soluble starch synthase I activity in maize (cv B73) endosperm. Plant J 6: 151–159
Neuffer MG, Chang M-T (1989) Induced mutations in biological and agronomic research. Vortr Pflanzenzüchtg. 16: 165–178
Preiss J (1992) Biology and molecular biology of starch synthesis and its regulation. In: Miflin BJ (ed) Oxford surveys of plant molecular and cell biology (Vol. 7). Oxford University Press, pp 59–144
Rohde W, Becker D, Salamini F (1988) Structural analysis of the waxy locus from Hordeum vulgare. Nucleic Acids Res 16: 7185–7186
Shure M, Wessler S, Fedoroff N (1983) Molecular identification and isolation of the waxy locus in maize. Cell 35: 225–233
Sivak MN, Wagner M, Preiss J (1993) Biochemical evidence for the role of the waxy protein from pea (Pisum sativum L.) as a granule-bound starch synthase. Plant Physiol. 103: 1355–1359
Smith AM (1990) Evidence that the waxy protein of pea (Pisum sativum L.) is not the major starch-granule-bound starch synthase. Planta 182: 599–604
Smith AM, Martin C (1993) Starch biosynthesis and the potential for its manipulation. In: Grierson D (ed) Biosynthesis and manipulation of plant products. Blackie and Son, Glasgow, pp 1–54
Visser RGF, Hergersberg M, van der Leij FR, Jacobsen E, Witholt B, Feenstra WJ (1989) Molecular cloning and partial characterisation of the gene for granule-bound starch synthase from a wildtype and an amylose-free potato (Solanum tuberosum L.). Plant Sci 64: 185–192
Author information
Authors and Affiliations
Additional information
We are very grateful to Dr. Roger Ellis (SCRI, Dundee, Scotland) for the gift of barley seeds, and to Drs Roger Ellis, Alan Schulman and Cathie Martin for helpful advice and comments during the course of this work.
Rights and permissions
About this article
Cite this article
Hylton, C.M., Denyer, K., Keeling, P.L. et al. The effect of waxy mutations on the granule-bound starch synthases of barley and maize endosperms. Planta 198, 230–237 (1996). https://doi.org/10.1007/BF00206248
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00206248