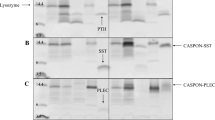
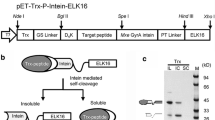
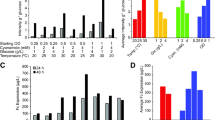
Summary
An approach to produce 13C-and 15N-enriched proteins is described. The concept is based on intracellular production of the recombinant proteins in Escherichia coli as fusions to an IgG-binding domain, Z, derived from staphylococcal protein A. The production method provides yields of 40–200 mg/l of isotope-enriched fusion proteins in defined minimal media. In addition, the Z fusion partner facilitates the first purification step by IgG affinity chromatography. The production system is applied to isotope enrichment of human insulin-like growth factor II (IGF-II), bovine pancreatic trypsin inhibitor (BPTI), and Z itself. High levels of protein production are achieved in shaker flasks using totally defined minimal medium supplemented with 13C6-glucose and (15NH4)2SO4 as the only carbon and nitrogen sources. Growth conditions were optimized to obtain high protein production levels and high levels of isotope incorporation, while minimizing 13C6-glucose usage. Incorporation levels of 13C and/or 15N isotopes in purified IGF-II, BPTI, and Z were confirmed using mass spectrometry and NMR spectroscopy. More than 99% of total isotope enrichment was obtained using a defined isotope-enriched minimal medium. The optimized systems provide reliable, high-level production of isotope-enriched fusion proteins. They can be used to produce 20–40 mg/l of properly folded Z and BPTI proteins. The production system of recombinant BPTI is state-of-the-art and provides the highest known yield of native refolded BPTI.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BPTI:
-
bovine pancreatic trypsin inhibitor
- DTT:
-
dithiothreitol
- Gdn-HCl:
-
guanidinium hydrochloride
- IAA:
-
β-indole acrylic acid
- IGF-II:
-
insulin-like growth factor II
- PBS:
-
phosphate-buffered saline
- PDMS:
-
plasma desorption mass spectrometry
- PFPA:
-
pentafluoro propionic acid
- RP-HPLC:
-
reversed-phase high performance liquid chromatography
- Z:
-
IgG-binding protein domain derived from staphylococcal protein A.
References
Abeygunawardana, C., Weber, D.J., Frick, D.N., Bessman, M.J. and Mildvan, A.S. (1993) Biochemistry 32, 13071–13080.
Altman, J.D., Henner, D., Nilsson, B., Anderson, S. and Kuntz, I.D. (1991) Protein Eng., 4, 593–600.
Archer, S.J., Bax, A., Roberts, A.B., Sporn, M.B., Ogawa, Y., Piez, K.A., Waetherbee, J.A., Tsang, M.L.-S., Lucas, R., Zheng, B.-L., Wenker, J. and Torchia, D.A. (1993) Biochemistry 32, 1152–1163.
Balbás, P., Soberón, X., Merino, E., Zurita, M., Lomeli, H., Valle, F., Flores, N. and Bolivar, F. (1986) Gene, 50, 3–40.
Boucher, W., Laue, E.D., Campbell-Burk, S.L. and Domaille, P.J. (1992) J. Am. Chem. Soc., 114, 2262–2264.
Brutscher, B., Simorre, J.-P., Caffrey, M.S. and Marion, D. (1994) J. Magn. Reson. Ser. B, 105, 77–82.
Cedergren, L., Andersson, R., Jansson, B., Uhlen, M. and Nilsson, B. (1993) Protein Eng. 6, 441–448.
Clore, G.M., Bax, A., Wingfield, P.T. and Gronenborn, A.M. (1990) Biochemistry, 29, 5671–5676.
Clore, G.M. and Gronenborn, A.M. (1991) Science, 252, 1390–1399.
Clore, G.M. and Gronenborn, A.M. (1994) Protein Sci., 3, 372–390.
Clowes, R.T., Boucher, W., Hardman, C.H., Domaille, P.J. and Laue, E.D. (1993) J. Biomol. NMR, 3, 349–354.
de, Dios, A.C., Pearson, J.G. and Oldfield, E. (1993) Science, 260, 1491–1496.
Fesik, S.W., Gampe, R.T., Holzman, T.F., Egan, D.A., Edalji, R., Luly, J.R., Simmer, R., Helfrich, R., Kishore, V. and Rich, D.H. (1990) Science, 250, 1406–1409.
Fesik, S.W., Gampe, R.T., Eaton, H.L., Gemmecker, G., Olejniczak, E.T., Neri, P., Holzman, T.F., Egan, D.A., Edalji, R., Simmer, R., Helfrich, R., Hochlowski, J. and Jackson, M. (1991) Biochemistry, 30, 6574–6583.
Forsberg, G., Palm, G., Ekebacke, A., Josephson, S. and Hartmanis, M. (1990) Biochem. J., 271, 357–363.
Griesinger, C. and Eggenberger, U. (1992) J. Magn. Reson., 97, 426–434.
Griesinger, C. and Eggenberger, U. (1992) J. Magn. Reson., 97, 426–434.
Grodberg, J. and Dunn, J.J. (1988) J. Bacteriol., 170, 1245–1253.
Grzesiek, S. and Bax, A. (1993) Acc. Chem. Res., 26, 131–138.
Hammarberg, B., Nygren, P.Å., Holmgren, E., Elmblad, A., Tally, M., Hellman, U., Moks, T. and Uhlén, M.. (1989) Proc. Natl Acad. Sci. USA, 86, 4367–4371.
Hammarberg, B., Tally, M., Samuelsson, E., Wadensten, H., Holmgren, E., Hartmanis, M., Hall, K., Uhlén, M. and Moks, T. (1991) J. Biol. Chem., 266, 11058–11062.
Hansen, A.P., Petros, A.M., Mazar, A.P., Pederson, T.M., Rueter, A. and Fesik, S.W. (1992) Biochemistry, 31, 12713–12718.
Hurle, M.R., Marks, C.B., Kosen, P.A., Anderson, S.A. and Kuntz, I.D. (1990) Biochemistry, 29, 4410–4419.
Ikura, M., Kay, L.E. and Bax, A. (1990) Biochemistry 29, 4659–4667.
Ikura, M., Clore, G.M., Gronenborn, A.M., Zhu, G., Klee, C.B. and Bax, A. (1992) Science, 256, 632–637.
Kay, L.E., Torchia, D.A. and Bax, A. (1989) Biochemistry, 28, 8972–8979.
Kay, L.E., Clore, G.M., Bax, A. and Gronenborn, A.M. (1990) Science, 243, 411–414.
Kriwacki, R.W., Hill, R.B., Flanagan, J.M., Caradonna, J.P. and Prestegard, J.H. (1993) J. Am. Chem. Soc. 115, 8907–8911.
Laemmli, U.K. (1970) Nature, 227, 680–685.
Laws, D.D., de, Dios, A.C. and Oldfield, E. (1993) J. Biomol. NMR, 3, 607–612.
Logan, T.M., Olejniczak, E.T., Xu, R.X. and Fesik, S.W. (1992) FEBS Lett., 314, 413–418.
Logan, T.M., Thériault, Y. and Fesik, S.W. (1994) J. Mol. Biol. 236, 637–648.
Lundström, H., Brobjer, M., Österlöf, B. and Moks, T. (1990) Biotechnol. Bioeng. 36, 1056–1062.
Lyons, B.A., Tashiro, M., Cedergren, L., Nilsson, B. and Montelione, G.T. (1993) Biochemistry, 32, 7839–7845.
Makhatadze, G.I., Kim, K.-S., Woodward, C. and Privalov, P. (1993) Protein Sci., 2, 2028–2036.
Maurer, R., Meyer, B.J. and Ptashne, M. (1980) J. Mol. Biol. 139, 147–161.
Miller, J.H. (1972) Experiments in Molecular Genetics, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY.
Moks, T., Abrahmsén, L., Holmgren, E., Bilich, M., Olsson, A., Uhlén, M., Pohl, G., Sterky, C., Hultberg, H., Josephson, S., Holmberg, A., Jörnvall, H. and Nilsson, B. (1987) Biochemistry, 26, 5239–5244.
Montelione, G.T. and Wagner, G. (1989) J. Am. Chem. Soc., 111, 5474–5475.
Montelione, G.T., Winkler, M.E., Rauenbuehler, P. and Wagner, G. (1989) J. Magn. Reson., 82, 198–204.
Montelione, G.T. and Wagner, G. (1990) J. Magn. Reson., 83, 183–188.
Montelione, G.T., Emerson, S.D. and Lyons, B.A. (1992a) Biopolymers, 32, 327–334.
Montelione, G.T., Lyons, B.A., Emerson, S.D. and Tashiro, M. (1992b) J. Am. Chem. Soc., 114, 10974–10975.
Muhandiram, D.R., Xu, G.Y. and Kay, L.E. (1993) J. Biomol. NMR, 3, 463–470.
Neidhart, F.C. (1987) Escherichia coli and Salmonella typhimurium: Cellular and Molecular Biology, American Society for Microbiology, Washington, DC.
Nilsson, B., Moks, T., Jansson, B., Abrahmsén, L., Elmblad, A., Holmgren, E., Henrichson, C., Jones, T.A. and Uhlén, M. (1987) Protein Eng., 1, 107–113.
Nilsson, B. and Abrahmsén, L. (1990) Methods Enzymol., 185, 144–161.
Nilsson, B., Forsberg, G. and Hartmanis, M. (1991) Methods Enzymol., 198, 3–16.
Nilsson, B., Forsberg, G., Moks, T., Hartmanis, M. and Uhlén, M. (1992) Curr. Opin. Struct. Biol., 2, 569–575.
Peng, J.W. and Wagner, G. (1992) Biochemistry, 31, 8573–8586.
Qian, Y.Q., Otting, G., Billeter, M., Müller, M., Gehring, W.J. and Wüthrich, K. (1993) J. Mol. Biol., 234, 1070–1083.
Reilly, D. and Fairbrother, W.J. (1994) J. Biomol. NMR, 4, 459–462.
Samuelsson, E., Moks, T., Nilsson, B. and Uhlén, M. (1994) Biochemistry, 33, 4207–4211.
Spera, S. and Bax, A. (1991) J. Am. Chem. Soc., 113, 5490–5492.
Sugimura, K. and Higashi, N. (1988) J. Bacteriol., 170, 3650–3654.
Venters, R.A., Calderone, T.L., Spicer, L.D. and Fierke, C.A. (1991) Biochemistry, 30, 4491–4494.
Vuister, G.W., Wang, A.C. and Bax, A. (1993) J. Am. Chem. Soc., 115, 5334–5335.
Wagner, G. (1993) J. Biomol. NMR, 3, 375–385.
Wishart, D.S., Sykes, B.D. and Richards, F.M. (1991) J. Mol. Biol., 222, 311–333.
Yamashiro, D. and Li, C.H. (1985) Int. J. Pept. Protein Res., 26, 299–304.
Yansura, D.B. (1990) Methods Enzymol., 185, 161–166.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jansson, M., Li, YC., Jendeberg, L. et al. High-level production of uniformly 15N-and 13C-enriched fusion proteins in Escherichia coli . J Biomol NMR 7, 131–141 (1996). https://doi.org/10.1007/BF00203823
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00203823