Abstract
To prevent drug accumulation and adverse effects the dose of hydrophilic angiotensin-converting enzyme (ACE) inhibitors, e. g. lisinopril, must be reduced in patients with renal failure. To obtain a rational basis for dose recommendations, we undertook a prospective clinical trial. After 15 days of lisinopril treatment pharmacokinetic and pharmacodynamic parameters were determined in patients with advanced renal failure (n=8; endogenous creatinine clearance [CLCR]: 18 ml·min−1·1.73m−2) and in healthy subjects with normal renal function (n=16; CLCR: 107 ml·min−1·1.73m−2). The volunteers received 10 mg lisinopril once daily, the daily dose in patients (1.1–2.2 mg) was adjusted to the individual CLCR according to the method of Dettli [13].
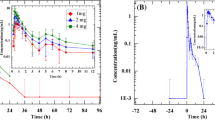
After 15 days of lisinopril treatment the mean maximal serum concentration (C max) in patients was lower than in volunteers (30.7 vs 40.7 ng·ml−1, while the mean area under the concentration-time curve (AUC 0–24 h) was higher (525 vs 473 ng·h−1·ml−1). ACE activity on day 15 was almost completely inhibited in both groups. Plasma renin activity, angiotensin I and angiotensin II levels documented marked inhibition of converting enzyme in volunteers and patients. Furthermore, average mean arterial blood pressure in patients decreased by 5 mmHg and proteinuria from 3.9–2.7 g per 24 h after 15 days of treatment with the reduced dose of lisinopril.
Adjustment of the dose of lisinopril prevents significant accumulation of the drug in patients with advanced renal failure during chronic therapy. Mean serum levels did not exceed this in subjects with normal renal function receiving a standard dose. Despite substantial dose reduction, blood pressure and proteinuria decreases were observed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Anderson S, Rennke HG, Brenner BM (1986) Therapeutic advantage of CEI in arresting progressive renal disease associated with systemic hypertension in the rat. J Clin Invest 77: 1993–2000
Kamper A, Strandgaard S, Leyssac PP (1992) Effect of enalapril on the progression of chronic renal failure. A randomized controlled trial. Am J Hypertens 5: 423–430
Landais P, Hannedouche T, Mignon F, Chanard J, Durand D, Fournier A, Godin M, Goldfarb B, Grünfeld JP (1992) Final results of the prospective multicenter study of antihypertensive therapy in chronic renal failure. 19th Congress of the EDTA, Paris, pp 85 a
Zuchelli P, Zuccala A, Borghi M, et al (1992) Long-term comparison between captopril and nifedipine in the progression of renal insufficiency. Kidney Int 42: 452–458
Kelly JG, Doyle G, Donohoe J, Laher M, Long C, Cooper WD (1987) Acute and chronic dose pharmacokinetics of lisinopril: effects of renal impairment. Br J Clin Pharmacol 23: 629–638
Drummer OH, Workman BS, Miach PJ, Jarrott B, Louis WJ (1987) The pharmacokinetics of captopril and captopril disulfide conjugates in uremic patients on maintenance dialysis: comparison with patients with normal renal function. Eur J Clin Pharmacol 32: 267–271
Fruncillo RJ, Rocci ML, Vlasses PH, Mojaverian P, Shepley K (1987) Disposition of enalapril and enalaprilat in renal insufficiency. Kidney Int 31 [Suppl 20]: S117-S122
Begg EJ, Bailey RR, Lynn KL, Robson RA, Frank GJ (1989) The pharmakokinetics of ACE inhibitors in patients with renal impairment. J Hypertens 7 [Suppl 5]: S29-S32
Case DE (1989) The clinical pharmacology of lisinopril. J Hum Hypertens 3 [Suppl 1]: 127–132
Ulm EH, Hichens M, Gomez HJ, Till AE, Hand EL, Vassil TC, Biollaz J, Brunner HR, Schelling JL (1982) Enalapril maleat and a lysine analogue (MK-521): disposition in man. Br J Clin Pharmacol 14: 357–362
Schaik BAM van, Geyskes GG, Boer P (1987) Lisinopril in hypertensive patients with and without renal failure. Eur J Clin Pharmacol 32: 11–16
Schaik BAM van, Geyskes GG, Wouw PA van der, Rooij HH van, Porsius AJ (1988) Pharmacokinetics of lisinopril in hypertensive patients with normal and impaired renal function. Eur J Clin Pharmacol 34: 61–65
Dettli L (1977) Elimination kinetics and dosage adjustment of drugs in patients with kidney disease. Prog Pharmacol 1: 1–34
Hichens M, Hand EL, Mulcahy WS (1981) Radioimmunoassay for angiotensin-converting enzyme inhibitors. Ligand Quarterly 4: 43
Weisser K, Schloos J, Lehmann K, Düsing R, Vetter H, Mutschler E (1991) Pharmacokinetics and converting enzyme inhibition after morning and evening administration of oral enalapril to healthy subjects. Eur J Clin Pharmacol 40: 95–99
Kwan KC, Bohidar NR, Hwang SS (1984) Estimation of an effective half-life. In: Benet LZ, Levy G (eds) Pharmacokinetics — a modern view. Plenum Press, New York, pp 147–161
Till AE, Gomez HJ, Hichens M, Bolognese JA (1984) Pharmacokinetics of repeated single oral doses of enalapril maleate (MK-421) in normal volunteers. Biopharm Drug Dispos 5: 273–280
Weisser K, Schloos J (1991) The role of enzyme and substrate concentrations in the evaluation of serum angiotensin converting enzyme (ACE) inhibition by enalaprilat in vitro. Chronobiol Int 8: 460–476
Nussberger J, Fasanella d'Amore T, Parchet M, Waeber B, Brunner DB, Brunner HR, Kler L, Brocon AR, Francis RJ (1987) Repeated administration of the CEI cilazapril to normal volunteers. J Cardiovasc Pharmacol 9: 39–44
Nussberger J, Brunner DB, Waeber B, Brunner HR (1985) True versus immunoreactive angiotensin II in human plasma. Hypertension 7 [Suppl 1]: 1–7
Hartung J, Elpelt B, Klösener KH (1985) Statistik, Oldenbourg, pp 166
Dickstein K (1987) Hemodynamic, hormonal and pharmacokinetic aspects of treatment with lisinopril in congestive heart failure. J Cardiovasc Pharmacol 9/S3: 73–81
Beermann B, Gomez HJ, Till AE, Junggren IL (1986) Pharmacokinetics of lisinopril in healthy volunteers. Acta Pharmacol Toxicol 59/S5/130: 66
Beermann B, Junggren IL, Cocchetto D, Cirillo VJ, Gomez HJ, Hichens M, Till AE, Bolognese JA (1985) Lisinopril steady-state kinetics in healthy subjects. J Clin Pharmacol 25: 471
Wade JR, Meredith PA, Hughes DM, Elliott HL (1992) The effect of saturation of ACE binding sites on the pharmacokinetics of enalaprilat in man. Br J Clin Pharmacol 33: 155–160
Shionoiri H, Minamisawa K, Ueda S, Abe Y, Ebina T, Sugimoto K, Matsukawa T, Gotoh E, Ishii M (1990) Pharmacokinetics and antihypertensive effects of lisinopril in hypertensive patients with normal and impaired renal function. J Cardiovasc Pharmacol 16: 594–600
Heeg JE, Jong PE, Van Der Hem GK, Zeeuw DD (1987) Reduction of proteinuria by angiotensin converting enzyme inhibition. Kidney Int 32: 78–83
Jackson B, Cubela RB, Conway EL, Johnston CI (1988) Lisinopril pharmacokinetics in chronic renal failure. Br J Clin Pharmacol 25: 719–724
Donohoe JF, Kelly J, Laher MS, Doyle GD (1988) Lisinopril in the treatment of hypertensive patients with renal impairment. Am J Med 85 [Suppl 3 B]: 31–34
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Neubeck, M., Fliser, D., Pritsch, M. et al. Pharmacokinetics and pharmacodynamics of lisinopril in advanced renal failure. Eur J Clin Pharmacol 46, 537–543 (1994). https://doi.org/10.1007/BF00196112
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00196112