Summary
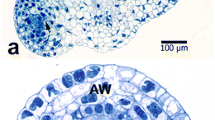
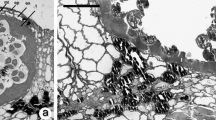
A study of pollen development in wheat was made using transmission electron microscopy (TEM). Microspores contain undifferentiated plastids and mitochondria that are dividing. Vacuolation occurs, probably due to the coalescence of small vacuoles budded off the endoplasmic reticulum (ER). As the pollen grain is formed and matures, the ER becomes distended with deposits of granular storage material. Mitochondria proliferate and become filled with cristae. Similarly, plastids divide and accumulate starch. The exine wall is deposited at a rapid rate throughout development, and the precursors appear to be synthesized in the tapetum. Tapetal cells become binucleate during the meiosis stage, and Ubisch bodies form on the plasma membrane surface that faces the locule. Tapetal plastids become surrounded by an electron-translucent halo. Rough ER is associated with the halo around the plastids and with the plasma membrane. We hypothesize that the sporopollenin precursors for both the Ubisch bodies and exine pollen wall are synthesized in the tapetal plastids and are transported to the tapetal cell surface via the ER. The microspore plastids appear to be involved in activities other than precursor synthesis: plastid proliferation in young microspores, and starch synthesis later in development. Plants treated with the chemical hybridizing agent RH0007 show a pattern of development similar to that shown by untreated control plants through the meiosis stage. In the young microspore stage the exine wall is deposited irregularly and is thinner than that of control plants. In many cases the microspores are seen to have wavy contours. With the onset of vacuolation, microspores become plasmolyzed and abort. The tapetal cells in RH0007-treated locules divide normally through the meiosis stage. Less sporopollenin is deposited in the Ubisch bodies, and the pattern is less regular than that of the control. In many cases, the tapetal cells expand into the locule. At the base of one of the locules treated with a dosage of RH0007 that causes 95% male sterility, several microspores survived and developed into pollen grains that were sterile. The conditions at the base of the locule may have reduced the osmotic stress on the microspores, allowing them to survive. Preliminary work showed that the extractable quantity of carotenoids in RHOOO7-treated anthers was slightly greater than in controls. We concluded that RH0007 appears to interfere with the polymerization of carotenoid precursors into the exine wall and Ubisch bodies, rather than interfering with the synthesis of the precursors.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bennett MD, Rao MK, Smith JB, Bayliss MW (1973) Cell development in the anther, the ovule, and the young seed of Triticum aestivum L. var. ‘Chinese Spring’. Philos Trans R Soc London Ser B 266:39–81
Brooks JS, Shaw G (1968) The post-tetrad ontogeny of the pollen wall and the chemical structure of the sporopollenin of Lilium henryi. Grana Palynol 8:227–234
Brooks MH, Brooks JS, Chien L (1966) The anther tapetum in cytoplasmic-genetic male-sterile sorghum. Am J Bot 53:902–908
Christensen JE, Horner HT, Jr (1974) Pollen pore development and its spatial orientation during microsporogenesis in the grass Sorghum bicolor. Am J Bot 61:604–623
Christensen JE, Horner HT, Jr, Lersten NR (1972) Pollen wall and tapetal orbicular wall development in Sorghum bicolor (Gramineae). Am J Bot 59:43–58
deVries AP, Ie TS (1970) Electron-microscopy on anther tissue and pollen of male-sterile and fertile wheat (Triticum aestivum L.). Euphytica 19:103–120
Dickinson JG, Helop-Harrison J (1970) The ribosome cycle, nucleoli, and cytoplasmic nucleoids in the meiocytes of Lilium. Protoplasma 69:187–200
Echlin P, Godwin H (1969) The ultrastructure and ontogeny of pollen in Helleborus foetidus L. III. The formation of the pollen grain wall. J Cell Sci 5:459–477
El-Ghazaly G, Jensen WA (1986) Studies of the development of wheat (Triticum aestivum) pollen. I. Formation of the pollen wall and Ubisch bodies. Grana 25:1–29
Guilford WJ, Schneider DM, Labovitz J, Opella SJ (1988) High-resolution solid state C-NMR spectroscopy of sporopollenins from different plant taxa. Plant Physiol 89:134–136
Heslop-Harrison J (1968) Some fine structural features of intine growth in the young microspore of Lilium henryi. Port Acta Biol Ser A 10:235–244
Heslop-Harrison J (1971) The cytoplasm and its organelles during meiosis. In: Heslop-Harrison J (ed) Pollen: development and physiology. Appleton-Century-Crofts, New York, pp 16–31
Hu S, Wang M, Hsu L (1977) Electron-microscopic observations on the microsporogenesis in male sterile and its maintainer lines of wheat. Sci Sin 20:625–630
Hu S, Zhu C, Xu L, Li X, Shen J (1979) Ultrastructure of male gametophyte in wheat. I. The formation of generative and vegetative cells. Acta Bot Sin 21:208–214
Lee SJ, Warmke HE (1979) Organelle size and number in fertile and T-cytoplasmic male-sterile corn. Am J Bot 66:141–148
Lee SJ, Earle ED, Gracen VE (1980) The cytology of pollen abortion in S cytoplasmic male-sterile corn anthers. Am J Bot 67:237–245
Lee SJ, Gracen VE, Earle ED (1979) The cytology of pollen abortion in C-cytoplasmic male-sterile corn anthers. Am J Bot 66:656–667
Maheshwari P (1950) Introduction to embryology of angiosperms. McGraw-Hill, New York
Overman MA, Warmke HE (1972) Cytoplasmic male sterility in sorghum. II. Tapetal behavior in fertile and sterile anthers. J Hered 65:227–234
Peterson RF (1965) Wheat: botany, cultivation and utilization, Interscience, New York, pp 22–23
Reynolds ES (1963) The use of lead citrate at high pH as an electron opaque stain in electron microscopy. J Cell Biol 17:208–212
Rowley JR (1962a) Nonhomogeneous sporopollenin in microspores of Poa annua L. Grana Palynol 3:3–20
Rowley JR (1962b) Stranded arrangement of sporopollenin in the exine of microspores of Poa annua. Science 137:526–528
Rowley JR (1963) Ubisch body development in Poa annua. Grana Palynol 4:25–36
Rowley JR (1964) Formation of the pore in pollen in Poa annua. In: Linskens HF (ed) Pollen physiology and fertilization. North Holland, Amsterdam, pp 59–69
Rudramuniyappa CK, Panchaksharappa MG (1980) Pollen development in Triticum durum Desf.: a histochemical study. J S Afr Bot 46:33–43
Schulz P, Jensen WA (1981) Pre-fertilization ovule development in Capsella: ultrastructure and ultracytochemical localization of acid phosphatase in the meiocyte. Protoplasma 107:27–45
Skvarla JJ, Larson DA (1966) Fine structural studies of Zea mays pollen. I. Cell membranes and exine ontogney. Am J Bot 53:1112–1125
Spurr AR (1969) A low viscosity expoxy resin embedding medium for electron microscopy. J Ultrastruct Res 26:31–43
Taylor JH (1959) Autoradiographic studies of nucleic acids and proteins during meiosis in Lilium longiflorum. Am J Bot 46:477–484
Warmke HE, Overman MA (1972) Cytoplasmic male sterility in Sorghum. I. Callose behavior in fertile and sterile anthers. J Hered 63:103–108
Warmke HE, Lee SJ (1977) Mitochondrial degeneration in Texas cytoplasmic male-sterile corn anthers. J Hered 68:213–222
Warmke HE, Lee SJ (1978) Pollen abortion in T cytoplasmic male-sterile corn (Zea mays): a suggested mechanism. Science 200:561–563
Young BA, Schulz-Schaeffer J, Carroll TW (1979) Anther and pollen development in male-sterile intermediate wheatgrass plants derived from wheat x wheatgrass hybrids. Can J Bot 57:602–618
Zhu C, Hu S, Xu L, Li W, Shen J (1979) The ultrastructure of sperm cells in wheat pollen grains (in Chinese). Sci Sin 22:1017–1021
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mizelle, M.B., Sethi, R., Ashton, M.E. et al. Development of the pollen grain and tapetum of wheat (Triticum aestivum) in untreated plants and plants treated with chemical hybridizing agent RH0007. Sexual Plant Reprod 2, 231–253 (1989). https://doi.org/10.1007/BF00195584
Issue Date:
DOI: https://doi.org/10.1007/BF00195584