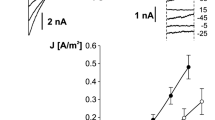
Abstract
Slowly activating vacuolar (SV-type; Hedrich and Neher 1987, Nature 329: 833–835) ion channels provide the predominant membrane conductance of the vacuolar-lysosomal compartment of Vicia faba L. guard cells and sugar beet (Beta vulgaris L.) taproots. Applying the patch-clamp technique to isolated vacuoles of both tissues, the electrical and pharmacological properties of guard-cell SV-type currents were studied and compared to the sugar beet channel with regard to its modulation by cytoplasmic Ca2+ and pH. This outward rectifier of V. faba guard cells showed a half-maximum activation at 55–60 mV with an apparent gating charge equivalent of z ≈ 4. Studies on the single-channel and whole-vacuole level revealed an extremely high conductance of 280 pS for the guard-cell channels at a mean density of 0.37 μm-2 compared to taproots (120–140 pS at about 0.16 channels per μm2). Guard-cell SV-type channels are weakly selective for cations over anions and lack saturation at KC1 concentrations of up to 1 M. Since in the absence of physiological K+ concentrations, Ca2+ is the major permeable ion, relative changes in the amounts of the two ions might control the permeation process. In spite of their different origins and physiological functions, in guard cells and beet taproot cells, cytoplasmic Ca2+ and protons, both considered as candidates for intracellular signalling in plants, modulate the voltage dependence of SV-type channels. While the two effectors do not alter the single-channel conductance, they strongly interact with the voltage sensor. The calmodulin (CaM) antagonists N-(6-aminohexyl)-5-chloro-l-naphthalenesulfonamide hydrochloride (W-7), trifluoperazine (TFP) and calmidazolium hydrochloride (R 24571) effectively blocked the channel in an antagonist-specific manner. In agreement with the properties of a Ca2+-permeable channel, CaM could be involved in the modulation of the activation threshold of the SV-type channel. We therefore conclude that guard-cell SV-type channels, which might be responsible for the release of K+, Cl- and to a smaller extent Ca2+ during stomatal closure, could serve as an intracellular sensor for changes in cytosolic calcium (calcium-CaM) and pH.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- CaM:
-
calmodulin
- Gluc- :
-
gluconate
- DIDS:
-
4,4,-diisothiocyano-2,2-stilbenedisulfonic acid
- Mes:
-
2-(N-morpholino)-ethanesulfonic acid
- R 24571:
-
calmidazolium hydrochloride
- SV:
-
slowly activating vacuolar
- TEA+ :
-
tetraethylammonium
- TFP:
-
trifluoperazine
- W-7:
-
N-(6-aminohexyl)-5-chloro-1-naphthalenesulfonamide hydrochloride
References
Aldrich RW, Yellen G (1983) Analysis of nonstationary channel kinetics. In: Sakmann B, Neher E (eds) Single channel recordings. Plenum Press, London, pp 287–300
Alexandre J, Lassalles JP, Kado RT (1990) Opening of Ca2+ channels in isolated red beet root vacuole membrane by inositol 1,4,5-trisphosphate. Nature 343: 567–570
Allen GJ, Sanders D (1994a) Two voltage-gated calcium release channels coreside in the vacuolar membrane of broad bean guard cells. Plant Cell 6: 685–694
Alien GJ, Sanders D (1994b) Osmotic stress enhances the competence of Beta vulgaris vacuoles to respond to inositol 1,4,5-triphosphate. Plant J 6: 687–695
Amodeo G, Zeiger E (1994) A cationic channel in the guard cell tonoplast of Allium cepa. Plant Physiol 105: 999–1006
Anderson JM (1983) Purification of plant calmodulin. Methods Enzymol 102: 9–16
Bentrup FW (1989) Cell physiology and membrane transport. Prog Bot 51: 70–79
Bertl A, Slayman CL (1990) Cation-selective channels in the vacuolar membrane of Saccharomyces: Dependence on calcium, redox state, and voltage. Proc Natl Acad Sci USA 87: 7824–7828
Bertl A, Blumwald E, Coronado R, Eisenberg R, Findlay G, Gradmann D, Hille B, Köhler K, Kolb HA, MacRobbie E, Meissner G, Miller C, Neher E, Palade P, Pantoja O, Sanders D, Schroeder J, Slayman C, Spanswick R, Walker A, Williams A (1992) Electrical measurements on endomembranes. Science 258: 873–874
Bethke PC, Jones RL (1994) Ca2+-Calmodulin modulates ion channel activity in storage protein vacuoles of barley aleurone cells. Plant Cell 6: 277–285
Blatt MR (1992) K+ channels of stomatal guard cells. Characteristics of the inward rectifier and its control by pH. J Gen Physiol 99: 15–644
Blatt MR, Armstrong F (1993) K+ channels of stomatal guard cells: Abscisic-acid-evoked control of the outward rectifier mediated by cytoplasmic pH. Planta 191: 330–341
Butler A, Tsunoda S, McCobb DP, Wei A, Salkhoff L (1993) mSlo, a complex mouse gene encoding “maxi” calcium-activated potassium channels. Science 261: 221–224
Colombo R, Cerana R, Lado P, Peres A (1988) Voltage-dependent channels permeable to K+ and Na+ in the membrane of Acer pseudoplatanus vacuoles. J Membr Biol 103: 227–236
Colombo R, Cerana R, Lado P, Peres A (1989) Regulation by calcium of voltage-dependent tonoplast K+ channels. Plant Physiol Biochem 27: 557–562
Colquhoun D, Sigworth FJ (1983) Fitting and statistical analysis of single-channel records. In: Sakmann B, Neher E (eds) Single channel recording. Plenum Press, London, pp 191–263
Cosgrove DJ, Hedrich R (1991) Stretch-activated chloride, potassium, and calcium channels coexisting in plasma membranes of guard cells of Vicia faba L. Planta 186: 143–153
Coyaud L, Kurkdjian A, Kado R, Hedrich R (1987) Ion channels and ATP-driven pumps involved in ion transport across the tonoplast of sugarbeet vacuoles. Biochim Biophys Acta 902: 263–268
DeBiasi M, Drewe JA, Kirsch GE, Brown AM (1993) Histidine substitution identifies a surface position and confers Cs+ selectivity on a K+ pore. Biophys J 65: 1235–1242
Evans DE, Briars SA, Williams LE (1991) Active calcium transport by plant membranes. J Exp Bot 42: 285–303
Felle H (1988a) Cytoplasmic free calcium in Riccia fluitans L. and Zea mays L.: Interaction of Ca2+ and pH? Planta 176: 248–255
Felle H (1988b) Short-term pH regulation in plants. Physiol Plant 74: 583–591
Felle H (1991) Aspects of Ca2+ homeostasis in Riccia fluitans: Reactions to perturbations in cytosolic-free Ca2+. Plant Sci 74: 27–33
Gehring CA, Irving HR, Parish RW (1990) Effects of auxin and abscisic acid on cytosolic calcium and pH in plant cells. Proc Natl Acad Sci USA 87: 9645–9649
Gilroy S, Huges WA, Trewavas AJ (1987) Calmodulin antagonists increase free cytosolic calcium levels in plant protoplasts in vivo. FEES Lett 212: 133–137
Goldman DE (1943) Potential, impedance and rectification in membranes. J Gen Physiol 352: 685–701
Hamill OP, Marty A, Neher E, Sakmann B, Sigworth FJ (1981) Improved patch-clamp-techniques for high-resolution current recording from cells and cell-free membrane patches. Pfluegers Arch 391: 85–100
Harper JF, Sussman MR, Schaller GE, Putnam-Evans C, Charbonneau H, Harmon AC (1991) A calcium-dependent protein kinase with a regulatory domain similar to calmodulin. Science 252: 951–954
Hedrich R, Kurkdjian A (1988) Characterization of an anion-permeable channel from sugar beet vacuoles: Effect of inhibitors. EMBO J 7: 3661–3666
Hedrich R, Marten I (1993) Malate-induced feedback regulation of plasma membrane anion channels could provide a CO2 sensor to guard cells. EMBO J 12: 897–901
Hedrich R, Neher, E (1987) Cytoplasmic calcium regulates voltage dependent ion channels in plant vacuoles. Nature 329: 833–835
Hedrich R, Schroeder JI (1989) The physiology of ion channels and electrogenic pumps in higher plants. Annu Rev Plant Physiol 40: 539–569
Hedrich R, Schroeder JI, Fernandez JM (1986a) Patch-clamp studies on higher plant cells: A perspective. Trends Biochem Sci 12: 49–52
Hedrich R, Schroeder JI, Fernandez JM (1986b) Patch-clamp studies of ion transport in isolated plant vacuoles. FEBS Lett 3910: 228–232
Hedrich R, Barbier-Brygoo H, Felle H, Flügge UI, Lüttge U, Matthuis FJM, Marx S, Prins HBA, Raschke K, Schnabl H, Schroeder JI, Struve I, Taiz L, Ziegler P (1988) General mechanism for solute transport across the tonoplast of plant vacuoles: A patch-clamp survey of ion channels and proton pumps. Bot Acta 101: 7–13
Hedrich R, Kurkdjian A, Guern J, Flügge UI (1989) Comparative studies on the electrical properties of the H+ translocating AT-Pase, and pyrophosphatase of the vacuolar-lysosomal compartment. EMBO J 8: 2835–2841
Hedrich R, Busch H, Raschke K (1990) Ca2+ and nucleotide dependent regulation of voltage dependent anion channels in the plasma membrane of guard cells. EMBO J 9: 3889–3892
Hedrich R, Marten I, Lohse G, Dietrich P, Winter H, Lohaus G, Heldt HW (1994) Malate-sensitive anion channels enable guard cells to send changes in the ambient CO2 concentration. Plant J 6: 44–50
Hepler PK, Wayne RO (1985) Calcium and plant development. Annu Rev Plant Physiol 36: 397–439
Hille B (1992) Ionic channels of excitable membranes. Sinauer Associates Inc. Publishers, Sunderland, Massachusetts
Hodgkin AL, Katz B (1949) The effect of sodium ions on the electrical acitvity of the giant axon of the squid. J Physiol 108: 37–77
Humble GD, Raschke K (1971) Stomatal opening quantitatively related to potassium transport. Plant Physiol 48: 447–453
Ilan N, Schwartz A, Moran N (1994) External pH effects on the depolarization-activated K channels in guard cell protoplasts of Vicia faba. J Gen Physiol 103: 807–831
Irving HR, Gehring CA, Parish RW (1992) Changes in cytosolic pH and calcium of guard cells precede stomatal movements. Proc Natl Acad Sci USA 89: 1790–1794
Johannes E, Brosnan JM, Sanders D (1992) Parallel pathways for intracellular Ca2+ release from the vacuole of higher plants. Plant J 2: 97–102
Keller BU, Hedrich R, Raschke K (1989) Voltage-dependent anion channels in the plasma membrane of guard cells. Nature 341: 450–453
Lemtiri-Chlieh F, MacRobbie EAC (1994) Role of calcium in the modulation of Vicia guard cell potassium channels by ABA: A patch-clamp study. J Membr Biol 137: 99–107
Lewis CA (1979) Ion-concentration dependence of the reversal potential and the single channel conductance of ion channels at the frog neuromuscular junction. J Physiol 286:17–445
Llinás R, Sugimori M, Silver RB (1991) Imaging preterminal calcium concentration microdomains in the squid giant synapse. Biol Bull 181: 316–317
Maathuis FJM, Prins HBA (1991a) Patch clamp studies on root cell vacuoles of a salt-tolerant and a salt-sensitive Plantago species. Plant Physiol 92: 23–28
Maathuis FJM, Prins HBA (1991b) Inhibition of inward rectifying tonoplast channels by a vacuolar factor: Physiological and kinetic implications. J Membr Biol 122: 251–258
Mackion AES (1984) Calcium fluxes at plasmalemma and tonoplast. Plant Cell Environ. 7: 407–413
MacRobbie EAC (1988) Control of ion fluxes in stomatal guard cells. Bot Acta 101: 140–148
MacRobbie EAC (1990) Calcium-dependent and calcium-independent events in the initiation of stomatal closure by abscisic acid. Proc Soc Lond B 241: 214–219
Matile P (1978) Biochemistry and function of vacuoles. Annu Rev Plant Physiol 29: 193–213
Miller AJ, Sanders D (1987) Depletion of cytosolic free calcium induced by photosynthesis. Nature 326: 397–400
Neher E (1992) Corrections for liquid junction potentials in patch-clamp experiments. Methods Enzymol 207: 123–131
Pantoja O, Dainty J, Blumwald E (1989) Ion channels in vacuoles from halophytes and glycophytes. FEBS Lett 255: 92–96
Pantoja O, Gelli A, Blumwald E (1992a) Voltage-dependent calcium channels in plant vacuoles. Science 255: 1567–1570
Pantoja O, Dainty J, Blumwald E (1992b) Cytoplasmic chloride regulates cation channels in the vacuolar membrane of plant cells. J Membr Biol 125: 219–229
Penny MG, Bowling DJF (1974) A study of potassium gradients in the epidermis of intact leaves of Commelina communis L. in relation to stomatal opening. Planta 119: 17–25
Perry CA, Leigh RA, Tomos AD, Wyse RE, Hall JL (1987) The regulation of turgor pressure during sucrose mobilisation and salt accumulation by excised storage-root tissue of red beet. Planta 170: 353–361
Raschke K (1979) Movements of stomata. In: Haupt W, Feinleib ME (eds) Encyclopedia of plant physiology. Vol. 7: Physiology of movements. Springer Verlag, Berlin, pp 383–441
Raschke K, Hedrich R (1989) Patch clamp measurements on isolated guard cell protoplasts and vacuoles. Methods Enzymol 174: 312–329
Roberts WM, Jacobs RA, Hudspeth AJ (1990) Colocalization of ion channels involved in frequency selectivity and synaptic transmission at presynaptic active zones in hair cells. J Neurosci 10: 3664–3684
Robinson RA, Stokes RH (1959) Electrolyte solutions. Butterworths Scientific Publications, London
Sanders D, Johannes E, Hedrich R (1990) Opening plant calcium channels. Nature 344: 593–594
Schroeder JI, Hagiwara S (1989) Cytosolic calcium regulates ion channels in the plasma membrane of Vicia faba guard cells. Nature 338: 427–430
Schroeder JI, Hedrich R (1989) Involvement of ion channels and active transport in osmoregulation and signaling of higher plant cells. Trends Biol Sci 14: 187–192
Schroeder JI, Raschke K, Neher E (1987) Voltage dependence of K+ channels in guard cell protoplasts. Proc Natl Acad Sci USA 84: 4108–4112
Schulz-Lessdorf B, Dietrich P, Marten I, Lohse G, Busch H, Hedrich R (1994) Coordination of plasma membrane and vacuolar membrane ion channels during stomatal movement. In: Blatt MR, Leigh RA, Sanders D (eds) Membrane transport in plants and fungi: molecular mechanisms and control (Symposia of the Society of Experimental Biology, Number XLVIII) The Company of Biologists Ltd, Cambridge, pp 99–112
Schulz-Lessdorf B, Hedrich R (1992) pH and Ca2+ modulate the activity of ion channels in the vacuolar membrane of guard cells-possible interaction with calmodulin. Poster Abstract [377], Botanikertagung Berlin
Siebke K, Yin ZH, Raghavendra AS, Heber U (1992) Vacuolar pH oscillations in mesophyll cells accompany oscillations of photosynthesis in leaves: Interdependence of cellular compartments, and regulation of electron flow in photosynthesis. Planta 186: 526–531
Sigworth F (1983) An example of analysis. In: Sakmann B, Neher E (eds) Single channel recording. Plenum Press, London, pp 301–321
Speer M, Kaiser WM (1991) Ion relations of symplastic and apoplastic space in leaves from Spinacia oleracea L. and Pisum sativum L. under salinity. Plant Physiol 97: 990–997
Sze H (1985) H+-translocating ATPases: Advances using membrane vesicles. Annu Rev Plant Physiol 36: 175–208
Tarczynski MC, Outlaw WH Jr (1990) Kinetic datum collection in real time from single cell activities. Arch. Biochim Biophys 280: 71
Thuleau P, Ward JM, Ranjeva R, Schroeder JI (1994) Voltage-dependent calcium-permeable channels in the plasma membrane of a higher plant cell. EMBO J 13: 2970–2975
Wallace RW, Tallant EA, Cheung WY (1983) Assay of calmodulin by Ca2+-dependent phosphodiesterase. Methods Enzymol 102: 39–46
Ward JM, Schroeder JI (1994) Calcium-activated K+ channels and calcium-induced calcium release by slow vacuolar ion channels in guard cell vacuoles implicated in the control of stomatal closure. Plant Cell 6: 669–683
Weiser T, Bentrup FW (1990) (+)-Tubocurarine is a potent inhibitor of cation channels in the vacuolar membrane of Chenopodium rubrum L. FEBS Lett 277: 220–222
Weiser T, Blum W, Bentrup FW (1991) Calmodulin regulates the Ca2+-dependent slow-vacuolar ion channel in the tonoplast of Chenopodium rubrum suspension cells. Planta 185: 440–442
Yin ZH, Neimanis S, Wagner U, Heber U (1990) Light-dependent pH changes in leaves of C3 plants.I. Recording pH changes in various cellular compartments by fluorescent probes. Planta 182: 244–252
Author information
Authors and Affiliations
Additional information
We thank R. Benz (Institute of Biotechnology, Würzburg, Germany), F. Conti, (Istituto di cibernetica e Biofisica, Genova, Italy) and C.L. Slayman (Department of Cellular and Molecular Physiology, Yale School of Medicine, New Haven, Conn., USA) for helpful discussion of the manuscript and C. Zeilinger (Institute of Biophysics, Hannover, Germany) and G. Hinz (Institute of Plant Physiology, Göttingen, Germany) for purification of wheat-germ CaM. Studies were funded by Deutsche Forschungsgemeinschaft grants to R.H.
Rights and permissions
About this article
Cite this article
Schulz-Lessdorf, B., Hedrich, R. Protons and calcium modulate SV-type channels in the vacuolar-lysosomal compartment — channel interaction with calmodulin inhibitors. Planta 197, 655–671 (1995). https://doi.org/10.1007/BF00191574
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00191574