Summary
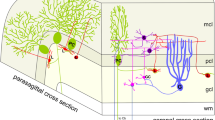
This investigation attempted to determine the mode of distribution and synaptic targets of the cartwheel cell axon in the guinea pig dorsal cochlear nucleus (DCoN). Antiserum against PEP-19, a putative calcium-binding neuropeptide, was employed at the light and electron microscopic levels. We show that in the hindbrain of the guinea pig, cerebellar Purkinje cells and DCoN cartwheel cells are the most densely immunoreactive neurons. The PEP-19 immunoreaction product is localized to all neuronal compartments of these cells. Primary targets of cartwheel cell axons are the DCoN pyramidal cells, the large efferent neurons of layer 2. These neurons receive numerous immunoreactive synaptic boutons on their cell bodies and apical and basal dendritic arbors. A PEP-19-immunoreactive axonal plexus, largely formed by cartwheel cell axons, highlights layer 3, co-extensively with the basal arbors of pyramidal cells. This plexus is oriented predominantly in the transstrial plane of the DCoN, in parallel with the sheetlike basal dendritic arbor of pyramidal neurons and with the isofrequency bands of primary cochlear nerve fibers. PEP-19-positive boutons contain pleomorphic synaptic vesicles and form symmetric synaptic junctions, indicative of inhibitory innervation. In addition, immunoreactive boutons, similar to those synapsing on pyramidal neurons, were observed on the cell bodies and main dendritic trunks of cartwheel neurons, indicating a system of recurrent collaterals. Furthermore, a small number of PEP-19-positive axons of unknown origin reach the caudal rim of the posteroventral cochlear nucleus. Within the territory of distribution of the cartwheel cell axon are the dendrites of at least two other types of DCoN neuron, the vertical cells of Lorente de Nó and the giant cells. These neurons may represent additional targets of the cartwheel cell axon, but this remains to be ascertained with specific methods. Our data demonstrate that the cartwheel neurons modulate the activity of pyramidal neurons and, therefore, play a key role in shaping the output of the DCoN superficial layers.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adams JC, Mugnaini E (1987) Patterns of glutamate decarboxylase immunostaining in the feline cochlear nuclear complex studied with silver enhancement and electron microscopy. J Comp Neurol 262:375–401
Arnesen AR, Osen KK (1978) The cochlear nerve in the cat: topography, cochleotopy and fiber spectrum. J Comp Neurol 178:661–678
Berrebi AS, Mugnaini E (1987) The dorsal cochlear nucleus in mutant mice. Anat Rec 218:17A
Berrebi AS, Mugnaini E (1988a) Targets and distribution of the cartwheel cell axon in the guinea pig dorsal cochlear nucleus. Soc Neurosci Abstr 14:489
Berrebi AS, Mugnaini E (1988b) Effects of the murine mutation ‘nervous’ on neurons in cerebellum and dorsal cochlear nucleus. J Neurocytol 17:465–484
Berrebi AS, Morgan JI, Mugnaini E (1987) Loss of cartwheel neurons in the dorsal cochlear nucleus of the mouse mutants, Lurcher, Purkinje Cell Degeneration and Staggerer. Soc Neurosci Abstr 13:1259
Berrebi AS, Morgan JI, Mugnaini E (1990) The Purkinje cell class may extend beyond the cerebellum. J Neurocytol 19:643–654
Blackstad TW, Osen KK, Mugnaini E (1984) Pyramidal neurons of the dorsal cochlear nucleus: a Golgi and computer reconstruction study in cat. Neurosci 13:827–854
Caspary DM (1990) Electrophysiological studies of glycinergic mechanisms in auditory brainstem structures. In: Ottersen OP, Storm-Mathisen J (eds) Glycine neurotransmission. Wiley, Chichester, pp 453–483
Caspary DM, Pazara KE, Kossl M, Faingold CL (1987) Strychnine alters the fusiform cell output from the dorsal cochlear nucleus. Brain Res 417:273–282
Cohen ES, Brawer JR, Morest DK (1972) Projections of the cochlea to the dorsal cochlear nucleus in the cat. Exp Neurol 35:470–479
DeCamilli P, Miller PE, Levitt P, Walter U, Greengard P (1984) Anatomy of cerebellar Purkinje cells in the rat determined by a specific immunohistochemical marker. Neurosci 11:761–817
Dunn ME, Vetter DE, Berrebi AS, Krider HM, Mugnaini E (1988) Different types of mossy fiber terminals in the guinea pig cochlear nucleus. Soc Neurosci Abstr 14:487
Dunn ME, Vetter DE, Berrebi AS, Krider HM, Mugnaini E (1992) The mossy fiber-granule cell-cartwheel cell system in the mammalian cochlear complex. In: Ainsworth WA, Evans EF, Hackney CM (eds) Advance in Speech, Hearing and Language Processing. Vol. III, Cochlear nucleus: structure and function in relation to modelling. JAI Press Ltd., London, in press
Friedrich VL Jr, Mugnaini E (1981) Electron microscopy: preparation of neural tissues for electron microscopy. In: Heimer L, Robards MJ (eds) Neuroanatomical tract-tracing methods. Plenum Press, New York, pp 345–376
Godfrey DA, Kiang NYS, Norris BE (1975) Single unit activity in the dorsal cochlear nucleus of the cat. J Comp Neurol 162:269–284
Hackney CM, Osen KK, Kolston J (1990) Anatomy of the cochlear nuclear complex of guinea pig. Anat Embryol 182:123–149
Harvey JA, Winsky L, Schindler CW, McMaster SE, Welsh JP (1988) Asymmetric uptake of 2-deoxy-D-[14C] glucose in the dorsal cochlear nucleus during Pavlovian conditioning in the rabbit. Brain Res 449:213–224
Hirsch JA, Oertel D (1988) Synaptic connections in the dorsal cochlear nucleus of mice, in vitro. J Physiol (Lond) 396:549–562
Itoh K, Kamiya H, Mitani A, Yasui Y, Takada M, Mizuno N (1987) Direct projections from the dorsal column nuclei and the spinal trigeminal nuclei to the cochlear nuclei in cat. Brain Res 400:145–150
Jack JJB, Noble D, Tsien RW (1975) Electric current flow in excitable cells. Clarendon Press, Oxford University Press, London
Jahnsen H (1986a) Electrophysiological characteristics of neurones in the guinea-pig deep cerebellar nuclei in vitro. J Physiol (Lond) 372:129–147
Jahnsen H (1986b) Extracellular activation and membrane conductances of neurones in the guinea-pig deep cerebellar nuclei in vitro. J Physiol (Lond) 372:149–168
Kane ES (1974) Synaptic organization in the dorsal cochlear nucleus of the cat: a light and electron microscopic study. J Comp Neurol 155:301–330
Llinás R, Mühlethaler M (1988a) An electrophysiological study of the in vitro, perfused brain stem-cerebellum of the adult guinea-pig. J Physiol (Lond) 404:215–240
Llinás R, Mühlethaler M (1988b) Electrophysiology of guinea-pig cerebellar nuclear cells in the in vitro brain stem-cerebellum preparation. J Physiol (Lond) 404:241–258
Lorente de Nó R (1933) Anatomy of the eighth nerve. III. General plan of structure of the primary cochlear nuclei. Laryngoscope 43:327–350
Lorente de Nó R (1981) The primary acoustic nuclei. Raven Press, New York
Manis PB (1988) Intracellular recordings from pyramidal cells of the guinea pig dorsal cochlear nucleus. J Neurosci Meth 24:201
Manis PB (1989) Responses to parallel fiber stimulation in the guinea pig dorsal cochlear nucleus in vitro. J Neurophysiol 61:149–161
Manis PB (1990) Membrane properties and discharge characteristics of guinea pig dorsal cochlear nucleus neurons studied in vitro. J Neurosci 10:2338–2351
Manis PB, Brownell WE (1983) Synaptic organization of eighth nerve afferents to the cat dorsal cochlear nucleus. J Neurophysiol 50:1156–1181
Masterton RB, Granger EM (1988) Role of the acoustic striae in hearing: contribution of dorsal and intermediate striae to detection of noises and tones. J Neurophysiol 60:1841–1860
Mignery GA, Südhof TC, Takei K, DeCamilli P (1989) Putative receptor for inositol 1,4,5-trisphosphate similar to ryanodine receptor. Nature 342:192–195
Moore JK, Moore RY (1987) Glutamic acid decarboxylase-like immunoreactivity in brainstem auditory nuclei of the rat. J Comp Neurol 260:157–174
Mugnaini E (1985) GABA neurons in the superficial layers of the rat dorsal cochlear nucleus: light and electron microscopic immunocytochemistry. J Comp Neurol 235:61–81
Mugnaini E, Dahl A-L (1983) Zinc-aldehyde fixation for light microscopic immunocytochemistry of nervous tissues. J Histochem Cytochem 31:1435–1438
Mugnaini E, Morgan JI (1987) The neuropeptide cerebellin is a marker for two similar neuronal circuits in rat brain. Proc Natl Acad Sci USA 84:8692–8696
Mugnaini E, Oertel WH (1985) An atlas of the distribution of GABA-ergic neurons and terminals in the rat CNS as revealed by GAD immunohistochemistry. In: Björklund A, Hökfelt T (eds) Handbook of chemical neuroanatomy, vol 4. Elsevier, Amsterdam New York, pp 436–608
Mugnaini E, Osen KK, Dahl A-L, Friedrich VL Jr, Korte G (1980a) Fine structure of granule cells and related interneurons (termed Golgi cells) in the cochlear nuclear complex of the cat, rat and mouse. J Neurocytol 9:537–570
Mugnaini E, Warr WB, Osen KK (1980b) Distribution and light microscopic features of granule cells in the cochlear nuclei of the cat, rat and mouse. J Comp Neurol 191:581–606
Mugnaini E, Berrebi AS, Dahl A-L, Morgan JI (1987) The polypeptide PEP-19 is a marker for Purkinje neurons in cerebellar cortex and cartwheel neurons in dorsal cochlear nucleus. Arch Ital Biol 126:41–67
Mugnaini E, Dahl A-L, Morgan JI (1988) Cerebellin is a postsynaptic neuropeptide. Synapse 2:125–138
Nieuwenhuys R. Nicholson C (1969) Aspects of the histology of the cerebellum of mormyrid fishes. In: Llinás R (ed) Neurobiology of cerebellar evolution and development. American Medical Association, Chicago, pp 135–169
Noda Y, Pirsig W (1974) Anatomical projection of the cochlea to the cochlear nuclei of the guinea pig. Arch Otorhinolaryngol 208:107–120
Noort J van (1969) The structure and connections of the inferior colliculus. An investigation of the lower auditory system. Van Gorcum, Leiden
Oertel D, Wu SH (1989) Morphology and physiology of cells in slice preparations of the dorsal cochlear nucleus of mice. J Comp Neurol 283:228–247
Oliver DL, Potashner SJ, Jones DR, Morest DK (1983) Selective labeling of spiral ganglion and granule cells with D-aspartate in the auditory system of cat and guinea pig. J Neurosci 3:455–472
Osen KK (1970) Course and termination of the primary afferents in the cochlear nuclei of the cat. An experimental anatomical study. Arch Ital Biol 108:21–51
Osen KK (1985) “Ectopic” neurons of the cochlear nuclei. Neurosci Lett 22 [Suppl]:S167
Osen KK (1988) Anatomy of the mammalian cochlear nuclei: a review. In: Syka J, Masterton RB (eds) Auditory pathway-structure and function. Plenum Press, London New York, pp 65–76
Osen KK, Ottersen OP, Storm-Mathisen J (1987) Glycine-like immunoreactivity in the rat dorsal cochlear nucleus (DCN). Neuroscience 22 [Suppl]:S788
Osen KK, Ottersen OP, Storm-Mathisen J (1990) Colocalization of glycine-like and GABA-like immunoreactivities. A semiquantitative study of individual neurons in the dorsal cochlear nucleus of cat. In: Ottersen OP, Storm-Mathisen J (eds) Glycine neurotransmission. Wiley, Chichester, pp 417–451
Rall W (1964) Theoretical significance of dendritic trees for neuronal input-output relations. In: Reiss RF (ed) Neural theory and modeling. Proceedings of the 1962 Ojai Symposium. Stanford University Press, Standford, pp 73–97
Rhode WS, Smith PH, Oertel D (1983) Physiological response properties of cells labeled intracellularly with horseradish peroxidase in cat dorsal cochlear nucleus. J Comp Neurol 213:426–447
Roberts RC, Ribak CE (1987) GABAergic neurons and axon terminals in the brainstem auditory nuclei of the gerbil. J Comp Neurol 258:267–280
Rose JE, Galambos R, Hughes JR (1959) Microelectrode studies of the cochlear nuclei of the cat. Bull Johns Hopkins Hosp 104:211–251
Ryan AF, Furlow Z, Woolf NK, Keithley EM (1988) The spatial representation of frequency in the rat dorsal cochlear nucleus and inferior colliculus. Hear Res 36:181–190
Sando I (1965) The anatomical interrelationships of the cochlear nerve fibers. Acta Otolaryngol (Stockh) 59:417–436
Smith PH, Rhode WS (1985) Electron microscopic features of physiologically characterized, HRP-labeled fusiform cells in the cat dorsal cochlear nucleus. J Comp Neurol 237:127–143
Sternberger LA (1979) Immunocytochemistry, 2nd edn. Wiley, New York
Weinberg RJ, Rustioni A (1987) A cuneocochlear pathway in the rat. Neuroscience 20:209–219
Weinberg RJ, Rustioni A (1989) Brainstem projections to the rat cuneate nucleus. J Comp Neurol 282:142–156
Wenthold RJ, Hunter C (1990) Immunocytochemistry of glycine and glycine receptors in the central auditory system. In: Ottersen OP, Storm-Mathisen J (eds) Glycine neurotransmission. Wiley, Chichester, pp 391–416
Wenthold RJ, Zempel JM, Parakkal MH, Reeks KA, Altschuler RA (1986) Immunocytochemical localization of GABA in the cochlear nucleus of the guinea pig. Brain Res 380:7–18
Wenthold RJ, Huie D, Altschuler RA, Reeks KA (1987) Glycine immunoreactivity localized in the cochlear nucleus and superior olivary complex. Neuroscience 22:897–912
Wickesberg RE, Oertel D (1988) Tonotopic projection from the dorsal to the anteroventral cochlear nucleus of mice. J Comp Neurol 268:389–399
Wouterlood FG, Mugnaini E (1984) Cartwheel neurons of the dorsal cochlear nucleus. A Golgi-electron microscopic study in the rat. J Comp Neurol 227:136–157
Wouterlood FG, Mugnaini E, Osen KK, Dahl A-L (1984) Stellate neurons in the dorsal cochlear nucleus of the rat studied with Golgi-impregnation electron microscopy; synaptic connections and mutual coupling by gap junctions. J Neurocytol 13:639–664
Young ED (1984) Response characteristics of neurons in the cochlear nuclei. In: Berlin C (ed) Hearing science. College Hill Press, San Diego, pp 423–460
Young ED, Brownell WE (1976) Responses to tones and noise of single cells in dorsal cochlear nucleus of unanesthetized cats. J Neurophysiol 39:282–300
Ziai R, Pan Y-CE, Hulmes JD, Sangameswaran L, Morgan JI (1986) Isolation, sequence, and developmental profile of a brain-specific polypeptide, PEP-19. Proc Natl Acad Sci USA 83:8420–8423
Ziai MR, Sangameswaran L, Hempstead JL, Danho W, Morgan JI (1988) An immunochemical analysis of the distribution of a brain-specific polypeptide, PEP-19. J Neurochem 51:1771–1776
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Berrebi, A.S., Mugnaini, E. Distribution and targets of the cartwheel cell axon in the dorsal cochlear nucleus of the guinea pig. Anat Embryol 183, 427–454 (1991). https://doi.org/10.1007/BF00186433
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00186433