Abstract
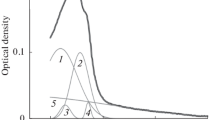
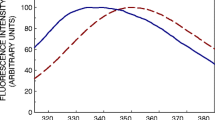
The environment of aromatic aminoacids in the thermal transition of brain tubulin has been studied by several spectroscopic techniques (Fourth Derivative, Difference Absorption, Fluorescence and Circular Ditchroism), in order to study its denaturation. An irreversible, temperature-induced, structural transition was found at around 48°C. In order to establish the relative degree of hydrophobicity of tubulin aromatic residues, before and after the thermal transition, difference and fourth derivative absorption spectra at different temperatures were compared with spectra of tyrosine and tryptophan model compounds in different media. It was found that at high temperatures, tubulin acquires a partially denatured stable state, with a significant amount of residual structure still preserved. This state is characterized by a general increase of the exposure of tyrosine residues to the medium, while the environment of tryptophans becomes more hydrophobic.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Andreu JM, Muñoz JA (1986) Interaction of tubulin with octyl glucoside and deoxycholate. 1 Binding and hydrodynamic studies. Biochemistry 25:5220–5230
Andreu JM, Timasheff SN (1982) Conformational states of tubulin liganded to colchicine, tropolone methyl ether and podophyllotoxin. Biochemistry 21:6465–6476
Andreu JM, de la Torre J, Carrascosa JL (1986) Interaction of tubulin with octyl glucoside and deoxycholate. 2 protein conformation, binding of colchicine ligands and microtubule assembly. Biochemistry 25:5231–5239
Audenaert R, Heremans L, Heremans K, Engelborghs Y (1989) Secondary structure analysis of tubulin and microtubules with Raman spectroscopy. Biochim Biophys Acta 996:110–115
Bello J (1977) Interpretation of thermal perturbation spectra of proteins. Int J Peptide Protein Res 10:71–79
Butler WL (1979) Fourth derivative spectra. Methods Enzymol 56:501–515
Clark CD, Martin SR, Bayley PM (1981) Conformation and assembly characteristics of tubulin and microtubule protein from bovine brain. Biochemistry 20:1924–1932
De la Viña S, Andreu A, Medrano FJ, Nieto JM, Andreu JM (1988) Tubulin structure probed with antibodies to synthetic peptides. Mapping of their major types of limited proteolysis fragments. Biochemistry 27:5352–5365
Demchenko AS, Ladokhin AS (1988) Temperature-dependence shift of fluorescence spectra without conformational changes in protein: studies of dipolar relaxation in the melittin molecule. Biochim Biophys Acta 955:352–360
Donovan JW(1973) UV difference spectroscopy. New techniques and applications. Methods Enzymol 27:497–525
Duñach M, Sabés M, Padrós E (1983) Fourth derivative spectrophotometry analysis of tryptophan environment in proteins. Eur J Biochem 134:123–128
Greenfield N, Fasman G (1969) Computed circular dichroism spectra for the evaluation of protein conformation. Biochemistry 8:4108–4116
Hastie SB (1989) Spectroscopic and kinetic features of allocolchicine binding to tubulin. Biochemistry 28:7753–7760
Kuznetsova IM, Khaitlina SYu, Konditerov SN, Surin AM, Turoverov KK (1988) Changes of structure and intramolecular mobility in the course of Actin denaturation. Biophys Chem 32:73–78
Lakowicz JR (1983) Principles of fluorescence spectroscopy. Plenum Press, New York
Lee LG, Frigon RP, Timasheff FN (1973) The chemical characterization of calf brain microtubule protein subunits. J Biol Chem 248:7253–7262
Lee JC, Corfman D, Frigon RP, Timasheff SN (1978) Conformational study of calf brain tubulin. Arch Biochem Biophys 185:4–14
Maccioni RM (1983) Microtubule assembly affected by the presence of denaturated tubulin. Biochim Biophys Res Commun 110:463–469
Maccioni RB, Seeds NW (1982) Involvement of tryptophan residues in colchicine binding and the assembly of tubulin. Biochim Biophys Res Commun 108:896–903
Na GC, Timasheff SN (1981) Interaction of calf brain tubulin with glycerol. J Mol Biol 151:165–178
Nicola NA, Leach SJ (1976) Interpretation and applications of thermal difference spectra of proteins. Int J Peptide Protein Res 8:393–415
Padrós F, Duñach M, Morros A, Sabés M, Mañosa J (1984) Fourth-derivative spectrophotometry of proteins. Trends Biochem Sci 9:508–510
Padrós E, Morros A, Mañosa J, Duñach M (1982) The state of tyrosine and phenylalanine residues in proteins analyzed by fourth-derivative spectrophotometry. Eur J Biochem 127:117–122
Ponstingl H, Krauhs E, Little M, Kempf T, Hoffer-Warbinek R, Ade W (1982) Aminoacid sequence of α- and β-tubulins from pig brain: heterogeneity and regional similarity to muscle proteins. Cold Spring Harbor Symp. Quant. Biol. 46:191–197
Prakash V, Timasheff SN (1982) Ageing of tubulin at neutral pH. J Mol Biol 160:499–515
Weisenberg RC, Borisy IY, Taylor E (1968) The colchicine-binding protein of mammalian brain and its relation to microtubules. Biochemistry 7:4466–4479
Author information
Authors and Affiliations
Additional information
Offprint requests to: A. Mozo-Villarías
Rights and permissions
About this article
Cite this article
Mozo-Villarías, A., Morros, A. & Andreu, J.M. Thermal transitions in the structure of tubulin. Eur Biophys J 19, 295–300 (1991). https://doi.org/10.1007/BF00183318
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00183318