Abstract
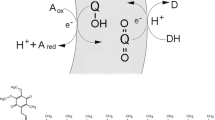
The flux of amino acids and other nutrient solutes such as phosphate across lipid bilayers (liposomes) is 105 slower than facilitated inward transport across biological membranes. This suggests that primitive cells lacking highly evolved transport systems would have difficulty transporting sufficient nutrients for cell growth to occur. There are two possible ways by which early life may have overcome this difficulty: (1) The membranes of the earliest cellular life-forms may have been intrinsically more permeable to solutes; or (2) some transport mechanism may have been available to facilitate transbilayer movement of solutes essential for cell survival and growth prior to the evolution of membrane transport proteins. Translocation of neutral species represents one such mechanism. The neutral forms of amino acids modified by methylation (creating protonated weak bases) permeate membranes up to 1010 times faster than charged forms. This increased permeability when coupled to a transmembrane pH gradient can result in significantly increased rates of net unidirectional transport. Such pH gradients can be generated in vesicles used to model protocells that preceded and were presumably ancestral to early forms of life. This transport mechanism may still play a role in some protein translocation processes (e.g., for certain signal sequences, toxins and thylakoid proteins) in vivo.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- LUV:
-
large unilamellar vesicle
- ΔpH:
-
transmembrane pH gradient
- PAH:
-
polyaromatic hydrocarbon
References
Boyd D, Beckwith J (1990) The role of charged amino acids in the localization of secreted and membrane proteins. Cell 62:1031–1033
Chakrabarti AC, Deamer DW (1992) Permeability of lipid bilayers to amino acids and phosphate. Biochim Biophys Acta 1111: 171–177
Chakrabarti AC, Clark-Lewis I, Harrigan PR, Cullis PR (1992) Uptake of basic amino acids and peptides into liposomes in response to transmembrane pH gradients. Biophys J 61:228–234
Chakrabarti AC (1994) Permeability of membranes to amino acids and modified amino acids: mechanisms involved in translocation. Amino Acids (in press)
Chakrabarti AC, Clark-Lewis 1, Cullis PR (1994) Influence of charge, charge distribution and hydrophobicity on the uptake of short model peptides into liposomes in response to transmembrane pH gradients. Biochemistry (submitted)
Cline K, Ettinger WF, Theg SM (1992) Protein-specific energy requirements for protein transport across or into thylakoid membranes: two lumenal proteins are transported in the absence of ATP. J Biol Chem 267:2688–2696
Crofts AR (1967) Amine uncoupling and energy transfer in chloroplasts. J Biol Chem 242:3352–3359
Deamer DW, Prince RC, Crofts AR (1972) The response of fluorescent amines to pH gradients across liposome membranes. Biochim Biophys Acta 274:323–335
Deamer DW, Oro J (1980) Role of lipids in prebiotic structures. Biosystems 12:167–175
Deamer DW (1986) Role of amphiphilic compounds in the evolution of membrane structure on the early earth. Orig Life Evol Biosphere 17:3–25
Deamer DW, Pashley RM (1989) Amphiphilic components of the Murchison carbonaceous chondrite: surface properties and membrane formation. Orig Life Evol Biosphere 19:21–38
Deamer DW, Harang E (1990) Light-dependent pH gradients are generated in liposomes containing ferrocyanide. Biosystems 24: 1–4
Deamer DW (1992a) Polycyclic aromatic hydrocarbons: primitive pigment systems in the prebiotic environment. Adv Space Res 12: 183–189
Deamer DW (1992b) Prebiotic conditions and the first living cells. In: Lipps J (ed) Fossil prokaryotes and protists. Blackwell Scientific, Oxford, pp 11–18
Gierasch L (1989) Signal sequences. Biochemistry 28:923–930
Hope MJ, Redelmeier TE, Wong KF, Rodrigueza W, Cullis PR (1989) Phospholipid asymmetry in large unilamellar vesicles induced by transmembrane pH gradients. Biochemistry 28:4181–4187
Madden TD, Harrigan PR, Tai LCL, Bally MB, Mayer LD, Redelmeier TE, Loughrey HC, Tilcock CPS, Reinish LW, Cullis PR (1990) The accumulation of drugs within large unilamellar vesicles exhibiting a proton gradient: a survey. Chem Phys Lipids 53: 37–46
Maduke M, Roise D (1993) Import of s mitochondrial presequence into protein-free phospholipid vesicles. Science 260:364–367
Morowitz HJ, Heinz B, Deamer DW (1988) The chemical logic of a minimum protocell. Orig Life Evol Biosphere 18:281–287
Morowitz HJ, Deamer DW, Smith T (1991) Biogenesis as a evolutionary process. J Mol Evol 33:207–208
Morowitz HJ (1992) Beginnings of cellular life. Yale University Press, New Haven, CT
Nooner DW, Sherwood E, More MA, Oro J (1977) Cyanamide mediated syntheses under plausible primitive earth conditions. III: synthesis of peptides. J Mol Evol 10:211–220
Olsnes S, Moskaug JO, Stenmark H, Sandvig K (1988) Diphtheria toxin entry: protein translocation in the reverse direction. Trends Biochem Sci 13:348–351
Parker MW, Tucker AD, Tsernoglou D, Pattus F (1990) Insights into membrane insertion based on studies of colicins. Trends Biochem Sci 15:126–129
Rapoport TA (1992) Transport of proteins across the endoplasmic reticulum membrane. Science 258:931–936
Rees-Jones R, Al-Awgati Q (1984) Proton-translocating adenosinetriphosphatase in rough and smooth microsomes from rat liver. Biochemistry 23:2236–2240
Schuldiner S, Fiskes H, Kanner BI (1978) Role of transmembrane pH gradient in epinephrine transport by chromafin granule membrane vesicles. Proc Natl Acad Sci 75:3713–3716
Segel I (1976) Biochemical calculations, 2nd ed. John Wiley, New York, p 81
Skerjanc I (1990) Mitochondrial import: properties of precursor proteins. Biochem Cell Biol 68:9–16
Stillwell W (1976) Facilitated diffusion of amino acids across bimolecular lipid membranes as a model for selective accumulation of amino acids in a primordial protocell. Biosystems 8:111–117
Thevenod F, Kemmer TP, Christian AL, Schultz I (1989) Characterization of MgATP-driven H+ uptake into a microsomal vesicle fraction from rat pancreatic acinar cells. J Membr Biol 107:263–275
Author information
Authors and Affiliations
Additional information
Correspondence to: A.C. Chakrabarti
Rights and permissions
About this article
Cite this article
Chakrabarti, A.C., Deamer, D.W. Permeation of membranes by the neutral form of amino acids and peptides: Relevance to the origin of peptide translocation. J Mol Evol 39, 1–5 (1994). https://doi.org/10.1007/BF00178243
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00178243