Summary
Experiments in slices of rat nucleus accumbens were carried out in order to investigate whether the release of acetylcholine in this tissue is modulated through dopamine receptors. The slices were preincubated with 3H-choline and then superfused and stimulated electrically twice for 2 min each at a frequency of 3 Hz.
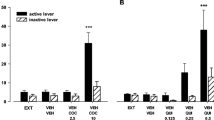
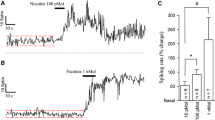
The electrically evoked overflow of tritium averaged 2.9–3.9% of the tritium content of the tissue in the various groups. The D2-selective agonist quinpirole (0.01–1 μmol/l) reduced the evoked overflow of tritium by maximally 56%, an effect antagonized by the D2-selective antagonist (−)-sulpiride (1 μmol/l). The D1-selective agonist 2,3,4,5-tetrahydro-7,8-dihydroxy-1-phenyl-1H-3-benzazepine (SKF 38393) caused a slight decrease only at the high concentration of 10 μmol/l. (−)-Sulpiride (0.1–10 μmol/l) moderately increased the evoked overflow of tritium when given alone. The dopamine uptake inhibitor nomifensine (10 μmol/l) caused a decrease, and in its presence the increase produced by (−)-sulpiride became much more marked, amounting to maximally 149%. (+)-Sulpiride (0.1–1 μmol/l) failed to change the evoked overflow of tritium in the presence of nomifensine. The dopamine-releasing agent (±)-amphetamine (1 μmol/l) also reduced the evoked overflow, an effect abolished by (−)-sulpiride. Finally, bretylium (1 mmol/l), which blocks the release of dopamine, increased the evoked overflow. (−)-Sulpiride (1 μmol/l) lost its facilitatory effect in slices treated with bretylium.
We conclude that the release of acetylcholine in rat nucleus accumbens, like its release in the nucleus caudatusputamen, is modulated through dopamine D2-receptors. The receptors are activated by endogenous dopamine under the conditions of these experiments.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Baud P, Arbilla S, Langer SZ (1985) Inhibition of the electrically evoked release of [3H]acetylcholine in rat striatal slices: an experimental model for drugs that enhance dopaminergic neurotransmission. J Neurochem 44:331–337
Björklund A, Lindvall O (1984) Dopamine-containing systems in the CNS. In: Björklund A, Hökfelt T (eds) Handbook of chemical neuroanatomy, vol 2. Elsevier, Amsterdam, pp 55–122
Cantrill R, Arbilla S, Zivkovic B, Langer SZ (1983) Amphetamine enhances latent dopaminergic neurotransmission in the rat striatum. Effects on 3H-acetylcholine release. Naunyn-Schmiedeberg's Arch Pharmacol 322:322–324
Consolo S, Ladinsky H, Bianchi S, Ghezzi D (1977) Apparent lack of a dopaminergic-cholinergic link in the rat nucleus accumbens septi-tuberculum olfactorium. Brain Res 135:255–263
Consolo S, Wu CF, Fusi R (1987) D-1 receptor-linked mechanism modulates cholinergic neurotransmission in rat striatum. J Pharmacol Exp Ther 242:300–305
Cubeddu LX, Hoffmann IS (1983) Frequency-dependent release of acetylcholine and dopamine from rabbit striatum: its modulation by dopaminergic receptors. J Neurochem 41:94–101
de Belleroche J, Gardiner IM (1983) Action of apomorphine, bromocriptine and lergotrile on γ-aminobutyric acid and acetylcholine release in nucleus accumbens and corpus striatum. J Neural Transm 58:153–168
de Belleroche JS, Neal MJ (1982) The contrasting effects of neuroleptics on transmitter release from the nucleus accumbens and corpus striatum. Neuropharmacology 21:529–537
Dubocovich ML, Weiner N (1981) Modulation of the stimulation-evoked release of [3H]dopamine in the rabbit retina. J Pharmacol Exp Ther 219:701–707
Gold R, Bluth R (1985) Dopaminergic modulation of acetylcholine release from slices of rat nucleus accumbens. Biogenic Amines 2:211–218
Heimreich I, Reimann W, Hertting G, Starke K (1982) Are presynaptic dopamine autoreceptors and postsynaptic dopamine receptors in the rabbit caudate nucleus pharmacologically different? Neuroscience 7:1559–1566
Hernandez L, Hoebel BG (1988) Food reward and cocaine increase extracellular dopamine in the nucleus accumbens as measured by microdialysis. Life Sci 42:1705–1712
Hertting G, Zumstein A, Jackisch R, Hoffmann I, Starke K (1980) Modulation by endogenous dopamine of the release of acetylcholine in the caudate nucleus of the rabbit. Naunyn-Schmiedeberg's Arch Pharmacol 315:111–117
Hoffmann IS, Cubeddu LX (1984) Differential effects of bromocriptine on dopamine and acetylcholine release modulatory receptors. J Neurochem 42:278–282
Joyce JN, Marshall JF (1987) Quantitative autoradiography of dopamine D2 sites in rat caudate-putamen: localization to intrinsic neurons and not to neocortical afferents. Neuroscience 20:773–795
Kelly E, Nahorski SR (1987) Dopamine D-2 receptors inhibit D-1 stimulated cyclic AMP accumulation in striatum but not limbic forebrain. Naunyn-Schmiedeberg's Arch Pharmacol 335:508–512
König JFR, Klippel RA (1963) The rat brain. A stereotaxic atlas of the forebrain and lower parts of the brain stem. Krieger, Huntington, New York
Lehmann J, Langer SZ (1983) The striatal cholinergic interneuron: synaptic target of dopaminergic terminals? Neuroscience 10:1105–1120
Mao CC, Cheney DL, Marco E, Revuelta A, Costa E (1977) Turnover times of gamma-aminobutyric acid and acetylcholine in nucleus caudatus, nucleus accumbens, globus pallidus and sub stantia nigra: effects of repeated administration of haloperidol. Brain Res 132:375–379
McGeer PL, Kimura H, McGeer EG, Peng JH (1982) Cholinergic systems in the CNS. In: Bradford HF (ed) Neurotransmitter interaction and compartmentation. Plenum, New York, pp 253–289
Setler PE, Sarau HM, Zirkle CL, Saunders HL (1978) The central effects of a novel dopamine agonist. Eur J Pharmacol 50:419–430
Stadler H, Gadea Ciria M, Bartholini G (1975) “In vivo” release of endogenous neurotransmitters in cat limbic regions: effect of chlorpromazine and of electrical stimulation. Naunyn-Schmiedeberg's Arch Pharmacol 288:1–6
Starke K (1979) Presynaptic regulation of release in the central nervous system. In: Paton DM (ed) The release of catecholamines from adrenergic neurons. Pergamon, Oxford, pp 143–183
Starke K, Montel H, Gayk W, Merker R (1974) Comparison of the effects of clonidine on pre- and postsynaptic adrenoceptors in the rabbit pulmonary artery. Naunyn-Schmiedeberg's Arch Pharmacol 285:133–150
Starke K, Späth L, Lang JD, Adelung C (1983) Further functional in vitro comparison of pre- and postsynaptic dopamine receptors in the rabbit caudate nucleus. Naunyn-Schmiedeberg's Arch Pharmacol 323:298–306
Stoof JC, Kebabian JW (1982) Independent in vitro regulation by the D-2 dopamine receptor of dopamine-stimulated efflux of cyclic AMP and K+-stimulated release of acetylcholine from rat neostriatum. Brain Res 250:263–270
Stoof JC, Verheijden PFHM (1986) D-2 receptor stimulation inhibits cyclic AMP formation brought about by D-1 receptor stimulation in rat neostriatum but not nucleus accumbens. Eur J Pharmacol 129:205–206
Stoof JC, Thieme RE, Vrijmoed-de Vries MC, Mulder AH (1979) In vitro acetylcholine release from rat caudate nucleus as a new model for testing drugs with dopamine-receptor activity. Naunyn-Schmiedeberg's Arch Pharmacol 309:119–124
Stoof JC, Verheijden PFHM, Leysen JE (1987) Stimulation of D2-receptors in rat nucleus accumbens slices inhibits dopamine and acetylcholine release but not cyclic AMP formation. Brain Res 423:364–368
Wichmann T, Illing RB, Starke K (1987) Evidence for a neurotransmitter function of acetylcholine in rabbit superior colliculus. Neuroscience 23:991–1000
Author information
Authors and Affiliations
Additional information
Send offprint requests to K. Starke at the above address
Rights and permissions
About this article
Cite this article
Wedzony, K., Limberger, N., Späth, L. et al. Acetylcholine release in rat nucleus accumbens is regulated through dopamine D2-receptors. Naunyn-Schmiedeberg's Arch Pharmacol 338, 250–255 (1988). https://doi.org/10.1007/BF00173396
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00173396