Abstract
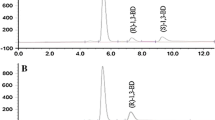
A number of bacteria and yeast was screened for asymmetric reduction of prochiral chloroacetone into chiral 1-chloro-2-propanol, which is chemically convertible into chiral 1,2-epoxypropane. In this way Rhodotorula glutinis produced optically pure S-1,2-epoxypropane with 98% enantiomeric excess and in a relatively high final concentration. The enzyme that catalysed the asymmetric reduction was an NAD(P)H-dependent alcohol dehydrogenase. Reduction of racemic 3-chloro-2-butanone resulted in mixtures of cis and trans-2,3-epoxybutane, indicating that no enantioselective reduction of this haloketone occurred.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adlercreutz P (1991) Novel biocatalyst for the asymmetric reduction of ketones: permeabilized cells of Gluconobacter oxydans. Enzyme Microb Technol 13:9–14
Carvalho M de, Okamoto MT, Moran PJS, Rodrigues AR (1991) Baker's yeast reduction of α-haloacetophenones. Tetrahedron 47:2073–2080
Fauve A, Veschambre H (1990) Regiospecificity and enantiospecificity in microbiological reduction of acylic β-diketones. Biocatalysis 3:95–109
Furuhashi K (1986) A fermentation process for the production of optically active epoxides. Chem Economy Eng Rev 18:21–26
Furuhashi K, Taoka A, Uchida S, Karube I, Suzuki S (1981) Production of 1,2-epoxyalkanes from 1-alkenes by Nocardia corallina B-276. Eur J Appl Microbiol Biotechnol 12:39–45
Habets-Crützen AQH, Bont JAM de (1985) Inactivation of alkene oxidation by epoxides in alkene- and alkene-grown bacteria. Appl Microbiol Biotechnol 22:428–433
Habes-Crützen AQH, Carlier SJN, Bont JAM de, Wistuba D, Schurig V, Hartmans S, Tramper J (1985) Stereospecific formation of 1,2-epoxypropane, 1,2-epoxybutane and 1-chloro-2,3-epoxypropane by alkene-utilizing bacteria. Enzyme Microbiol Technol 7:17–21
Hirschbein BL, Whitesides GM (1982) Laboratory-scale enzymatic/chemical synthesis of d- and l-β-chlorolactic acid and d-and l-potassium glycidate. J Am Chem Soc 104:4458–4460
Hou CT, Patel R, Laskin AI, Barnabe N, Barist I (1983) Epoxidation of short-chain alkenes by resting-cell suspensions of propane-grown bacteria. Appl Environ Microbiol 46:171–177
Imuta M, Kawai K, Ziffer H (1980) Product stereospecificity in the microbial reduction of α-haloaryl ketones. J Org Chem 45:3352–3355
Keinan E, Hafeli EK, Seth KK, Lamed R (1986) Thermostable enzymes in organic synthesis. Asymmetric reduction of ketones with alcohol dehydrogenase from Thermoanaerobium brockii. J Am Chem Soc 108:162–169
Maconi E, Aragozzini F (1989) Stereoselective reduction of noncyclic ketones with lactic acid bacteria. Appl Microbiol Biotechnol 31:29–31
Man JC de, Rogosa M, Sharpe ME (1960) A medium for the cultivation of lactobacilli. J Appl Bacteriol 23:130–135
Nakamura K, Kawai Y, Kitayama T, Miyai T, Ogawa M, Mikata Y, Higaki M, Ohno A (1989) Asymmetric reduction of ketones with microbes. Bull Inst Chem Res Kyoto Univ 67:157–168
Servi S (1990) Baker's yeast as a reagent in organic synthesis. Synthesis 1:1–25
Sih CJ, Chen CS (1984) Mikrobielle asymmetrische Katalyse. Enantioselektive Reduktion von Ketonen. Angew Chem 96:556–565
Schurig V, Bürkle W (1982) Extending the scope of enantiomer resolution by complexation gas chromatography. J Am Chem Soc 104:7573–7580
Smet de MJ, Kingma J, Wynberg H, Witholt B (1983) Pseudomonas oleovorans as a tool in bioconversions of hydrocarbons: growth, morphology and conversion characteristics in different two-phase systems. Enzyme Microb Technol 5:352–360
Subramanian V (1986) Oxidation of propene and 1-butene by Methylococcus capsulatus and Methylosinus trichosporium. J Ind Microbiol 1:119–127
Ward OP, Young CS (1990) Reductive biotransformations of organic compounds by cells or enzymes of yeast. Enzyme Microb Technol 12:483–493
Weijers CAGM, Bont JAM de (1991) Enantioselective degradation of 1,2-epoxyalkanes by Nocardia H8. Enzyme Microb Technol 13:306–308
Weijers CAGM, Ginkel CG van, Bont JAM de (1988a) Enantiomeric composition of lower epoxyalkanes produced by methane-, alkane- and alkene-utilizing bacteria. Enzyme Microb Technol 10:214–218
Weijers CAGM, Haan A de, Bont JAM de (1988b) Chiral resolution of 2,3-epoxyalkanes by Xanthobacter Py2. Appl Microbiol Biotechnol 27:337–340
Author information
Authors and Affiliations
Additional information
Correspondence to: C. A. G. M. Weijers
Rights and permissions
About this article
Cite this article
Weijers, C.A.G.M., Litjens, M.J.J. & de Bont, J.A.M. Synthesis of optically pure 1,2-epoxypropane by microbial asymmetric reduction of chloroacetone. Appl Microbiol Biotechnol 38, 297–300 (1992). https://doi.org/10.1007/BF00170075
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00170075