Summary
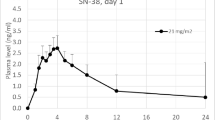
A Phase I study of caracemide evaluating a short intravenous infusion repeated every 21 days is presented. Patients were entered at 85 mg/m2 with subsequent escalation levels of 170, 425, 595, and 795 mg/m2. Mild to moderate nausea and vomiting occurred at all dose levels. An apparent allergic reaction was observed at the 425 mg/m2 level. A “burning pain” originating in the mucosal areas of the head and neck, progressing to the chest and abdomen, was noted at the 425 mg/m2 level. Because of this observation, the infusion time was extended to 4 h. At the 795 mg/m2, this toxicity precluded completion of the 4 h infusion. Pharmacokinetic evaluation disclosed blood levels of 0.74–2.31 μg/ml at the 425 mg/m2 during the 0.5 h infusion. At the same dose for a 4 h infusion time, blood levels were 0.15–0.18 μg/ml. At 595 mg/m2 administered as a 4 h infusion, blood levels increased to 0.33 ± 0.14 μg/ml. The drug was cleared rapidly from the blood compartment with a half-life of 2.5 min and a total body clearance of 11.5 1/min/m2. No partial or complete response was observed. However, an advanced colon carcinoma patient experienced subjective pain relief with a decrease in carcinoembryonic antigen. The dose-limiting toxicity of caracemide using the 4 h infusion was an intolerable “burning pain” with a maximum tolerated dose of 795 mg/m2. Further characterization of this dose-limiting toxicity is required prior to further clinical evaluation of caracemide.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Clinical Brochure. Caracemide (NSC-253272). Division of Cancer Treatment, National Cancer Institute, Bethesda, MD, 1983
Moore EC, Loo TL: Inhibition of ribonucleotide reductase by caracemide. Cancer Treat Rep 68:1293–1294, 1984
Carter SK: Study design principles for clinical evaluation of new drugs as developed by the chemotherapy program of the National Cancer Institute. In: M Staquet (ed) The Design of Clinical Trials in Cancer Therapy, Futura Publishing Co., Mount Kisco, NY, pp 242–289, 1972
Chabot GG, Pazdur R, Evans L, Marier S, Valeriote F, Baker LH: Caracemide stability, preclinical pharmacology in mice and preliminary pharmacology in humans (Abstr). Proc Am Soc Clin Oncol 26:354, 1985
Neuman RA, Farquahar D, Meyn R, Moore EC, Massia S, Korp JD, McKinney M: Biochemical pharmacology of N-acetyl-N-(methylcarbamoyloxy)-N′-methylurea (Caracemide). Biochem Pharmacol 35:2781–2787, 1986
Miller AB, Hoogstraten B, Staquet M, Winkler A: Reporting results of cancer treatment. Cancer 47:207–214, 1981
Gibaldi M, Perrier D: Pharmacokinetics, Marcel Dekker, Inc., New York, NY, pp 27–33, 1975
Raber MM, Legha S, Dimery I, Kavanaugh J, Adams F, Krakoff I: Phase I Study of Caracemide (Abstr). Proc Am Soc Clin Oncol 4:46, 1985
Investigational Drug Branch, Cancer Therapy Evaluation Program, National Cancer Institute. Minutes of the Phase I Working Group, March 20–21, 1985
Adams F, Kavanagh J, Raber M, Fernandez F, Richelson E: Neuropsychiatric manifestations of caracemide toxicity in a Phase I trial (Abstr). Proc Am Soc Clin Oncol 4:50, 1985
Lu K, Savaraj N, Feun LG, Neuman RA, Robbins VW, Gray KN, Loo TL: Pharmacokinetics of caracemide (NSC- 253272) in dogs (Abstr). Proc Am Soc Clin Oncol 26:359, 1985
Lin DP, Chabot GG, Valeriote FA, Baker LH: Identification of degradation products of caracemide, a new anticancer drug (Abstr). Annual Anachem Symposium, Amer Chem Soc, 1985
Memorandum of Phase I Investigators. Investigational Drug Branch, National Cancer Institute, Bethesda MD, December 24, 1985
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pazdur, R., Chabot, G.G. & Baker, L.H. Phase I study and pharmacokinetics of caracemide (NSC-253272) administered as a short infusion. Invest New Drugs 5, 365–371 (1987). https://doi.org/10.1007/BF00169976
Issue Date:
DOI: https://doi.org/10.1007/BF00169976