Abstract
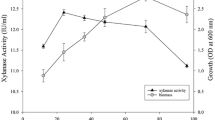
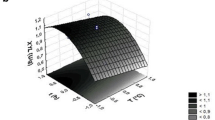
The thermophilic, xylanolytic, anaerobic organism, Dictyoglomus sp. B1, was cultivated in batch and continuous cultures in media containing insoluble beech-wood xylan. The extracellular xylanase activity levels obtained for the two cultivation methods were compared. Experiments were performed separately to determine the optimum substrate concentration, dilution rate, pH and temperature for xylanase production. Maximum xylanase activity was found at a substrate concentration of 1.5 g xylan/l, a dilution rate of 0.112 h−1, pH 8.0 and at 7°C. Different combinations of these optimum values were used in a 23 factorial experiment to investigate whether an increase in the xylanase production/activity could be achieved. A maximum xylanase activity of 2312 U/l was found when fermentors were operated at 73°C with a substrate concentration of 1.5 g xylan/l, pH 8.0, and a dilution rate of 0.112 h−1. Thus, the optimum xylanase activity in the factorial experiment was obtained when the conditions that gave the maximum xylanase activities in the individual experiments were combined. Optimum xylanase activity obtained in the 23 factorial experiment was 6.2 times higher than the activity found in the initial batch culture (373 U/l) and 3.0 times higher than the activity of a batch culture (783 U/l) grown at the same optimum conditions as the factorial experiment. The higher specific xylanase activity (217 U/mg protein) found in the 23 factorial experiment was 4.1 times higher than the specific activity in the initial batch culture (53 U/mg protein).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Angelidaki I, Petersen SP, Ahring BK (1990) Effects of lipids on thermophilic anaerobic digestion and reduction of lipid inhibition upon addition of bentonite. Appl Microbiol Biotechnol 33:467–472
Antranikian G, Zablowski P, Gottschalk G (1987) Conditions for the overproduction and excretion of thermostable α-amylase and pullulanase from Clostridium thermohydrosulfuricium DSM 567. Appl Microbiol Biotechnol 27:75–81
Bachmann SL, McCarthy AJ (1991) Purification and cooperative activity of enzymes constituting the xylan-degrading system of Thermomonospora fusca. Appl Environ Microbiol 57:2121–2130
Bajpai P, Bajpai PK (1992) Biobleaching of kraft pulp. Process Biochem 27:319–325
Bérenger JF, Frixon C, Bigliardi J, Creuzet N (1985) Production, purification and properties of thermostable xylanases of Clostridium stercorarium. Can J Microbiol 31:635–643
Bradford M (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
DSM—Deutsche Sammlung von Mikroorganismen und Zellkulturen GmbH (1989) Catalogue of strains 1989. German collection of Microorganisms and Cell Cultures, 4th edn
Edwards C (1990) Thermophiles. In: Edwards C (ed) Microbiology of extreme environments. Open University, Oxford, pp 1–32
Kantelinen A, Viikari A, Lindo M, Rättø M Rauna M, Sundquist J (1988) Hemicellulases and their potential role in bleaching. In: Proceedings of the 1988 International Pulp Bleaching Congress, Tappi. pp 1–9
Khasin A, Alchanati I, Shoham Y (1993) Purification and characterization of a thermostable xylanase from Bacillus stearothermophilus T-6. Appl Environ Microbiol 59:1725–1730
Linko M, Poutanen K, Viikari L (1989) New developments in the application of enzymes for biomass processing. In: Coughlan MP (ed) Enzyme systems for lignocellulose degradation. Elsevier, New York pp 331–346
Mathrani IM, Ahring BK (1991) Isolation and characterization of a strictly xylan-degrading Dictyoglomus from a man-made, thermophilic anaerobic environment. Arch Microbiol 157:13–17
Mathrani IM, Ahring BK (1992) Thermophilic and alkalophilic xylanases from several Dictyoglomus isolates. Appl Microbiol Biotechnol 38:23–27
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Nakanishi K, Marui M, Yasui T (1992) Comparison of xylan and methyl β-xyloside-induced xylanases from Streptomyces sp. J Ferment Bioeng 74:392–394
Priem B, Dobberstein J, Emeis CC (1991) Production of β-1,4-sugars in continuous culture by Aureobasidium pullulans CBS 58475. Biotechnol Lett 13:149–154
Rättö M, Mathrani IM, Ahring BK, Viikari L (1994) Application of thermostable xylanases of Dictyoglomus sp. in enzymatic treatment of kraft pulps. Appl Microbiol Biotechnol 41:130–133
Röthlisberger P, Fiechter A, Zimmermann W (1992) Production of thermostable xylanases in batch and continous culture by Thermomonospora fusca KW 3. Appl Microbiol Biotechnol 37:416–419
Simpson HD, Haufler UR, Daniel RM (1991) An extremely thermostable xylanase from the thermophilic eubacterium Thermotoga. Biochem J 277:413–417
Sørensen AH, Winther-Nielsen M, Ahring BK (1991) Kinetics of lactate, acetate, propionate in upadapted and lactate-adapted thermophilic, anaerobic sewage sludge: the influence of sludge adaption for start-up of thermophilic UASB-reactors. Appl Microbiol Biotechnol 34:823–827
Takahashi N, Koshijima T (1988) Molecular properties of lignin-carbohydrate complexes from beech (Fagus crenata) and pine (Pinus densiflora) woods. Wood Sci Technol 22:177–189
Tangnu SK, Blanch HW, Wilke CR (1981) Production of xylanase by Streptomyces xylophagus nov. sp. Acta Biotechnol 1:31–40
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Adamsen, A.K., Lindhagen, J. & Ahring, B.K. Optimization of extracellular xylanase production by Dictyoglomus sp. B1 in continuous culture. Appl Microbiol Biotechnol 44, 327–332 (1995). https://doi.org/10.1007/BF00169924
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00169924