Abstract
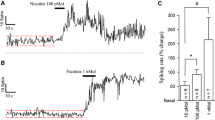
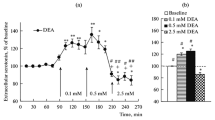
The influence of nitric oxide on acetylcholine release in the ventral striatum was investigated by the push-pull superfusion technique in the conscious, freely moving rat. Superfusion with the nitric oxide donors S-nitroso-N-acetylpenicillamine or with 3-morpholino-sydnonimine caused a pronounced increase in striatal acetylcholine release. This effect was prevented by superfusion with tetrodotoxin. Pre-superfusion with the guanylyl cyclase inhibitor methylene blue abolished the effect of 3-morpholino-sydnonimine. Superfusion of the ventral striatum with the guanylyl cyclase inhibitor LY83583 decreased acetylcholine release by 60% of basal release, whereas the less specific guanylyl cyclase inhibitor methylene blue was ineffective in this respect. Superfusion of the ventral striatum with inhibitors of nitric oxide synthase also led to different effects on basal acetylcholine release. Superfusion with L-NG-methylarginine did not influence basal acetylcholine release, whereas superfusion with L-NG-nitroarginine or with L-NG-nitroarginine methyl ester led to a substantial decrease in acetylcholine output, the latter compound being more effective. The effect of L-NG-nitroarginine was abolished by simultaneous superfusion with L-arginine.
The effects of NO donors and of LY83583 suggest that NO increases acetylcholine release, probably by a cGMP-dependent mechanism. The effectiveness of nitric oxide synthase inhibitors shows that the activity of striatal neurons is under the permanent influence of nitric oxide, that leads, via a direct or indirect mechanism, to continuous enhancement of acetylcholine release.
In conclusion, our findings suggest that NO synthesized in the ventral striatum acts as an intercellular messenger which modulates acetylcholine release.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Archer LS, Hampl V (1992) NG-monomethyl-L-arginine causes nitric oxide synthesis in isolated arterial rings: Trouble in paradise. Biochem Biophys Res Commun 188:590–596
Ariano MA (1984) Rat striatal cyclic nucleotide-reactive cells and acetylcholinesterase reactive interneurons are separate populations. Brain Res 296:160–163
Böhme GA, Bon C, Lemaire M, Reibaud M, Piot O, Stutzmann JM, Doble A, Blanchard JC (1993) Altered synaptic plasticity and memory formation in nitric oxide synthase inhibitor-treated rats. Proc Natl Acad Sci USA 90:9191–9194
Buxton ILO, Cheek DJ, Eckman D, Westfall PD, Sanders KM, Keef KD (1993) NG-nitro-L-arginine methyl ester and other alkyl esters of arginine are muscarinic receptor antagonists. Circ Res 72:387–395
Cespuglio R, Burlet S, Lambas-Senas L, Robert F, Jouvet M (1994) Voltammetric detection of NO throughout the sleep-wake cycle of the rat. Proceedings of the 6th International Conference on in vivo Methods, Seignosse ‘Les Tuquets’, France, pp 215–216
Damsma G, Lammerts van Bueren D, Westerink BHC, Horn AS (1987) Determination of acetylcholine and choline in the femtomol range by means of HPLC, a postcolumn enzyme reactor, and electrochemical detection. Chromatographia 24:827–831
Dwyer MA, Bredt DS, Snyder SH (1991) Nitric oxide synthase: Irreversible inhibition by L-NG-nitroarginine in brain in vitro and in vivo. Biochem Biophys Res Commun 176:1136–1141
Estall LB (1993) Inhibition of nitric oxide production selectively impairs learning and memory in the rat. Pharmacol Biochem Behav 49:959–962
Garthwaite J (1991) Glutamate, nitric oxide and cell-cell signalling in the nervous system. Trends Neurosci 14:60–67
Green LC, Wagner DA, Glogowski J, Skipper LP, Wishnok JS, Tannenbaum RS (1982) Analysis of nitrate, nitrite and [15N]-nitrate in biological fluids. Anal Biochem 126:131–138
Greenberg LH, Troyer E, Ferrendelli JA, Weiss B (1978) Enzymatic regulation of the concentration of cyclic GMP in mouse brain. Neuropharmacology 17:737–745
Guevara-Guzman R, Emson PC, Kendrick KM (1994) Modulation of in vivo striatal transmitter release by nitric oxide and cyclic GMP. J Neurochem 62:807–810
Haley JE, Wilcox GL, Chapman PF (1992) The role of nitric oxide in hippocampal long-term potentiation. Neuron 8:211–216
Heinzel B, John M, Klatt B, Bohme E, Mayer B (1992) Ca2+/calmodulin-dependent formation of hydrogen peroxide by brain nitric oxide synthase. Biochem J 281:627–630
Klatt P, Schmidt K, Brunner F, Mayer B (1994) Inhibitors of brain nitric oxide synthase: binding kinetics, metabolism, and enzyme inactivation. J Biol Chem 269:1674–1680
Klüver H, Barrera E (1953) A method for the combined staining of cells and fibers in the nervous system. J Neuropathol Exp Neurol 12:400–403
Lambert EL, Whitten JP, Baron BM, Cheng HC, Doherty NS, McDonald IA (1991) Nitric oxide synthesis in the CNS, endothelium and macrophages differs in its sensitivity to inhibition by arginine analogues. Life Sci 48:69–75
Malta E, Macdonald PS, Dusting GJ (1988) Inhibition of vascular smooth muscle relaxation by LY83583. Naunyn-Schmiedeberg's Arch Pharmacol 337:459–464
Matsuoka I, Sakurai K, Ono T, Nakanishi H (1987) Involvement of endogeneous noradrenaline release in methylene blue-induced contraction of isolated rabbit aorta. Jpn J Pharmacol 44:23–33
Mayer B, Schmid M, Klatt P, Schmidt K (1993) Reversible inactivation of endothelial nitric oxide synthase by NG-nitro-L-arginine. FEBS Lett 333:203–206
Meffert MK, Premack BA, Schulman H (1994) Nitric oxide stimulates Ca2+ independent synaptic vesicle release. Neuron 12:1235–1244
Moncada S, Palmer RMJ, Higgs EA (1991) Nitric oxide: Physiology, pathophysiology, and pharmacology. Pharmacol Rev 43:109–142
Okamura T, Yoshida K, Toda N (1990) Suppression by methylene blue of prostaglandin I2 synthesis in isolated dog renal arteries. J Pharmacol Exp Ther 254:198–203
Paxinos G, Watson C (1986) The rat brain in stereotaxic coordinates. Academic Press, Sydney
Phelps PE, Houser CR, Vaughn JE (1985) Immunocytochemical localization of choline acetyltransferase within the rat neostriatum: a correlated light and electron microscopic study of cholinergic neurons and synapses. J Comp Neurol 238:286–307
Prast H, Philippu A (1992a) Nitric oxide releases acetylcholine in the basal forebrain. Eur J Pharmacol 343:139–140
Prast H, Philippu A (1992b) Release of endogenous acetylcholine in the hypothalamus of conscious rats. Naunyn-Schmiedeberg's Arch Pharmacol 346:1–3
Prast H, Fischer H, Grass K, Philippu A (1994) Nitric oxide is a modulator of acetylcholine and glutamate release. Proceedings of the 6th International Conference on in vivo Methods, Seignosse ‘Les Tuquets’, France, pp 261–262
Schmidt MJ, Sawyer BD, Truex LL, Marshall WS, Fleisch JH (1985) LY83583: An agent that lowers intracellular levels of cyclic guanosine 3′, 5′-monophosphate. J Pharmacol Exp Ther 232:764–769
Shibuki K, Okada D (1991) Endogenous nitric oxide release required for long-term synaptic depression in the cerebellum. Nature 349:326–328
Southam E, Garthwaite J (1991) Comparative effects of some nitric oxide donors on cyclic cGMP levels in rat cerebellar slices. Neurosci Lett 130:107–111
Vincent SR, Kimura H (1992) Histochemical mapping of nitric oxide synthase in the rat brain. Neuroscience 46:755–784
Werner-Felmayer G, Prast H, Werner ER, Philippu A, Wachter H (1993) Inhibition of GTP cyclohydrolase I by bacterial lipopolysaccharide in the rat. FEBS Lett 322:223–226
Wood PL, Emmett MR, Tadimeti SR, Cler J, Mick S, Iyengar S (1990) Inhibition of nitric oxide synthase blocks N-methyl-D-aspartate-, quisqualate-, kainate-, harmaline-, and pentylenetetrazole-dependent increases in cerebellar cyclic GMP in vivo. J Neurochem 55:346–348
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Prast, H., Fischer, H., Werner, E. et al. Nitric oxide modulates the release of acetylcholine in the ventral striatum of the freely moving rat. Naunyn-Schmiedeberg's Arch Pharmacol 352, 67–73 (1995). https://doi.org/10.1007/BF00169191
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00169191