Abstract
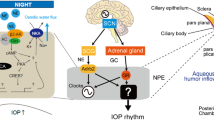
There has been a suspicion on the part of many clinicians and research scientists that intraocular pressure can be regulated by neural and/or humoral influences upon the rate of aqueous humor formation. It has been difficult, if not impossible, to separate specific influences of the central nervous system upon intraocular pressure from vascular induced or other secondary alterations. The past two decades have witnessed a great deal of study of the role of the adrenergic nervous system upon the regulation of intraocular pressure. From the investigations it is possible to formulate an integrated concept that can place years of work and speculation on a firm molecular foundation. The secretory tissue of the eye, the ciliary processes, contain an enzyme receptor complex, comprised by receptor complex, comprised by receptor bound membrane proteins, the catalytic moiety of the enzyme, a guanyl nucleotide regulatory protein (or N protein) and other features. The enzyme can be activated by well known neurohumoral or humoral agents that consist of catecholamines, glycoprotein hormones produced by the hypothalmic pituitary axis, and other related compounds, including placental gonadotropin. These compounds cause the ciliary epithelia to produce cyclic AMP at an accelerated rate. Cyclic AMP, as a second messenger, causes, either directly or indirectly, a decrease in the rate of aqueous humor formation that may be modulated by cofactors. Clinical syndromes fit the experimental data so that an integrated explanation can be given for the reduced intraocular pressure witnessed under certain central nervous system and adrenergic influences. The molecular biology of this concept provides important leads for future investigations that bear directly both upon the regulation of intraocular pressure and upon glaucoma.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Araie, M., M. Sawa, S. Nagataki & S. Mishima. Aqueous humor dynamics in man as studied by oral fluorescein. Jpn. J. Ophthalmol. 24: 346–362 (1980).
Becker, B. Vanadate and aqueous humor dynamics. Proctor Lecture Invest. Ophthalmol. Vis. Sic. 19: 1156–1165 (1980).
Becker, B. & J.S. Friedenwald. Clinical aqueous outflow. Arch. Ophthalmol. 50: 557–571 (1953).
Bietti, G.B., M.G. Bucci, J. Pecori-Giraldi et al. Etude sur l'action hypotonisante oculaire du Propranolol et du Propranolol-Pilocarpine par voie locale. Bull. Soc. Ophthalmol. Fr. 84: 509 (1971).
Brand, I. Intraokulare Hypotonie bie Myotonikern. Ophthalmologica 129; 81–88 (1955).
Bromberg, B.B., D.S. Gregory & M.L. Sears. Beta-adrenergic receptors in ciliary processes of the rabbit. Invest. Ophthalmol. Vis. Sci. 19: 203–207 (1980).
Coakes, R.L. & R.F. Brubaker. A method of measuring aqueous humor flow and corneal permeability using a fluorophotometry monogram. Invest. Ophthalmol. Vis. Sci. 18: 288–302 (1979).
De Grosz, E. La Tension intra-oculaire et la grossesse. Ann.d'Ocul. (Budapest) 167–177 (mars 1937).
Duke-Elder, W.S. The aetiology of simple glaucoma. Trans. Ophthalmol. Soc. U.K. 77: 205–228 (1957).
Eakins, K. The effect of intravitreous injections of norepinephrine, epinephrine and isoproterenol on the intraocular pressure and aqueous humor dynamics of rabbit eyes. J. Pharmacol. Exp. Ther. 140: 79–84 (1963).
Elwyn, H. Pathogenesis of chronic simple glaucoma. Arch. Ophthalmol. 19: 986–1008 (1938).
Finidori-Lepicard, J., S. Schorderet-Slatkine, J. Hanoune & E.E. Baulieu. Progesterone inhibits membrane-bound adenylate cyclase in Xenopis laevis oocytes. Nature 292: 255–256 (1981).
Gaasterland, D., C. Kupfer, K. Ross & H.L. Gabelnick. Studies of aqueous humor dynamics in man. 3. Measurements in young normal subjects using norepinephrine and isoproterenol. Invest. Ophthalmol. 12: 267–279 (1973).
Gierkowa, A., E. Samoehowiec & R. Halatek. Influence of gonadotropins and gonadotropic releasing hormones on the intraocular pressure. Klin. Oczna 46: 1005–1008 (1976).
Gregory, D.S., L.P. Bausher, B.B. Bromberg & M.L. Sears. The beta-adrenergic receptor and adenyl cyclase of rabbit ciliary processes. In: New Directions in Ophthalmic Research, ML. Sears, editor, Yale University Press, New Haven, Chapter 8, pp. 127–145 (1981).
Gregory, D.S., M.L. Sears, L. Bausher, H. Mishima & A. Mead. Intraocular pressure and aqueous flow are decreased by cholera toxin. Invest. Ophthalmol. Vis. Sci. 20: 371–381 (1981).
Hamburger, K. Experimentelle Glaukomtherapie. Klin. Monatsbl. Augenheilkunde 71: 810–811 (1923).
Hess, L. Pathogenesis of glaucoma. Arch. Ophthalmol. 33: 392–396 (1946).
Horven, L. & H. Gjonnaess. Corneal indentation pulse and intraocular pressure in pregnancy. Arch. Ophthalmol. 91: 92–98 (1974).
Imre, J. Pregnancy and the eye, their endocrinological relations. XV Concilium Ophthalmol. Egypte, Vol. III, 213–226 (1937).
Jones, R.F. & D.M. Maurice. New methods of measuring the rate of aqueous flow in man with fluorescein. Exp. Eye Res. 5: 208–220 (1966).
Krupin, T., S.M. Podos & B. Becker. The effect of optic nerve transection on osmotic alterations of intraocular pressure. Am. J. Ophthalmol. 70: 214–220 (1970).
Langham, M.E. & C.B. Taylor. The influence of pre- and postganglionic section of the cervical sympathetic on the intraocular pressure of rabbits and cats. J. Physiol. 152: 437–446 (1960).
Liu, H.K. & G.C.Y. Chiou. Continuous, simultaneous, and instant display of aqueous humor dynamics with a microspectrophotometer and a sensitive drop counter. Exp. Eye Res. 32: 583–592 (1981).
Magitot, A. Sur l'origine intra-cranienne de l'atrophie optique glaucomateurse. Ann. Ocul. (Paris) 180: 321 (1947).
Meyer, E.J., C.R. Roberts, H.M. Leibowitz, B. McGowan & R.E. Houle. Influence of norethynodrel with mestranol on intraocular pressure in glaucoma. II. A controlled double-blind study. Arch. Ophthalmol. 75: 771–773 (1966).
Mishima, H., M. Sears, L. Bausher, & D. Gregory. Ultracytochemistry of cholera toxin binding sites in ciliary processes. Cell Tissue Res. 223: 241–253 (1982).
Nagai, M., T. Ban & T. Koratsu. Studies on the changes of the intraocular pressure induced by electrical stimulation of the hypothalamus. Med. j. Osaka Univ. 2: 87–95 (1951).
Nathanson, J.A. Human ciliary process adrenergic receptor: pharmacological characterization. Invest. Ophthalmol. Vis. Sci. 21: 798–804 (1981).
Neufeld, A.H., L.M. Jampol, & M.L. Sears. Cyclic-AMP in the aqueous humor: the effects of adrenergic agents. Exp. Eye Res. 14: 242–250 (1972).
Neufeld, A.H. & M.L. Sears. Cyclic-AMP in the ocular tissues of the rabbit, monkey and human. Invest. Ophthalmol. 13: 475–477 (1974).
Neufeld, A.H., D.K. Dueker, T. Vegge & M.L. Sears. Adenosine 3′,5′-monophosphate increases the outflow of aqueous humor from the rabbit eye. Invest. Ophthalmol. 14: 40–42 (1975).
Neufeld, A.H. & M.L. Sears. Adenosine 3′,5′-monophosphate analogue increases the outflow facility of the primate eye. Invest. Ophthalmol. 14: 688–689 (1975).
Neufeld, A.H. Epinephrine and timolol: How do these drugs lower intraocular pressure? Ann. Ophthalmol. 13: 1109–1111 (1981).
Niebroj, T., A. Gierek, & E. Samochowiec: Urinary excretion of luteinizing hormone in women with glaucoma. Pol. Med. J. 9: 526–528 (1970).
Niebroj, T, A. Gierek, & E. Samochowiec. The influence of chorionic gonadotropin (HCG) on water metabolism in the eye of patients suffering from glaucoma. Pol. Endocrinol. XXII, No. 3: 204–207 (1971).
Paterson, C.A. The effect of sympathetic nerve stimulation on the aqueous humor dynamics of the cocaine pretreated animal. Exp. Eye Res. 5: 37–44 (1966).
Phillips, C.I., G. Howitt, & J. Rowlands. Propranolol as ocular hypotensive agent. Br. J. Ophthalmol. 51: 222–226 (1967).
Radnot, M. Beitrag zur Entstehung der durch Kastration erzeugten Hypotonie. Klin. Monatsbl. Augenheilkunde 115: 524–526 (1949).
Riise, D. & S.E. Simonsen. Intraocular pressure in unilateral optic nerve lesion. Acta Ophthalmol. 47: 750–755 (1969).
Ross, R.A. & S.M. Drance. Effects of topically applied isoproterenol on aqueous dynamics in man. Arch. Ophthalmol. 83: 39–46 (1970).
Salomon, Y. Receptors for the glycoprotein hormones: LH, FSH, hCG and TSH. In: Cellular Receptors for Hormones and Neurotransmitters, D. Schulster and A. Levitzki, editors, Chichester, John Wiley & Son, Chapter 8, pp. 149–161 (1980).
Schmerl, E. & B. Steinberg. Separation of diencephalic centers concerned with pupillary motility and ocular tension. Am. J. Ophthalmol. 33: 1379–1381 (1950).
Sears, M.L. & E.H. Bárány. Effects of cervical sympathectomy with adrenergic inhibitors. Arch. Ophthalmol. 64: 839–848 (1960).
Sears, M.L. Friedenwald Lecture. Perspectives in glaucoma research. Invest. Ophthalmol. Vis. Sci. 17: 6–22 (1978).
Sears, M.L. The aqueous. In: Adler's Physiology of the Eye, 7th edition, R.A. Moses, editor. St. Louis, C.V. Mosby, Chapter 7, pp. 204–226 (1980).
Sears, M.L., D. Gregory, L. Bausher, H. Mishima & J. Stjernschantz. A receptor for aqueous humor formation. In: New Directions in Ophthalmic Research, M.L. Sears, editor, New Haven, Yale University Press, chapter 10, pp. 163–183 (1981).
Treister, G. & S. Mannor. Intraocular pressure and outflow facility. Arch. Ophthalmol. 83: 311–318 (1970).
Trope, G.E., B. Clark & S.J.S. Titinchi. Identification of beta adrenergic receptors in the pigmented mammalian irisciliary body diaphragm. Exp. Eye Res. 34: 153–157 (1982).
Vareilles, P., DL Silverstone, B. Plazonnet, J.-C. LeDouarec, M.L. Sears, & C.A. Stone. Comparison of the effects of Timolol and other adrenergic agents on intraocular pressure in the rabbit. Invest. Ophthalmol. Vis. Sci. 16: 987–996 (1977).
von Hippel, A. & A. Grunhagen. Uber der Einfluss der Nerven auf die Hohe des intraokularen Druckes. Alb. von Graefes Arch. Klin. Exp. Ophthalmol. 15: 27 (1870).
von Sallman, L. & O. Lowenstein. Responses of intraocular pressure, blood pressure and cutaneous vessels to electric stimulation in the diencephalon. Am. J. Ophthalmol. 39: 11–29 (1955).
Waitzman, M.B. & W.D. Woods. Some characterizations of an adenyl cyclase preparation from rabbit ciliary process tissue. Exp. Eye Res. 12: 99–111 (1971).
Weekers, R., Y. Delmarcelle & J. Gustin. Treatment of ocular hypotension by adrenalin and diverse sympathomimetic amines. Am. J. Ophthalmol. 40: 666–672 (1955).
Wentworth, W.O. & R.F. Brubaker. Aqueous humor dynamics in a series of patients with third neuron Horner's syndrome. Amer. J. Ophthalmol. 92: 407–415 (1981).
Wilke, K. Episcleral venous pressure and pregnancy. Acta Ophthalmol. Suppl. 125: 40–41 (1975).
Yablonski, M.E., T.J. Zimmerman, S.R. Waltman, & B. Becker. A fluorophotometric study of the effect of topical timolol on aqueous humor dynamics. Exp. Eye Res. 27: 134–142 (1978).
Zimmerman, T.J. & H.E. Kaufman. Timolol. A beta-adrenergic blocking agent for the treatment of glaucoma. Arch. Ophthalmol. 95: 601–604 (1977).
Author information
Authors and Affiliations
Additional information
This work was supported in part by grants from the Connecticut Lions Eye Research Foundation, Inc., Foresight, Inc., New Haven Foundation, and Research to Prevent Blindness, Inc.
Rights and permissions
About this article
Cite this article
Sears, M., Mead, A. A major pathway for the regulation of intraocular pressure. Int Ophthalmol 6, 201–212 (1983). https://doi.org/10.1007/BF00141129
Issue Date:
DOI: https://doi.org/10.1007/BF00141129