Summary
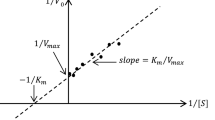
The investigated biocatalyst consists of gelatin-entrapped cells of Saccharomyces cerevisiae retaining invertase activity. Comparative examination of pH profile, apparent Km, saturation velocity and activation energy indicates that the entrapment procedure did not influence invertase affinity with sucrose but lead to some loss of activity probably due to either enzyme inactivation or cell-wall impairment as well as to substrate diffusion limitation in the gel matrix.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Cantarella M., Gianfreda L., Palescandolo R. & Scardi V. (1977) Biochem.J., 167, 313–315.
Filippusson H. & Hornby W.E. (1970). Biochem.J., 120, 215–219.
Gianfreda L., Parascandola P. & Scardi V. (1980). European J. Appl.Microbiol.Biotechnol., 11, 6–7.
Nelson N. (1944). J.Biol.Chem., 153, 375–380.
Usami S., Noda J. & Goto K. (1971). J.Ferment.Technol., 49, 598–603.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Parascandola, P., Scardi, V. Gelatin-entrapped whole-cell invertase. Biotechnol Lett 3, 369–374 (1981). https://doi.org/10.1007/BF00127620
Issue Date:
DOI: https://doi.org/10.1007/BF00127620