Abstract
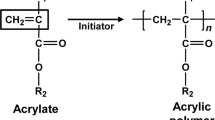
Polyacrylate (PA), which is widely used in disposable diapers, is synthesized by polymerization and cross-linking of acrylate. During the synthesis, 3–6% of the polyacrylate polymers is not incorporated into the absorbent material, but remains soluble. If the soluble PA is mobilized from a landfill, it could enter the groundwater. Therefore, the biodegradation and adsorption properties of soluble polymers contained in PA are determined in this study. The soluble PA is highly polydisperse, and the fraction tested has a weight-average molecular-weight of 16,700 and a range extending from 103 to 105. Sand-column tracer tests show that about 1% of the polyacrylate is unadsorbed, but the remainder has a retardation factor that averages at least 58. Biodegradation kinetics are determined in completely mixed biofilm reactors having a methanogenic consortium grown on glucose. The polyacrylate fraction, as well as glucose and acrylate, are removed and mineralized to CO2. The Monod parameters for the polyacrylate are: maximum specific rate of substrate utilization = 0.0016 gC/g biomass-day, and half-maximum-rate concentration = 0.79 gC/m3. Although these kinetics are much slower than for glucose and acrylate, significant degradation and mineralization are observed.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
American Society of Agronomy and Soil Science Society of American (1965). Methods of Solid Analysis, Madison, Wisconsin
Curtis GP, Roberts P & Reinhard M (1984). Sorption of organic solutes: Comparison between laboratory estimated retardation factors and field observations. Proc. 2nd Intl. Conf. on Groundwater Quality Research, March 1984, Tulsa, OK, pp. 35–38
Gottschalk G (1986). Bacterial Metabolism, 2nd ed., Springer-Verlag, New York
Hassett JJ, Means JC, Banwart WL, Woods SG (1980). Sorption Properties of Sediments and Energy-Related Pollutants. U.S. Environmental Protection Agency, Doc. No. EPA/600/3–80/041, p. 60, Washington DC
Kawai F, Fukaya M, Tani Y, & Ogata K (1977). Ferment. Technol. 55: 429
Lehrburger C (1988). Diapers in the Waste Stream: A Review of Waste Management and Public Policy Issues. National Assoc. Diaper Services, Sheffield, MA
Levenspiel O (1972). Chemical Reaction Engineering, John Wiley & Sons, Inc., New York
Martin JE, Howard KWF, & King LW (1987). Environmental behavior of 14C-tagged polyacrylate polymer: Column studies of flow and retardation in sand, Nuclear and Chem. Waste Management 7: 265–271
Merck & Co., Inc. (1989) The Merck Index, eleventh edition, Rahway, New Jersey
Miller ML (1968) The Structure of Polymers, Reinhold Publ. Corp., New York
Morita M & Watanabe Y (1977) Agric. Biol. Chem. 41: 1535
Perry RH, Green DW, & Maloney JO (1984) Perry's Chemical Engineers' Handbook, sixth ed., McGraw-Hill Book Co., New York
Rittmann BE, Crawford L, Tuck CK, & Namkung E (1986) In situ determination of kinetic parameters for biofilms. Isolation and characterization of oligotrophic biofilms. Biotechnol. Bioengr. 28: 1753–1760
Rittmann BE, Henry B, Odencrantz JE, & Sutfin JA (1992) Biological fate of a polydisperse acrylate polymer in anaerobic, sand-medium transport. Biodegradation (this issue)
Williamson K & McCarty PL (1976) Verification studies of the biofilm model for bacterial substrate utilization, J. Water Pollution Control Federation 48: 281–296
Zaikov GE & Livshits VS (1987) Biodegradable polymers. In: Pethrick RA (Ed.). Polymer Yearbook 4 (pp. 177–201). Harwood Academic Publishers, New York
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rittmann, B.E., Sutfin, J.A. & Henry, B. Biodegradation and sorption properties of polydisperse acrylate polymers. Biodegradation 2, 181–191 (1991). https://doi.org/10.1007/BF00124492
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00124492