Abstract
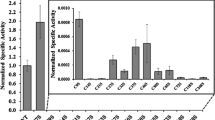
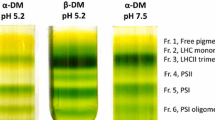
Violaxanthin deepoxidase (VDE) has been purified from spinach (Spinacia oleracea) leaves. The purification included differential sonication of thylakoid membranes, differential (NH4)2SO4 fractionation, gel filtration chromatography and finally either hydrophobic interaction chromatography or anion exchange chromatography. A total purification of more than 5000-fold compared to the original thylakoids enabled the identification of a 43 kDa protein as the VDE, in contrast to earlier reported molecular weight of 54–60 kDa. A detailed comparison was made for the VDE activity and polypeptide pattern for the different fractions throughout the purification and the best correlation was always found for the 43 kDa protein. The highest specific activity obtained was 256 μmol g−1 s−1 protein, which is at least 10-fold higher than reported earlier. We estimate that there is 1 VDE molecule per 20–100 electron transport chains. The 43 kDa protein was N-terminally sequenced, after protection of cysteine residues with β-mercaptoethanol and iodoacetamid, and a unique sequence of 20 amino acids was obtained. The amino acid composition of the protein revealed a high abundance of charged and polar amino acids and remarkably, 11 cysteine residues. Two other proteins (39.5 kDa and 40 kDa) copurifying with VDE were also N-terminally sequenced. The N-terminal part of the 39.5 kDa protein showed complete sequence identity both with the N-terminal part of cyt b 6 and an internal sequence of polyphenol oxidase.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- DMSO:
-
dimethylsulfoxid
- HIC:
-
hydrophobic interaction chromatography
- MGDG:
-
monogalactosyl diacylglycerol
- VDE:
-
violaxanthin deepoxidase
References
Arvidsson, P-O, Bratt, CE, Andréasson, L-E and Åkerlund, H-E (1993) The 28 kDa apoprotein of CP 26 in PS II binds copper. Photosynth Res 37: 217–225
Björkman, O and Demmig-Adams, B (1994) Regulation of photosynthethic light energy capture, conversion, and dissipation in leaves of higher plants. In: Schulze, A-D and Caldwell, MM (eds) Ecophysiology of Photosynthesis, pp 17–47. Springer-Verlag, Berlin
Bratt, CE, Arvidsson, P-O, Carlsson, M and Åkerlund, H-E (1995) Regulation of violaxanthin de-epoxidase activity by pH and ascorbate concentration. Photosynth Res 45: 169–175
Capaldi, RA and Vanderkooi, G (1972) The low polarity of many membrane proteins (solube proteins/polar amino acids / hydrophobicity / polarity index). Proc Natl Acad Sci USA 69: 930–932
Carlsson, M, Bratt, CE, Arvidsson, P-O and Åkerlund, H-E (1995) Regulation of violaxanthin de-epoxidase activity by pH and ascorbate concentration. In: Mathis, P (ed) Photosynthesis: From Light to Biosphere, Vol. IV, pp 55–58. Kluwer Academic Publishers, Dordrecht
Gruszecki, WI (1995) Different aspects of protective activity of the xanthophyll cycle under stress conditions. Acta Physiologiae Plantarum 17: 145–152
Hager, A (1975) Die reversiblen, lichtabhängigen Xanthophyllumwandlungen im Chloroplasten. Ber Dtsch Bot Ges 88: 27–44
Hager, A and Holocher, K (1994) Localization of the xanthophyllcycle enzyme violaxanthin de-epoxidase within the thylakoid lumen and abolition of its mobility by a (light-dependent) pH decrease. Planta 192: 581–589
Hager, A and Perz, H (1970) Veränderung der Lichtabsorption eines Carotinoids im Enzym (De-epoxidase)-Substrat (Violaxanthin)-Komplex. Planta (Berl.) 93: 314–322.
Laemmli, UK (1970) Cleavage of structual proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685
Ljungberg, U, Åkerlund, H-E and Andersson, B (1986) Isolation and characterisation of the 10-kDa and 22-kDa polypeptides of higher plant Photosystem II. Eur J Biochem 158: 477–462
Thayer, SS and Björkman, O (1990) Leaf xanthophyll content and composition in sun and shade determined by HPLC. Photosynth Res 23: 331–343
Yamamoto, HY and Kamite, L (1972) The effects of dithiothreitol on violaxanthin de-epoxidation and absorbance changes in the 500-nm region. Biochim Biophys Acta 267: 558–543
Yamamoto, HY and Higashi, RM (1978) Violaxanthin de-epoxidase; lipid composition and substrate specificity. Arch Biochem Biophys 190: 514–522
Author information
Authors and Affiliations
Additional information
A preliminary report of these results was presented at the Xth Int. Congress on Photosynthesis, Montpellier, France, 1995.
Rights and permissions
About this article
Cite this article
Arvidsson, PO., Bratt, C.E., Carlsson, M. et al. Purification and identification of the violaxanthin deepoxidase as a 43 kDa protein. Photosynth Res 49, 119–129 (1996). https://doi.org/10.1007/BF00117662
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00117662