Abstract
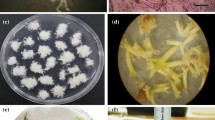
Walnut somatic embryos were multiplied by repetitive embryogenesis on a solid basal DKW medium at 25°C in the dark. When the embryos were isolated at early cotyledonary stage (1–2 mm long) from the primary embryos and cultured on the medium for 3 weeks, they developed into mature embryos showing white, enlarged cotyledons and shoot and root apex. After transfer to light on solid germination medium, however, few mature embryos (0–5%) germinated. Germination percentage increased to about 10% when the mature embryos were pretreated by a storage at 4°C in the dark for 2 months, or by desiccation at 25°C in the dark for 3 or 5 days under an air-humidity conditioned by saturated salt solutions (Mg(NO3)2.6H2O, or ZnSO4.7H2O). Similar results were obtained by the addition of gibberellic acid (GA3) to the germination medium. When mature embryos were desiccated and then placed on medical cotton compresses in liquid germination medium, 45% of the embryos germinated into complete plantlets. These plantlets continued their growth after transplanting to a mixture of peat and vermiculite in pots.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- GA3 :
-
gibberellic acid
- DKW medium:
-
Driver & Kuniyuki Walnut medium
References
Ammirato PV (1989) Recent progress in somatic embryogenesis. Intl. Assoc. Plant Tissue Culture Newsl. 57: 2–16
Buchheim J, Colburn SM & Ranch JP (1989) Maturation of soybean somatic embryos and the transition to plantlet growth. Plant Physiol. 89: 766–775
Cornu D (1988) Somatic embryogenesis in tissue culture of walnut (Juglans nigra, J. major and hybrids J. nigra x J regia). In: Ahuja MR (Ed) Somatic Cell Genetics of Woody Plants (pp 45–49). Kluwer Academic Publishers, Boston
Cornu D (1989) Walnut somatic embryogenesis, physiological and histological aspects. Ann. Sci. For. 46S: 133–135
Driver JA & Kuniyuki AH (1984) In vitro propagation of Paradox walnut rootstock. HortScience 19: 507–509
Ghazi TD, Cheema HV & Nabors MW (1986) Somatic embryogenesis and plant regeneration from embryogenic callus of soybean, Glycine max L. Plant Cell Rep. 5: 452–456
Gingas VM & Lineberger RD (1989) Asexual embryogenesis and plant regeneration in Quercus. Plant Cell Tiss. Org. Cult. 17: 191–203
Gray DG (1987) Quiescence in monocotyledonous and dicotyledonous somatic embryos induced by dehydration. HortScience 22: 810–814
Kermode A, Bewley JD, Dasgupta J & Misra S (1986) The transition from seed development to germination: a key role for desiccation? HortScience 21: 1113–1118
Lee BC, Shim SY & Lee SK (1988) Mass propagation and germination of somatic embryos in Juglans regia L. (English walnut). Res. Rep. Inst. For. Genet Korea 24: 99–106
Le Page-Degivry MT, Barthe P & Garello G (1990) Involvement of endogenous abscisic acid in onset and release of Helianthus annuus embryo dormancy. Plant Physiol. 92: 1164–1168
Marx S, Grosse W & Schneider-Poetsch HA (1988) A combined HPLC-ELISA assay applied to measure abscisic acid (ABA) and Abscisyl-β-D-Glucopyranoside in ripening embryos of walnut (Juglans regia L.). J. Plant Physiol. 133: 475–479
Merck Index (1983) Constant humidity solutions. Tenth edition, (pp misc-103). Merck & CO. Inc. Publ, Rahway, NJ, USA
Oishi MY & Bewley JD (1990) Distinction between the responses of developing maize kernels to fluridone and desiccation in relation to germinability, α-amylase activity, and abscisic acid content. Plant Physiol. 94: 592–598
Pearce D, Pharis RP, Rajasekaran K & Mullins MG (1987) Effects of chilling and ABA on (3H) Gibberellin A4 metabolism in somatic embryos of grape (Vitis vinifera L. x V. rupestris Scheele). Plant Physiol. 80: 381–385
Preece JE, Zhao J & Kung FH (1989) Callus production and somatic embryogenesis from white ash. HortScience 24: 377–380
Radojevic L (1988) Plant regeneration of Aesculus hippocastanum L. (Horse chestnut) through somatic embryogenesis. J. Plant Physiol. 132: 322–326
Rajasekaran K, Vine J & Mullins MG (1982) Dormancy in somatic embryos and seeds of Vitis: changes in endogenous abscisic acid during embryogeny and germination. Planta 154: 139–144
Rodriguez R, Revilla A, Albuerne M & Perez C (1989) Walnut (Juglans spp.). In: Bajaj YPS (Ed) Biotechnology in Agriculture and Forestry, Vol 5 (pp 99–126). Springer-Verlag, Berlin
Tulecke W (1987) Somatic embryogenesis in woody perennials. In: Bonga JM & Durzan DJ (Eds) Cell and Tissue Culture in Forestry, Vol 2 (pp 61–91). Martinus Nijhoff Publishers, Boston
Tulecke W & McGranahan GH (1985) Somatic embryogenesis and plant regeneration from cotyledons of walnut, Juglans regia L. Plant Sci. 40: 57–63
Tulecke W, McGranahan G & Ahmadi H (1988) Regeneration by somatic embryogenesis of triploid plants from endosperm of walnut, Juglans regia L. cv Mangerian. Plant Cell Rep. 7: 301–304
Wetzstein HY, Ault JR & Merkle SA (1989) Further characterization of somatic embryogenesis and plantlet regeneration in pecan (Carya illinoensis). Plant Sci. 64: 193–201
Wood BW (1982) Gibberellin-like substances in developing fruits of pecan. HortScience 17: 70–71
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Deng, MD., Cornu, D. Maturation and germination of walnut somatic embryos. Plant Cell Tiss Organ Cult 28, 195–202 (1992). https://doi.org/10.1007/BF00055517
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00055517