Abstract
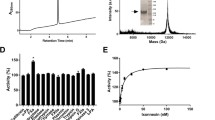
A low molecular mass anticoagulant (17 kDa) was isolated from the salivary glands of prefed female Hyalomma truncatum ticks by means of reverse phase and anion-exchange HPLC. Trypsin digestion and amino acid analysis confirmed the protein nature of the anticoagulant. The inhibitor appears to be uncompetitive with a Ki of 6.9×10−10M. The target of the anticoagulant is factor Xa at the junction of the extrinsic and intrinsic pathways. This may be crucial for the survival of the tick, making it feasible to investigate the possibility of vaccination with this antihaemostatic against tick feeding. In addition, tick anticoagulants may possibly have therapeutic application in controlling thrombosis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bradford, M.M., 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein, utilizing the principle of protein-dye binding. Anal. Biochem. 72: 248–254.
Cornish-Bowden, A., 1986. Why is uncompetitive inhibition so rare? A possible explanation with implications for the design of drugs and pesticides. FEBS., 203: 3–6.
Gordon, J.R. and Allen, J.R., 1991. Factors V and VII anticoagulant activities in the salivary glands of feeding Dermacentor andersoni ticks. J. Parasitol., 77: 167–170.
Jordan, S.P., Waxman, L., Smith, D.E. and Vlasuk, G.P., 1990. Tick anticoagulant peptide: kinetic analysis of the recombinant inhibitor with blood coagulation factor Xa. Biochemistry, 29, 11095–11100.
Limo, M.K., Voigt, W.P., Tumbo-Oeri, A.G., Njogi, R.M. and Ole-MoiYoi, O.K., 1991. Purification and characterization of an anticoagulant from the salivary glands of the ixodid tick, Rhipicephalus appendiculatus. Exp. Parasitol. 72: 418–429.
Margolis, J., 1958. Activation of plasma by contact with glass: evidence for a common reaction which releases plasma kinin and initiates coagulation. J. Physiol., 144: 1–22.
Mathews, C.K. and van Holde, K.E., 1990. The Regulation of Enzyme Activity In: Biochemistry, pp. 397–399. Benjamin/Cummings Publishing Company Inc., New York.
Meyer, B.H., Luus, H.G., Muller, F.O., Badenhorst, P.N., Lotter, M.G., Röthig, H.J., Grötsch, H. and Rozenkranz, B., 1992. Recombinant hirudin, a new anticoagulant, has no effect on feacal blood loss. Br. J. Clin. Pharmac., 33: 524–526.
Neeper, M.P., Waxman, L., Smith, D.E., Schulman, C.A., Sardona, M., Ellis, R.W., Schaffer, L.W., Siegl, P.K.S. and Vlasuk, G.P., 1990. Characterization of recombinant tick anticoagulant peptide: a highly selective inhibitor of blood coagulation factor Xa. J. Biol. Chem., 265: 17746–17752.
Segal, I.H., 1976. Enzymes In: Biochemical Calculations, p. 216. John Wiley & Sons, New York.
Vlasuk, G.P., Ramjit, D., Fujita, T., Dunwiddie, C.T., Nutt, E.M., Smith, D.E. and Shebuski, R.J., 1991. Comparison of the in vivo anticoagulant properties of standard heparin and the highly selective factor Xa inhibitors Antistasin and tick anticoagulant peptide (TAP) in a rabbit model of venous thrombosis. Thromb. Haemost., 65: 257–262.
Voet, D. and Voet, J.G., 1990. Molecular Physiology In: Biochemistry, p. 1093. John Wiley & Sons, New York.
Waxman, L., Smith, D.E., Arcuri, K.E. and Vlasuk, G.P., 1990. Tick anticoagulant peptide is a novel inhibitor of blood coagulation factor Xa. Science, 248: 593–596.
Waxman, L. and Connolly, T.M., 1993. Isolation of an inhibitor selective for collagen-stimulated platelet aggregation from the soft tick, Ornithodoros moubata. J. Biol. Chem., 268: 5445–5449.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Joubert, A.M., Crause, J.C., Gaspar, A.R.M.D. et al. Isolation and characterization of an anticoagulant present in the salivary glands of the bont-legged tick, Hyalomma truncatum . Exp Appl Acarol 19, 79–92 (1995). https://doi.org/10.1007/BF00052548
Issue Date:
DOI: https://doi.org/10.1007/BF00052548