Abstract
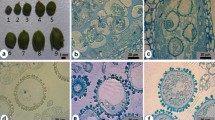
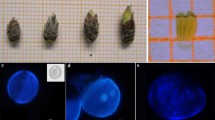
Three methods of microspore culture were tested for the induction of microspore embryogenesis in Camellia japonica L. cv. Elegans. Culture was performed on 17 different media consisting of Murashige and Skoog (MS) and N6 basal media with different combinations of carbon, growth regulators, serine and glutamine. Microspore suspensions plated over solid MS medium containing 4.5 μM 2,4-dichlorophenoxyacetic acid and 0.5 μM kinetin, with sucrose (MS6) or glucose (MS9) were seen as the best culture conditions for induction of embryogenesis. The development of microspore derived proembryos was obtained in MS medium supplemented with 2.2 μM N6-benzyladenine (MS10) and reached the highest level when the microspores were cultured in MS6 inducing medium. The development of microspore-derived embryos ceased at the maturation stage.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BA:
-
N6-benzyladenine
- 2,4-d :
-
2,4-dichlorophenoxyacetic acid
References
Bajaj, YPS (1990) Haploids in Crop Improvement I. Biotechnology in Agriculture and Forestry, Vol 12 (pp 101–541). Springer-Verlag, Berlin, Heidelberg
Chen, Z (1990) Rubber (Hevea brasiliensis Muell. Arg.): In vitro production of haploids. In: Baja, YPS (Ed) Biotechnology in Agriculture and Forestry, Vol 12 (pp 215–236). Springer-Verlag, Berlin, Heidelberg
Chen, Z, Chen, F, Chien, C, Wang, C-h, Chang, S-c, Hsu, H-c, Ou, H-h, Ho, Y-t & Lu, T-m (1978) A process of obtaining pollen plants of Hevea brasiliensis Muell.-Arg. Sci. Sin. 22: 81–90
Chu, CC (1978) The N6 medium and its applications to anther culture of cereal crops. Proc. Symp. Plant Tissue Culture Beijing (pp 43–50). Science Press, Peking
Lianfang, F (1990) Litchi (Litchi chinensis Sonn.): In vitro production of haploids. In: Bajaj, YPS (Ed) Biotechnology in Agriculture and Forestry, Vol 12 (pp 265–274). Springer-Verlag, Berlin, Heidelberg
McClintock, B (1929) A method for making aceto-carmin smears permanent. Stain Technol. 4: 53–56
Milewska-Pawliczuk, E (1990) Apple (Malus domestica Borkh.): In vitro induction of androgenesis. In: Bajaj, YPS (Ed) Biotechnology in Agriculture and Forestry, Vol 12 (pp 250–265). Springer-Verlag, Berlin, Heidelberg
Milewska-Pawliczuk, E & Kubicki, B (1977) Induction of androgenesis in vitro in Malus domestica. Acta Hort. 78: 271–276
Murashige, T & Skoog, F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15: 473–497
Pedroso, MC & Pais, MS (1992) A SEM and X-ray microanalysis study during induction of morphogenesis in Camellia japonica L. Plant Sci. 87: 99–108
Pedroso, MC & Pais, MS (1993) Direct embryo formation in leaves of Camellia japonica L. Plant Cell Rep. 12: 639–643
Pedroso-Ubach MC (1991) Contribuição para a preservção e o melhoramento de Camellia japonica L. Faculdade de Ciências da Universidade de Lisboa. Master Thesis (English Abstract) (pp 23–50;89–103)
Raghavan, V & Nagmani, R (1989) Cytokinin effects on pollen embryogenesis in cultured anthers of Hyoscyamus niger. Can. J. Bot. 67: 247–257
Sunderland, N & Dunwell, JM (1974) Pathways in pollen embryogenesis. In: Street, HE (Ed) Tissue Culture and Plant Science (pp 141–167). Academic Press, New York
Taylor, DC, Weber, N, Underhill, EW, Pomery, MK, Keller, WA, Scowcroft, WR, Wilen, RW, Moloney, MM & Molbrook, LA (1990) Storage-protein regulation and lipid accumulation in microspore embryos of Brassica napus L. Planta 181: 18–26
Wang, C, Chu, Z & Sun, C (1975) The induction of Populus pollen plants. Acta Bot. Sin. 17: 56–59
Widholm, JM (1972) The use of fluoresceine diacetate and phenosafranine for determining viability of cultured plant cells. Stain Technol. 47: 189–194
Zhang YX, Lespinasse Y & Chevreau E (1990) In vitro androgenesis in apple (Malus domestica Borkh.). Abstracts WIIth International Congress on Plant Tissue and Cell Culture. Amsterdam, June 24–29, A6-10, 185
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pedroso, M.C., Pais, M.S. Induction of microspore embryogenesis in Camellia japonica cv. Elegans. Plant Cell Tiss Organ Cult 37, 129–136 (1994). https://doi.org/10.1007/BF00043606
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00043606