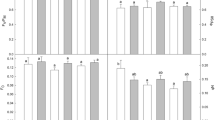
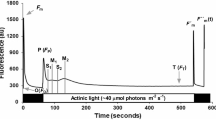
Abstract
The potential involvement of impaired photophosphorylation in the chilling sensitivity of photosynthesis in warm climate plant species has been a topic of investigation for more than two decades. With recent advances in the analysis of photosynthetic energy transduction in intact leaves, experiments are now possible that either address or avoid important uncertainties in the significance and interpretation of earlier in vitro work. Nevertheless, different laboratories using different techniques to analyze the effects of chilling in the light on photophosphorylation in intact cucumber (Cucumis sativus) leaves have come to very different conclusions regarding the role of impaired ATP formation capacity in the inhibition of net photosynthesis. In order to evaluate these discrepancies and bring this issue to a final resolution, in this investigation, we have made a detailed analysis of the decay of the flash-induced electrochromic shift and changes in chlorophyll fluorescence yield in cucumber leaves before, during and after a 5 h light-chill at chill temperatures of between 4 and 10°C. We feel that our findings address the major discrepancies in both data and interpretation as well as provide convincing evidence that photophosphorylation is not disrupted in cucumber leaves during or after light and chilling exposure. It follows that impaired photophosphorylation is not a contributing element to the inhibition of net photosynthesis that is widely observed in warm climate plants as a result of chilling in the light.
Article PDF
Similar content being viewed by others

Avoid common mistakes on your manuscript.
Abbreviations
- CF:
-
chloroplast coupling factor or CF1CF0-ATP synthase
- ΔA518 :
-
flash-induced electrochromic absorbance change measured at 518 nm
- DCCD:
-
N,N'-dicyclohexylcarbodiimide
- ΔμH + :
-
transmembrane electrochemical potential of hydrogen ions
- ‡Ψ:
-
the electrical charge component of ΔμH +
- ‡pH:
-
the hydrogen ion concentration component of ΔμH +
- F0 and Fm :
-
the yields of chlorophyll fluorescence from dark-adapted material when all Photosystem II centers are open (F0) or closed (Fm)
- F0' and Fm':
-
F0 and Fm measured in light-adapted material
- Fs :
-
steady-state chlorophyll fluorescence yield in light-adapted material
- QA :
-
primary quinone electron acceptor of Photosystem II
- PPFD:
-
photosynthetic photon flux density
References
Baker, NR, Long, SP and Ort, DR (1988) Photosynthesis and temperature, with particular reference to effects on quantum yield. In: Long, SP and Woodward, FI (eds) Plants and Temperature, pp 347–375. The Company of Biologists Ltd, Cambridge
Bradbury, M and Baker, NR (1981) Analysis of the slow phases of the in vivo chlorophyll fluorescence induction curve. Changes in the redox state of Photosystem II electron acceptors and fluorescence emission from Photosystems I and II. Biochim Biophys Acta 635: 542–551
Briantais, J-M, Vernotte, C, Picaud, M and Krause, GH (1979) A quantitative study of the slow decline of chlorophyll a fluorescence in isolated chloroplasts. Biochim Biophys Acta 548: 128–138
Butler, WL (1978) Energy distribution in the photochemical apparatus of photosynthesis. Annu Rev Plant Physiol 29: 345–378
Butler, WL (1980) Energy transfer between Photosystem II units in a connected package model of the photochemical apparatus of photosynthesis. Proc Natl Acad Sci USA 77: 4697–4701
Crofts, AR and Wraight, CA (1983) The electrochemical domain of photosynthesis. Biochim Biophys Acta 726: 149–185
Demming, B, Winter, K, Kruger, A and Czygan, F-C (1987) Photoinhibition and zeaxanthin formation in intact leaves. A possible role of the xanthophyll cycle in the dissipation of excess light energy. Plant Physiol 84: 218–224
Garber, MP (1977) Effect of light and chilling temperatures on chilling-sensitive and chilling-resistant plants. Plant Physiol 59: 981–985
Genty, B, Briantais, J-M, and Baker, NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta 990: 87–92
Hangarter, RP, Grandoni, P and Ort, DR (1987) The effects of chloroplast coupling factor reduction on the energetics of activation and on the energetics and efficiency of ATP formation. J Biol Chem 262: 13513–13519
Heber, U (1969) Conformational changes of chloroplasts induced by illumination of leaves in vivo. Biochim Biophys Acta 180: 302–319
Hutchison RS (1993) Thioredoxin-mediated reduction of photosynthetic carbon reduction enzymes following chilling in the light. PhD dissertation. University of Illinois, Urbana, IL
Junesch, U and Gräber, P (1987) Influence of the redox state and the activation of the chloroplast ATP synthase on proton-transport-coupled ATP synthesis/hydrolysis. Biochim Biophys Acta 893: 275–288
Junge, W and Witt, HT (1968) On the ion transport system of photosynthesis. Investigations on a molecular level. Z Naturforsch 23: 244–254
Kislyuk, IM and Vas'kovskii, MD (1972) Effect of cooling cucumber leaves on photosynthesis and photochemical reactions. Sov Plant Physiol 19: 688–692
van, Kooten, O and Snel, JFH (1990) The use of chlorophyll fluorescence nomenclature in plant stress physiology. Photosynth Res 25: 147–150
Kramer, DM and Crofts, AR (1989) Activation of the chloroplast ATPase measured by the electrochromic change in leaves of intact plants. Biochim Biophys Acta 976: 28–41
Kramer, DM, Wise, RR, Frederick, JR, Alm, DM, Hesketh, JD, Ort, DR and Crofts, AR (1990) Regulation of coupling factor in field-grown sunflower: A redox model relating coupling factor activity to the activities of other thioredoxin-dependent chloroplast enzymes. Photosynth Res 26: 213–222
Krause, GH (1973) The high-energy-state of the thylakoid membrane as indicated by chlorophyll fluorescence and chloroplast shrinkage. Biochim Biophys Acta 292: 715–728
Lill, H, Althoff, G and Junge, W (1987) Analysis of ionic channels by a flash spectrophotometric technique applicable to thylakoid membranes: CF0, the proton channel of the chloroplast ATP synthase, and, for comparison, gramicidin. J Membr Biol 98: 69–78
Lill, H and Junge, W (1989) CF0, the proton channel of chloroplast ATP synthase. After removal of CF1 it appears in two forms with highly different proton conductance. Eur J Biochem 179: 459–467
Mills, JD and Mitchell, P (1984) Thiol modulation of the chloroplast proton motive ATPase and its effect on photophorylation. Biochim Biophys Acta 764: 93–104
Morita, S, Itoh, S and Nishimura, M (1981) Acceleration of the decay of membrane potential after energization of chloroplasts in intact Zea mais leaves. Plant and Cell Physiol 22: 205–214
Murata, N and Sugahara, K (1969) Control of excitation transfer in photosynthesis. III. Light-induced decrease of chlorophyll a fluorescence related to photophosphorylation systems in spinach chloroplasts. Biochim Biophys Acta 189: 182–192
Nelson, N and Eytan, E (1979) Approach to the membrane sector of the chloroplast coupling device. In: Mukohata, Y and Packer, L (eds) Cation Flux across Biomembranes, pp 409–415. Academic Press, New York
Noctor, G, Rees, D, Young, A and Horton, P (1991) The relationship between zeaxanthin, energy-dependent quenching of chlorophyll fluorescence, and trans-thylakoid pH gradient in isolated chloroplasts. Biochim Biophys Acta 1057: 320–330
Ort, DR and Oxborough, K (1992) In situ regulation of chloroplast coupling factor activity. Ann Rev Plant Physiol Plant Mol Biol 43: 269–291
Ortiz-Lopez, A, Nie, GY, Ort, DR and Baker, NR (1990) The involvement of the photoinhibition of Photosystem II and impaired membrane energization in the reduced quantum yield of carbon assimilation in chilled maize. Planta 181: 78–84
Oxborough, K and Horton, P (1987) Characterization of the effects of antimycin A upon the high energy state quenching of chlorophyll fluorescence (qE) in spinach and pea chloroplasts. Photosynth Res 12: 119–128
Peeler, TC and Naylor, AW (1988a) A comparison of the effects of chilling on leaf gas exchange in pea (Pisum sativum L.) and cucumber (Cucumis sativus L.). Plant Physiol 86: 143–146
Peeler, TC and Naylor, AW (1988b) A comparison of the effects of chilling on thylakoid electron transfer in pea (Pisum sativum L.) and cucumber (Cucumis sativus L.) Plant Physiol 86: 147–151
Pick, U and Racker, E (1979) Purification and reconstitution of the N,N'-dicyclohexylcarbodiimide-sensitive ATPase complex from spinach chloroplasts. J Biol Chem 254: 2793–2799
Quick, WP and Horton, P (1984) Studies on the induction of chlorophyll fluorescence in barley protoplasts. II. Resolution of fluorescence quenching by redox state and the transthylakoid pH gradient. Proc R Soc London B 220: 371–382
Rees, D, Noctor, G, Ruban, AV, Crofts, J, Young, AJ and Horton, P (1992) pH-dependent chlorophyll fluorescence quenching in spinach thylakoids from light treated or dark adapted leaves. Photosynth Res 31: 11–19
Ruban, AV, Walters, RG and Horton, P (1992) The molecular mechanism of the control of excitation energy dissipation in chloroplast membranes: Inhibition of ΔpH-dependent quenching of chlorophyll fluorescence by dicyclo-hexylcarbodiimide. FEBS Lett 309: 175–179
Ruban, AV, Young, AJ and Horton, P (1993) Induction of nonphotochemical energy dissipation and absorbance changes in leaves. Plant Physiol 102: 741–750
Sassenrath, GF, Ort, DR and Portis, AR (1990) Impaired reductive activation of stromal biphosphatases in tomato leaves following low-temperature exposure at high light. Arch Biochem Biophys 282: 302–308
Terashima, I, Huang, L-K and Osmond, B (1989) Effects of leaf chilling on thylakoid functions, measured at room temperature, in Cucumis sativus L. and Oryza sativa L. Plant Cell Physiol 30: 841–850
Terashima, I, Yasuhito, K and Katoh, S (1991a) Exposure of leaves of Cucumis sativus L. to low temperatures in the light causes uncoupling of thylakoids I. Studies with isolated chloroplasts. Plant Cell Physiol 32: 1267–1274
Terashima, I, Yasuhito, K and Katoh, S (1991b) Exposure of leaves of Cucumis sativus L. to low temperatures in the light causes uncoupling of thylakoids II. Non-destructive measurements with intact leaves. Plant Cell Physiol 32: 1275–1283
Uribe, EG (1972) The interaction of N,N'-dicyclohexylcarbodiimide with the energy conservation systems of the spinach chloroplast. Biochemistry 11: 4228–4235
Wise, RR and Ort, DR (1989) Photophosphorylation after chilling in the light. Plant Physiol 90: 657–64.
Wise, RR, Terashima, I and Ort, DR (1990) The effect of chilling in the light on photophosphorylation. Analysis of discrepancies between in vitro and in vivo results. Photosynth Res 25: 137–139
Witt, HT (1979) Energy conversion in the functional membrane of photosynthesis. Analysis by light pulse and electric pulse methods. The central role of the electric field. Biochim Biophys Acta 505: 355–427
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Oxborough, K., Ort, D.R. In situ evidence that chilling in the light does not cause uncoupling of photophosphorylation or detachment of coupling factor in chilling-sensitive plants. Photosynth Res 43, 93–105 (1995). https://doi.org/10.1007/BF00042966
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00042966