Abstract
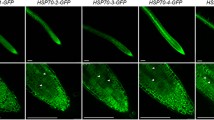
The heat shock (hs) response during plant growth and development was analyzed in tobacco and Arabidopsis using chimaeric β-glucuronidase reporter genes (hs-Gus) driven by a soybean hs promoter. Fluorimetric measurements and histochemical staining revealed high Gus activities in leaves, roots, and flowers exclusively after heat stress. The highest levels of heat-inducible expression were found in the vascular tissues. Without heat stress, a developmental induction of hs-Gus was indicated by the accumulation of high levels of Gus in transgenic tobacco seeds. There was no developmental induction of hs-Gus in Arabidopsis seeds. In situ hybridization to the RNA of the small heat shock protein gene Athsp17.6 in tissue sections revealed an expression in heat-shocked leaves but no expression in control leaves of Arabidopsis. However, a high level of constitutive expression of hs gene was detected in meristematic and provascular tissues of the Arabidopsis embryo. The developmental and tissue-specific regulation of the hs response is discussed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- hs:
-
heat shock
- Hsp:
-
heat shock protein(s)
- hs:
-
Gus: heat-inducible Gus gene(s)
- HSE:
-
heat shock element(s)
- HSF:
-
heat shock factor
- X-gluc:
-
5-bromo-4-chloro-3-indolyl-β-D-glucuronide
- Gus:
-
β-glucuronidase
- DAF:
-
days after flowering
- SAR:
-
scaffold attachment region
References
Almoguera C, Jordano J: Developmental and environmental concurrent expression of sunflower dry-seed-stored low-molecular-weight heat-shock protein and Lea mRNAs. Plant Mol Biol 19: 781–792 (1992).
Baumann G, Raschke E, Bevan M, Schöffl F: Functional analysis of sequences required for transcriptional activation of a soybean heat shock gene in transgenic tobacco plants. EMBO J 8: 2195–2202 (1987).
Bouchard RA: Characterization of expressed meiotic prophase repeat transcript clones of Lilium: meiosis specific expression, relatedness and affinities to small heat shock protein genes. Genome 33: 68–79 (1990).
Bradford MM: A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein dye binding. Anal Biochem 72: 248–254.
Coca MA, Almoguera C, Jordano J: Expression of sunflower low-molecular-weight heat-shock proteins during embryogenesis and persistence after germination: localization and possible functional implications. Plant Mol Biol 25: 479–792 (1994).
De Block M, Debrouwer D: In situ enzyme histochemistry on plastic embedded plant material. The development of an artefact-free β-glucuronidase assay. Plant J 2: 261–266 (1992).
DeRocher AE, Vierling E: Developmental control of small heat shock protein expression during pea seed maturation. Plant J 5: 93–102 (1994).
Dietrich PS, Bouchard RA, Casey ES, Sinibaldi RM: Isolation and characterization of a small heat shock protein gene from maize. Plant Physiol 96: 1268–1276 (1991).
Giraudat J, Hauge BM, Valon C, Smalle J, Parcy F, Goodman HM Isolation of the Arabidopsis ABI3 gene by positional cloning. Plant Cell 4: 1251–1261 (1992).
Györgyey J, Gartner A, Nemeth K, Magyar Z, Hirt H, Heberle-Bors E, Dudits D: Alfalfa heat shock genes are differentially expressed during somatic embryogenesis. Plant Mol Biol 16: 999–1007 (1991).
Helm KW, Abernethy RH: Heat shock proteins and their mRNAs in dry and early imbibing embryos of wheat. Plant Physiol 93: 1626–1633 (1990).
Helm KW, Vierling E: An Arabidopsis cDNA clone encoding a low molecular weight heat shock protein. Nucl Acids Res 17: 7995 (1989).
Helm KW, DeRocher A, Lauzon L, Vierling E: Expression of low molecular weight heat shock proteins during seed development. J Cell Biochem 14E: 301 (1990).
Hernandez LD, Vierling E: Expression of low molecular weight heat shock proteins under field conditions. Plant Physiol 101: 1209–1216 (1993).
Hogan BLM, Constantini F, Lacy E: Manipulating the Mouse Embryo: a Laboratory Manual, pp. 228–242. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY New York (1986).
Hübel A, Schöffl F: Arabidopsis heat shock factor: isolation and characterization of the gene and the recombinant protein. Plant Mol Biol, in press (1994).
Jakob U, Gaestel M, Engel K, Buchner J: Small heat shock proteins are molecular chaperones. J Biol Chem 268: 1517–1520 (1993).
Jefferson RA: Assaying chimeric genes in plants: the Gus gene fusion system. Plant Mol Biol Rep 5: 387–405 (1987).
Jinn TL, Wu SH, Yeh CH, Hsieh MH, Yeh YC, Chen YM, Lin CY: Immunological kinship of class I low molecular weight heat shock proteins and thermostabilization of soluble proteins in vitro among plants. Plant Cell Physiol 34: 1055–1062 (1993).
Kruse E, Liu Z, Kloppstech K: Expression of heat shock proteins during development of barley. Plant Mol Biol 23: 111–122 (1993).
Marrs KA, Casey ES, Capitant SA, Bouchard RA, Dietrich PS, Mettler IJ, Sinibaldi R: Characterization of two maize hsp90 heat shock protein genes and expression during heat shock, embryogenesis, and pollen development. Devel Genet 14: 27–41 (1993).
Pechan PM, Bartels D, Brown DCW, Schell J: Messenger-RNA and protein changes associated with induction of Brassica microspore embryogenesis. Planta 184: 161–165 (1991).
Rieping M, Schöffl F: Synergistic effects of upstream sequences, CCAAT box elements, and HSE sequences for enhanced expression of chimaeric heat shock genes in transgenic tobacco. Mol Gen Genet 321: 226–232 (1992).
Schmülling T, Schell J, Spena A: Promoters of the A, B and C genes of Agrobacterium rhizogenes are differentially regulated in transgenic plants. Plant Cell 1: 665–670 (1989).
Schöffl F: Aspekte der Temperaturstress-regulierten Genexpression bei höheren Pflanzen. In: Dechema-Monographie, vol. 95, pp. 323–338. Verlag Chemie, Weinheim (1984).
Schöffl F, Raschke E, Nagao RT: The DNA sequence analysis of soybean heat-shock genes and identification of possible regulatory promoter elements. EMBO J 3: 2491–2497 (1984).
Schöffl F, Rieping M, Baumann G, Bevan M, Angerm:uller S: The function of plant heat shock promoter elements in the regulated expression of chimaeric genes in transgenic tobacco. Mol Gen Genet 217: 246–253 (1989).
Schöffl F, Rieping M, Raschke E: Functional analysis of sequences regulating the expression of heat shock genes in transgenic plants. In: Lycett GW, Grierson D (eds) Genetic Engineering of Crop Plants, pp. 79–94. Butterworth, London (1990).
Schöffl F, Rieping M, Severin K: The induction of the heat shock response: activation and expression of chimaeric heat shock genes in transgenic tobacco. In: Herrmann RG, Larkins B (eds) Plant Molecular Biology, vol. 2, pp. 685–694. Plenum Press, New York (1991).
Schöffl F, Schröder G, Kliem M, Rieping M: An SAR sequence containing 395-bp DNA fragment mediates enhanced gene-dosage-correlated expression of a chimaeric heat shock gene in transgenic tobacco plants. Transgen Res 2: 93–100 (1993).
Severin K, Schöffl F: Heat-inducible hygromycin resistance in transgenic tobacco. Plant Mol Biol 15: 827–833 (1990).
Severin K, Wagner A, Schöffl F: A heat-inducible Adh gene in Arabidopsis as a reporter of negative selection in transgenic Arabidopsis. Transgen Res, in press (1994).
Takahashi T, Naito S, Komeda Y: The Arabidopsis hsp18.2 promoter/Gus gene fusion in transgenic Arabidopsis plants: a powerful tool for the isolation of regulatory mutants of the heat shock response. Plant J 2: 751–761 (1992).
Vierling E: The roles of heat shock proteins in plants. Annu Rev Plant Physiol Plant Mol Biol 42: 579–620 (1991).
Vierling E, Sun A: Developmental expression of heat shock proteins in higher plants. In: Cherry J (ed) Environmental stress in plants. Biochemical and physiological mechanism associated with environmental stress tolerance in plants, pp. 343–354. Springer-Verlag, New York (1987).
Zarsky V, Garrido D, Eller N, Tupy J, Vicente O, Schöffl F, Heberle-Bors E: The expression of a small heat shock gene is activated during induction of tobacco pollen embryogenesis by starvation. Plant Cell Environ (in press).
Zimmermann JL, Apuya N, Darwish K, O'Carroll C: Novel regulation of heat shock genes during carrot somatic embryo development. Plant Cell 1: 1137–1146 (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Prändl, R., Kloske, E. & Schöffl, F. Developmental regulation and tissue-specific differences of heat shock gene expression in transgenic tobacco and Arabidopsis plants. Plant Mol Biol 28, 73–82 (1995). https://doi.org/10.1007/BF00042039
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00042039