Abstract
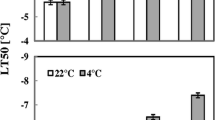
Cell suspension cultures were initiated from callus derived from xylem tissues of peach [Prunus persica (L.) Batsch]. Cold acclimation was induced (LT50 of-13°C) in cell suspensions at 3°C in the dark for 10 days. Freezing tolerance returned to the level of nonacclimated cells (LT50 of −4.5°C) when cold-acclimated cells were transferred to 24°C (in dark) for 3 days. Addition of 75 μM abscisic acid (ABA) to the growth medium failed to induce cold acclimation after cells were cultured for 5 days at 24°C. Microvacuolation, cytoplasmic augmentation and disappearance of starch grains were observed in cells that were cold-acclimated by exposure to low temperature. Similar ultrastructural alterations were not observed in ABA-treated cells. Several qualitative and quantitative changes in proteins were noted during both cold acclimation and ABA treatment. Both the ultrastructural and protein changes observed during cold acclimation were reversed during deacclimation. The relationship of these changes to cold acclimation in peach cell-cultures is discussed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- ABA:
-
abscisic acid
- 2,4-d :
-
2,4-dichlorophenoxyacetic acid
- IBA:
-
indole-3-butyric acid
- Ms:
-
Murashige & Skoog
- PMSF:
-
phenylmethylsulfonyl fluoride
- LT50 or Freezing Tolerance:
-
temperature that resulted in 50% decrease in TTC reduction
- TTC:
-
2,3,5-triphenyltetrazolium chloride
References
Arora R, Wisniewski ME & Scorza R (1992) Cold acclimation in genetically related (sibling) deciduous and evergreen peach (Prunus persica [L.] Batsch). Plant Physiol. 99: 1562–1568
Borochov A, Walker MA & Pauls KP (1989) Effect of cold acclimation on the morphological and physiological properties of alfalfa (Medicago sativa) suspension culture cells. J. Plant Physiol. 133: 671–677
Chen PM & Gusta LV (1982) Cold acclimation of wheat and smooth brome-grass cell suspensions. Can. J. Bot. 60: 1207–1211
Chen THH & Gusta LV (1983) Abscisic acid-induced freezing resistance in cultured plant cells. Plant Physiol. 73: 71–75
Christersson L (1978) The influence of photoperiod and temperature on the development of frost hardiness in seedlings of Pinus sylvestris and Picea abies. Physiol. Plant. 44: 288–294
Gnanapragasam S & Vasil IK (1992) Ultrastructural changes in suspension culture cells of Panicum maximum during cryopreservation. Plant Cell Rep. 11: 169–174
Guy CL (1990) Cold acclimation and freezing stress tolerance: role of protein synthesis. Annu. Rev. Plant Physiol. Plant Mol. Biol. 41: 187–223
Hellergren J (1983) Cold acclimation of suspension cultures of Pinus sylvestris in response to light and temperature treatments. Plant Physiol. 72: 992–995
Johnson-Flanagan Am & Singh J (1987) Alteration of gene expression during the induction of freezing tolerance in Brassica napus suspension cultures. Plant Physiol. 85: 699–705
Kuroda H & Sagisaka S (1993) Ultrastructural changes in cortical cells of apple (Malus pumila Mill.) associated with cold hardiness. Plant Cell Physiol. 34: 357–365
Lee SP, Zhu B, Chen THH & Li PH (1992) Induction of freezing tolerance in potato (Solanum commersonii) suspension cultured cells. Physiol. Plant 84: 41–48
Niki T & Sakai A (1981) Ultrastructural changes related to frost hardiness in the cortical parenchyma cells from mulberry twigs. Plant Cell Physiol. 22: 171–183
Orr W, Keller W & Singh J (1986) Induction of freezing tolerance in an embryogenic cell suspension culture of Brassica napus by abscisic acid at room temperature. J. Plant Physiol. 126: 23–32
Perry TO & Hellmers H (1973) Effects of abscisic acid on growth and dormancy of two races of red maple. Bot. Gaz. 134: 283–289
Pomeroy MK & Siminovitch D (1971) Seasonal cytological changes in secondary phloem parenchyma cells in Robinia pseudoacacia in relation to cold hardiness. Can. J. Bot. 49: 787–795
Reaney MJT & Gusta LV (1987) Factors influencing the induction of freezing tolerance by abscisic acid in cell suspension cultures of Bromus inermis Leyess and Medicago sativa L. Plant Physiol. 83: 423–427
Robertson AJ, Gusta LV, Reaney MJT & Ishikawa M (1987) Protein synthesis in bromegrass (Bromus inermis Leyss) cultured cells during the induction of frost tolerance by abscisic acid or low temperature. Plant Physiol. 84: 1331–1336
Robertson AJ, Gusta LV, Reaney MJT & Ishikawa M (1988) Identification of proteins correlated with increased freezing tolerance in bromegrass cell cultures. Plant Physiol. 86: 344–347
Sakai A & Yoshida S (1968) The role of sugar and related compounds in variations of freezing resistance. Cryobiology 5: 160–174
Sauter JJ & van Cleve B (1991) Biochemical and ultrastructural results during starch-sugar conversion in ray parenchyma cell of Populus during cold adaptation. J. Plant Physiol. 139: 19–26
Schiavone FM & Wisniewski ME (1990) Callus and cell suspension cultures from dormant stems of peach. HortScience 25: 483
Siminovitch D, Wilson CM & Briggs DR (1953) Studies on the chemistry of the living bark of the black locust in relation to its frost hardiness. Seasonal transformations and variations in the carbohydrates: starch-sucrose interconversions. Plant Physiol. 28: 383–400
Tanino K, Weiser CJ, Fuchigami LH & Chen THH (1990) Water content during abscisic acid induced freezing tolerance in bromegrass cells. Plant Physiol. 93: 460–464
Tanino KK, Chen THH, Fuchigami LH & Weiser CJ (1991) Abscisic acid-induced cellular alterations during the induction of freezing tolerance in bromegrass cells. J. Plant Physiol. 137: 619–624
Towill LE & Mazur P (1975) Studies on the reduction of 2,3,5-triphenyltetrazolium chloride as a viability assay for plant tissue cultures. Can. J. Bot. 53: 1097–1102
Tremblay M-F, Nadeau P & Lalonde M (1992) Effect of ABA on freezing resistance of Betula papyrifera and Alnus incana woody plant cell suspensions. Tree Physiol. 10: 317–326
Wallner SJ, Wu M-T & Anderson-Krengel SJ (1986) Changes in extracellular polysaccharides during cold acclimation of cultured pear cells. J. Amer. Soc. Hort. Sci. 111: 769–773
Wisniewski ME & Ashworth EN (1986) A comparison of seasonal ultrastructural changes in the stem tissues of peach (Prunus persica) that exhibit contrasting mechanisms of cold hardiness. Bot. Gaz. 147–417
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Arora, R., Wisniewski, M.E. Ultrastructural and protein changes in cell suspension cultures of peach associated with low temperature-induced cold acclimation and abscisic acid treatment. Plant Cell Tiss Organ Cult 40, 17–24 (1995). https://doi.org/10.1007/BF00041113
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00041113