Abstract
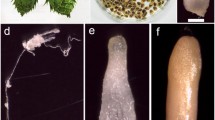
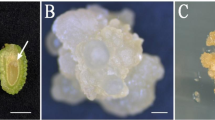
For the purpose of developing somatic embryogenesis in Prunus avium L., immature zygotic embryos, collected from five donor trees and sorted into two size classes (C1: 2.5–3.5 and C2: 3.6–4.5 mm), received various experimental treatments. When cultured for 10 days on an inductive medium containing 18.1 μM 2,4-dichlorophenoxyacetic acid (2,4-d) and 9.3 μM kinetin, then transferred to fresh medium without growth regulators, 2.5% of the C1 class cotyledons expressed direct somatic embryogenesis. C2 class cotyledons were less responsive. The response was also influenced by the chosen donor tree. In a few cases, spontaneous germination occurred. The presence of a root meristem was clearly demonstrated by histological examination of longitudinal sections. The replacement of half the amount of 2,4-d, present in the inductive medium mentioned above, by the same quantity of naphthaleneacetic acid reduced the incidence of somatic embryogenesis. Conversely, a rhizogenic response was strongly enhanced. When submitted to an inductive medium containing indoleacetic acid and zeatin without any subcultures for 3 months, C1 class cotyledons were the most morphogenic and developed leaves and cotyledon-like structures.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BA:
-
6-benzyladenine
- 2,4-d :
-
2,4-dichlorophenoxyacetic acid
- IAA:
-
indoleacetic acid
- IBA:
-
indolebutyric acid
- NAA:
-
α-naphthaleneacetic acid
References
Agresti A (1990) Categorial Data Analysis. John Wiley & Sons Inc (p. 558)
Bonneau L, Beranger-Novat N, Martin-Tanguy J & Monin J (1990) Polyamines and somatic embryogenesis in spindle tree (Euonymus europaeus L.). The effects of difluromethylarginine and difluoromethylornithine. In: Flores HE, Arteca RN & Shannon JC (Eds) Polyamines and Ethylene: Biochemistry, Physiology, and Interactions (pp 363–367). American Society of Plant Physiologists, Pennsylvania State University Press, University Park
Chalupa V (1990) Plant regeneration by somatic embryogenesis from cultured immature embryos of oak (Quercus robur L.) and linden (Tilia cordata Mill.) Plant Cell Rep. 9: 398–401
David H, Domon JM, Savy C, Miannay N, Sulmont G, Dargent R & David A (1992) Evidence for early stages of somatic embryo development in a protoplast-derived cell culture of Prunus avium. Physiol. Plant. 85: 301–307
De March G, Filloux D, Miannay N, Sulmont G, Grenier E & David A (1990) In vitro morphogenic responses of zygotic embryos in Prunus avium L. In: Le BiopôleVégétal (Ed) Seeds: Genesis of Natural & Artificial Forms-Biotechnologies '90; International Symposia in Picardie, Proceedings (pp 155–158). Le Biopôle, Amiens, France
Druart P (1981) Embryogenèse somatique et obtention de plantules chez Prunus incisa x serrula (GM9), cultivé in vitro. Bull. Rech. Gembloux 16: 205–220
Druart P (1990) Improvement of somatic embryogenesis of the cherry dwarf rootstock Inmil/GM9 by the use of different carbon sources. Acta Hort. 280: 125–127
Feucht W & Dausend D (1976) Root induction in vitro of easy-to-root Prunus pseudocerasus and difficult-to-root Prunus avium. Scienta Hort. 4: 49–54
Gabe M (1968) Les principes généraux de la technique histologique. In: Gabe M (Ed) Techniques Histologiques (pp 11–184). Masson et Cie, Paris
Geneve RL & Kester ST (1990) The initiation of somatic embryos and adventitious roots from developing zygotic embryo explants of Cercis canadensis L. cultured in vitro. Plant Cell Tiss. Org. Cult. 22: 71–76
Hammerschlag FA, Bauchan G & Scorza R (1985) Regeneration of peach from calus derived from immature embryos. Theor. Appl. Genet. 70: 248–251
James DJ, Mackenzie KAD & Malhotra SB (1987) The induction of hexaploidy in cherry rootstocks using in vitro regeneration techniques. Theor. Appl. Genet. 73: 589–594
Jones OP, Gayner A & Watkins R (1984) Plant regeneration from callus tissue cultures of the cherry rootstock Colt (Prunus avium x P. pseudocerasus) and the apple rootstock M. 25 (Malus pumila). J. Hort. Sci. 59: 463–467
Krogstrup P (1986) Embryolike structures from cotyledons and ripe embryos of Norway spruce (Picea abies). Can. J. For. Res. 16: 644–668
Lane WD & Cossio F (1986) Adventitious shoots from cotyledons of immature cherry and apricot embryos. Can. J. Plant Sci. 66: 953–959
Mante S. Scorza R & Cordts JM (1989) Plant regeneration from cotyledons of Prunus persica, Prunus domestica and Prunus cerasus. Plant Cell Tiss. Org. Cult. 19: 1–11
Morel G & Wetmore RH (1951) Fern callus tissue culture. Amer. J. Bot. 38: 141–143
Murashige T & Skoog F (1962) A revised medium for rapid growth and biossays with tobacco tissue cultures. Physiol. Plant. 15: 473–497
Ochatt SJ (1991) Strategies for plant regeneration from mesophyll protoplasts of the recalcitrant fruit and farm woodland species Prunus avium L. (Sweet/wild cherry), Rosaceae. J. Plant Physiol. 139: 155–160
Raj Bhansali R, Driver JA & Durzan DJ (1990) Rapid multiplication of adventitious somatic embryos in peach and nectarine by secondary embryogenesis. Plant Cell Rep. 9: 280–284
Riffaud JL & Cornu D (1981) Utilisation de la culture in vitro pour la multiplication de merisiers adultes (Prunus avium L.) sélectionnés en forê. Agronomie 1: 633–640
SAS Institute Inc (1988) SAS/STAT User's Guide Release 6.03 Edition. Cary, NC: SAS Institute Inc (p. 1028)
Sotak RJ, Sommer HE & Merkle SA (1991) Relation of the developmental stage of zygotic embryos of yellow-poplar to their somatic embryogenic potential. Plant Cell Rep. 10: 175–178
Tulecke W & McGranahan G (1985) Somatic embryogenesis and plant regeneration from cotyledons of walnut, Juglans regia L. Plant Sci. 40: 57–63
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
De March, G., Grenier, E., Miannay, N. et al. Potential of somatic embryogenesis in Prunus avium immature zygotic embryos. Plant Cell Tiss Organ Cult 34, 209–215 (1993). https://doi.org/10.1007/BF00036104
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00036104