Abstract
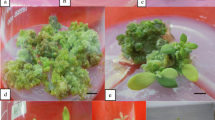
Conditions for plant regeneration from excised shoot tips of Vigna radiata were studied. Complete plants were regenerated directly without an intervening callus phase from shoot tips on basal medium (MS salts+B5vitamins). Regeneration frequency varied with genotype, explant size and growth regulator combinations in the medium. Addition of cytokinins induced a variable amount of callus at the base of the shoot tip, followed by multiple shoot formation. Benzyladenine (BA), kinetin and zeatin at 5×10-6 M each induced multiple shoots in 100% of the explants but the highest number of regenerants per explant (9) was produced with BA. The efficacy of BA for shoot multiplication was not improved when it was supplemented with naphthaleneacetic acid (NAA) or indoleacetic acid (IAA). NAA or adenine sulphate, when applied alone, induced complete plantlets. The growth regulator requirement of explants for the induction of multiple shoots varied with explant size. The shoot tip explants maintained proliferation ability on subculture. None of the treatments was effective in inducing shoot bud differentiation from callus. Regenerated shoots were rooted on MS basal medium and MS supplemented with either IAA or indolebutyric acid. The rooted plants were transferred to the field; 60% subsequently survived and grew.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BM:
-
basal medium [MS (Murashige & Skoog 1962) salts+B5 (Gamborg et al. 1968) vitamins]
- BA:
-
6-benzyladenine
- AdS:
-
adenine sulphate
- IAA:
-
indole-3-acetic acid
- NAA-1:
-
naphthaleneacetic acid
- IBA:
-
indolebutyric acid
References
Bajaj YPS & Dhanju MS (1979) Regeneration of plants from apical meristem tips of some legumes. Current Sci. 48: 906–907
Bhojwani SS (1981) A tissue culture method for propagation and low temperature storage of Trifolium repens genotypes. Physiol. Plant. 52: 187–190
Bhojwani SS, Mullins K & Cohen D (1984) Intervarietal variation for in vitro plant regeneration in the genus Trifolium. Euphytica 33: 915–921
Bruning JL & Kintz BL (1977) Computational hand-book of statistics. 2nd Edition, Scott, Foresman, Glenview, California
Dale PJ (1977) Meristem tip culture in Lolium, Festuca, Phleum and Dactylis. Plant Sci. Lett. 9: 333–338
Gamborg OL, Miller RA & Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Exp. Cell Res. 50: 151–158
Goel S, Mudgal AK & Gupta SC (1983) Development of plants from in vitro cultured shoot tips of Vigna mungo and V. radiata. Trop. Plant Sci. Res. 1: 31–33
Gulati A & Jaiwal PK (1990) Culture conditions effecting plant regeneration from cotyledon of Vigna radiata (L.) Wilczek. Plant Cell Tiss. Org. Cult. 23: 1–7
Hammatt N, Ghose TK & Davey MR (1986) Regeneration in legumes. In: Vasil IK (Ed) Cell Culture and Somatic Cell Genetics of Plants, Vol 3 (pp 67–85). Academic Press, Inc. Orlando, Florida
Hoque MI, Hoque MM, Begum A & Islam AS (1984) In vitro regeneration of plantlets from different explants of Vigna mungo (L.) Hepper. Bangladesh J. Bot. 13: 45–51
Jaiwal PK & Bhambie S (1990) Influence of growth regulating substances on the morphology of shoot apex of Vigna radiata (L.) Wilczek. Proc. Nat. Sci. Acad. India (in press)
Kartha KK (1982) Genepool conservation through tissue culture. In: Rao AN (Ed) Proc. Int. Sym. (pp 213–218) National University, Singapore
Kartha KK, Leung NL & Gamborg OL (1979) Freezepreservation of pea meristem in liquid nitrogen and subsequent plant regeneration. Plant Sci. Lett. 15: 7–15
Kartha KK, Leung NL & Pahl N (1980) Cryopreservation of strawberry meristems and mass propagation of plantlets. J. Amer. Soc. Hort. Sci. 105: 481–484
Kartha KK, Pahl K, Leung NL & Mroginski LA (1981) Plant regeneration from meristems of grain legumes: Soybean, cowpea, peanut, chickpea and bean. Can. J. Bol. 59: 1671–1679
Malmberg RL (1979) Regeneration of whole plants for callus culture of diverse genetic lines of Pisum sativum L. Planta 146: 243–244
Mathews H (1987) Morphogenetic responses from in vitro cultured seedling explants of mung bean (Vigna radiata L. Wilczek). Plant Cell Tiss. Org. Cult. 11: 233–240
Murashige T & Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15: 473–497
Rubluo A, Mroginski LA & Kartha KK (1982) Morphogenetic responses of pea leaflets cultured in vitro. In: Fujiwara A (Ed) Plant Tissue Culture (pp 151–152). The Japanese Association for Plant Tissue Culture, Tokyo
Rubluo A & Kartha KK (1985) In vitro culture of shoot apical meristem of various Phaseolus species and cultivars. J. Plant Physiol. 119: 425–433
Singh RP, Singh BD, Singh RM & Jaiswal H (1985) Genotypic differences in callus growth and organogenesis in greengram. Indian. J. Agric. Sci. 55: 612–615
Templeton-Somers KM & Collins WW (1986) Heritability of regeneration in tissue of sweet potato (Ipomea batatas). Theor. Appl. Genet. 71: 835–841
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gulati, A., Jaiwal, P.K. In vitro induction of multiple shoots and plant regeneration from shoot tips of mung bean (Vigna radiata (L.) Wilczek). Plant Cell Tiss Organ Cult 29, 199–205 (1992). https://doi.org/10.1007/BF00034353
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00034353