Summary
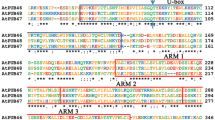
Soybean contains a multigene family which encodes the small subunit of ribulose-1,5-bisphosphate carboxylase (RuBPCss). A member of this gene family, SRS4, has been isolated from a soybean genomic DNA library. Its nucleotide sequence has been determined and compared to the sequence of SRS1, a previously characterized RuBPCss gene from soybean. Relevant regulatory sequences such as the PuPuCCAAT boxes, TATA box, the actual start of transcription and poly A addition sites are conserved between the two genes. Using a gene specific synthetic probe to the 3′ flanking region the steady state mRNA levels of SRS4, like SRS1, are shown to be very high in light grown soybean seedlings and low in seedlings grown in darkness. SRS1 and SRS4 are very closely related, the three exons being 96%, 93% and 96.5% homologous in nucleotide sequence. The polypeptide sequences are nearly identical with only one amino acid change in each of the three exons encoding the 178 amino acid precursor polypeptide. The two introns are about 75% homologous and the flanking regions are more than 85% homologous (700 base pairs on the 5′ end and 300 base pairs on the 3′ end). Furthermore, hybridization studies between lambda clones containing the SRS1 and SRS4 genes reveal that a region of strong homology extends at least 4 kb on the 5′ end and about 1.1 kb on the 3′ end. We propose that these two genes may be alloalleles or homeologous alleles. This proposal is consistent with soybean having an allotetraploid origin, and would imply that the divergence of two ancient Papilionoidae species gave rise to these two genes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Benoist C, Chambon P: Deletions covering the putative promoter region of early mRNAs of simian virus 40 do not abolish T-antigen expression. Proc Natl Acad Sci USA 77:3865–9, 1980.
Benoist C, Chambon P: In vivo sequence requirements of the SV40 early promoter region. Nature 279:336, 1981.
Berget SM: Are U4 small nuclear ribonuclear proteins involved in polyadenylation? Letters to Nature 309:179–182, 1984.
Berry-Lowe SL: The isolation and characterization of a ribulose-1,5-bisphosphate carboxylase small subunit gene in soybean. Dissertation, University of Georgia, 1985.
Berry-Lowe SL, Mc Knight TD, Shah DM, Meagher RB: The nucleotide sequence, expression and evolution of one member of a multigene family encoding the small subunit of ribulose-1,5-bisphosphate carboxylase in soybean. J Mol Appl Genet 1:483–498, 1982.
Berry-Lowe SL, Meagher RB: Transcriptional regulation of a gene encoding the small subunit of ribulose-1,5-bisphosphate carboxylase in soybean tissue is linked to the phytochrome response. Mol Cell Biol 5:1910–1917, 1985.
Beversdorf WD, Bingham ET: Male-sterility as a source of haploids and polypoids ofGlycine max. Can J Genet Cytol 19:283–287, 1977.
Bolivar F: Construction and characterization of new cloning vehicles, III. Derivatives of plasmid pBR322 carrying unique EcoRI sites for selection of EcoRI generated recombinant molecules. Gene 4:121–36, 1978.
Broglie R, Bellemaire G, Bartlett SG, Chua N-H, Cashmore AR: Cloned DNA sequences complementary to mRNA encoding precursors to the small subunit of ribulose-1,5-bisphosphate carboxylase and a chlorophyll a/b binding polypeptide. Proc Natl Acad Sci USA 78:7304–8, 1981.
Broglie R, Coruzzi G, Lamppa G, Keith B, Chua N-H: Structural analysis of nuclear gene coding for the precursor to the small subunit of wheat ribulose-1,5-bisphosphate carboxylase. Biotechnol 1:55–61, 1983.
Breathnach R, Chambon P: Organization and expression of eukaryotic split genes coding for proteins . Ann Rev Biochem 50:349–384.
Cashmore T: Nuclear genes encoding the small subunit of ribulose bisphosphate carboxylase. In: Kosuge T, Meridith C, Hollander A (eds) Genetic Engineering of Plants. An Agricultural Perspective. Plenum Press, New York, 1983, pp 29–38.
Coruzzi G, Broglie R, Cashmore A, Chua N-H: Nucleotide sequences of two pea cDNA clones encoding the small subunit of ribulose-1,5-bisphosphate carboxylase and the major chlorophyll a/b-binding thylakoid polypeptide. J Biol Chem 258: 1399–1402, 1983.
Coruzzi G, Broglie R, Edwards C, Chua N-H: Tissuespecific and light-regulated expression of pea nuclear gene encoding the small subunit of ribulose-1,5-bisphosphate carboxylase. EMBO Journal 3: 1671–1679, 1984.
Crane CF, Beversdorf WD, Bingham ET: Chromosome pairing and associations at meiosis in haploid soybean (Glycine max). Can J Genet Cytol 24: 293–300, 1982.
Crepet WL, Taylor DW: The diversification of the Leguminosae: First fossil evidence of the Mimosoidene and Papilionoideae. Science 228: 1087–1089, 1985.
Cutter GL, Bingham ET: Effect of soybean male-sterile gene ms on organization and function of the female gametophyte. Crop Science 17: 760–764, 1977.
Dean C, van den Elzen P, Tamaki S, Dunsmuir P, Bedbrook J: Differential expression of the eight genes of the petunia ribulose bisphosphate carboxylase small subunit multi-gene family. EMBO J 4: 3055–3061, 1985.
Delannay X, Rodgers DM, Palmer RG: Relative genetic contributions among ancestral lines to North American soybean cultivars. Crop Science 23: 944–949, 1983.
Dierks P, van Ooyen A, Mantei N, Wiessmann C: DNA sequences preceding the rabbit B-globin gene are required for formation in mouse L cells of β-globin RNA with the correct 5′ terminus. Proc Natl Acad Sci USA 78: 1411–1415, 1981.
Dunsmuir P, Smith S, Bedbrook J: A number of different nuclear genes for the small subunit of RuBPCase are transcribed in petunia. Nuc Acids Res 11: 4177–4185, 1983.
Eckenrode VK, Arnold J, Meagher RB: Comparison of the nucleotide sequence of soybean 18S rRNA with the sequences of other small-subunit rRNAs. J Mol Evol 21: 259–269, 1985.
Favaloro J, Freisman R, Kamen R: Transcription maps of polyoma virusspecific RNA: Analysis by two dimensional nuclease S1 gel mapping. In: Grossman L, Moldave K (eds) Methods in Enzymology. Academic Press, New York, 1980, Vol. 65, pp 718–749.
Gallagher TF, Ellis RF: Light stimulated transcription of genes for two chloroplast polypeptides in isolated pea leaf nuclei. EMBO J 1: 1493–1498.
Gottlieb LD: Conservation and duplication of isozymes in plants. Science 216: 373–380, 1982.
Grosveld GC, deBoer E, Shewmaker CK, Flavell RA: DNA sequences necessary for transcription of the rabbit B-globinin vivo. Nature 295: 120–126, 1982.
Hamada H, Petrino MG, Kakunaga T: A novel element with Z-DNA forming potential is widely found in evolutionarily diverse eukaryotic genomes. Biochem. 79: 6465–6469, 1982.
Hamada H, Seidman M, Howard BJ, Gorman CM: Enhanced gene expression by the poly(dT-dG)-poly(dC-dA) sequence. Cell 12: 2622–2630, 1984.
Hightower RC, Meagher RB: Divergence and differential expression of soybean actin genes. EMBO Journal 4: 1–8, 1985.
Hentschel C, Irminger J-C, Bucher P, Birnstiel ML: Sea urchia histone mRNA termini are located in gene region downstream from putative regulatory sequences. Nature 285: 147–151, 1980.
Herrera-Estrella L, van den Broeck G, Maenhaut R, van Montagu M, Schell J, Timko M, Cashmore A: Light inducible and chloroplast-associated expression of a chimaeric gene introduced intoNicotiana tabacum using a Ti plasmid vector. Nature 310: 115–120, 1984.
Hymowitz T: On the domestication of the soybean. Econ Bot 24:40B SB107.E1G1, 1970.
Jenson RG, Bahr JT: Ribulose-1,5-bisphosphate carboxylase-oxygenase. Ann Rev Plant Physiol 28: 379–400, 1977.
Johnson DA, Gautsch JW, Sportsman JR, Elder JH: Improved technique utilizing nonfat dry milk for analysis of proteins and nucleic acids transferred to nitrocellulose. Gene Anal Techn. 1: 3–8, 1984.
Kawashima N, Wildman SG: Studies on fraction 1 protein IV. Mode of inheritance of primary structure in relation to whether chloroplast or nuclear DNA contains the code for a choroplast protein. Biochim Biophys Acta 262: 42–9, 1972.
Maxam A, Gilbert W: Sequencing end-labeled DNA with base-specific chemical cleavages. In: Grossman L, Moldave K (eds) Methods in Enzymology, Academic Press, New York, 1980, Vol. 65, pp 499–560.
Meagher RB, Shepherd RJ, Boyer HW: The structure of cauliflower mosaic virus 1. A restriction endonuclease map of cauliflower mosaic virus DNA. Virology 80: 362–75, 1977.
Mishkind ML, Wessler SR, Schmidt GW: Functional determinants in transit sequences: Import and partial maturation by vascular plant chloroplasts of ribulose-1,5-bisphosphate carboxylase small subunit ofChamydomonas. J Cell Biol 100: 226–234, 1985.
Miyata T, Hayashida H: Recent divergence from a common ancestor of human IFN-alpha genes. Nature 295: 165–6, 1982.
Morelli G, Nagy F, Fraley RT, Rogers SG, Chua N-H: A short conserved sequence is involved in the light-inducibility of a gene encoding ribulose-1,5-bisphosphate carboxylase small subunit of pea. Nature 315: 200–204, 1985.
Muller J: Fossil-pollen records of extant angiosperms. Botanical Rev 47: 1–142, 1981.
Nagao RT, Shah DM, Eckenrode VK, Meagher RB: Multigene family of actin-related sequences isolated from a soybean genomic library. DNA 1: 1–9, 1981.
Nordheim A, Rich A: Negatively supercoiled simian virus 40 DNA contains Z-DNA segments within transcriptional enhancer elements. Nature 303: 674, 1983.
Perler F, Efstratiadis A, Lomedico P, Gilbert W, Kolodner R, Dogson J: The evolution of genes: The chicken preproinsulin gene. Cell 20: 555–66, 1980.
Polans NO, Weeden NF, Thompson WF: Inheritance organization and mapping of rbcS and cab multigene families in pea. Proc Natl Acad Sci USA 82: 5083–5087, 1985.
Proudfoot NJ: Eukaryotic promoters? Nature 279: 376, 1979.
Sasaki Y, Tomoda Y, Kamikubo T: Light regulates the gene expression of ribulose-1,5-bisphosphate carboxylase at the levels of transcription and gene dosage in greening pea leaves. FEBS Lett 173: 31–35, 1984.
Shah DM, Hightower RC, Meagher RB: Complete nucleotide sequence of a soybean actin gene. Proc Natl Acad Sci USA 79: 1022–6, 1982.
Shah DM, Hightower RC, Meagher RB: Genes encoding actin in higher plants: Intron positions are highly conserved but the coding sequences are not. J Mol Appl Gen 2: 111–126, 1983.
Silverthorne J, Tobin EM: Demonstration of transcriptional regulation of specific genes by phytochrome action. Proc Natl Acad Sci USA 81: 1112–1116, 1984.
Simpson J, Timko MP, Cashmore AR, Schell J, Van Montagu M, Herrera-Estrella L: Light-inducible and tissue-specific expression of a chimaeric gene under control of the 5′-flanking sequence of a pea chlorophyll a/b-binding protein gene. EMBO J 4: 2723–2729, 1985.
Smith HO, Birnstiel ML: A simple method for DNA restriction site mapping. Nucl Acids Res 3: 2387–98, 1976.
Sorrells ME, Bingham ET: Reproductive behavior of soybean haploids carrying the ms allele. Can J Genet Cytol 21: 449–455, 1979.
Stiekema WJ, Wimpee CF, Silverthorne J, Tobin EM: Phytochrome control of the expression of two nuclear genes encoding chloroplast proteins inLemna gibba L.G-3. Plant Physiol 72: 717–724, 1983.
Stiekema WJ, Wimpee CF, Tobin EM: Nucleotide sequence encoding the precursor of the small subunit of ribulose-1,5-bisphosphate carboxylase fromLemna gibba L.G-3. Nuc Acids Res 11: 8051–8061, 1983.
Viera J, Messing J: The pUC plasmids, an M13mp7-derived system for insertion mutagenesis and sequencing with synthetic universal primers. Gene 19: 259–268, 1982.
Weiher H, Konig M, Gruss P: Multiple point mutations affecting the Simian virus 40 enhancer. Science 219: 626–631, 1983.
Wimpee CS: PhD thesis. Organization and expression of light-regulated genes inLemna gibba L. G-3. Univ of Calif, 1984.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Grandbastien, M.A., Berry-Lowe, S., Shirley, B.W. et al. Two soybean ribulose-1,5-bisphosphate carboxylase small subunit genes share extensive homology even in distant flanking sequences. Plant Mol Biol 7, 451–465 (1986). https://doi.org/10.1007/BF00020329
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00020329