Abstract
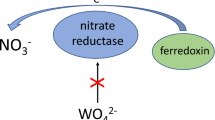
The first two genes of ferredoxin-dependent glutamate synthase (Fd-GOGAT) from a prokaryotic organism, the cyanobacterium Synechocystis sp. PCC 6803, were cloned in Escherichia coli. Partial sequencing of the cloned genomic DNA, of the 6.3 kb Hind III and 9.3 kb Cla I fragments, confirmed the existence of two different genes coding for glutamate synthases, named gltB and gltS. The gltB gene was completely sequenced and encodes for a polypeptide of 1550 amino acid residues (M r 168 964). Comparative analysis of the gltB deduced amino acid sequence against other glutamate synthases shows a higher identity with the alfalfa NADH-GOGAT (55.2%) than with the corresponding Fd-GOGAT from the higher plants maize and spinach (about 43%), the red alga Antithamnnion sp. (42%) or with the NADPH-GOGAT of bacterial source, such as Escherichia coli (41%) and Azospirillum brasilense (45%). The detailed analysis of Synechocystis gltB deduced amino acid sequence shows strongly conserved regions that have been assigned to the 3Fe-4S cluster (CX5CHX3C), the FMN-binding domain and the glutamine-amide transferase domain. Insertional inactivation of gltB and gltS genes revealed that both genes code for ferredoxin-dependent glutamate synthases which were nonessential for Synechocystis growth, as shown by the ferredoxin-dependent glutamate synthase activity and western-blot analysis of the mutant strains.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Anderson MP, Vance CP, Heichel GH, Miller SS: Purification and characterization of NADH-glutamate synthase from alfalfa root nodules. Plant Physiol 90: 351–358 (1989).
Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K: Current Protocols in Molecular Biology. Greene Publishing/Wiley-Interscience, New York (1992).
Avila C, Botella JR, Cánovas FM, Núñez de Castro I, Valpuesta V: Different characteristics of the two glutamate synthases in the green leaves of Lycopersicon esculentum. Plant Physiol 85: 1036–1039 (1987).
Avila C, Márquez AJ, Pajuelo P, Cannell ME, Wallsgrove RM, Forde BG: Cloning and sequence analysis of a cDNA for barley ferredoxin-dependent glutamate synthase and molecular analysis of photorespiratory mutants deficient in the enzyme. Planta 189: 475–483 (1993).
Benny AG, Boland MJ: Enzymes of nitrogen metabolism in legume nodules. Purification and properties of NADH-dependent glutamate synthase from lupin nodules. Eur J Biochem 79: 355–362 (1977).
Botella JR, Verbelen JP, Valpuesta V: Immunocytolocalization of ferredoxin-GOGAT in cells of green leaves and cotyledons of Lycopersicon esculentum: Plant Physiol 87: 255–257 (1988).
Boyer HW, Roulland-Dussoix D: A complementation analysis of the restriction and modification of DNA in Escherichia coli. J Mol Biol 41: 459–472 (1969).
Bradford MM: A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248–254 (1979).
Cai Y, Wolk CP: Use of a conditionally lethal gene in Anabaena sp. strain PCC 7120 to select for double recombinants and to entrap insertion sequences. J Bact 172: 3138–3145 (1990).
Chapman SK, White SA, Reid GA: Flavocytochrome b2. Adv Inorg Chem 36: 257–301 (1991).
Chauvat F, deVries L, van derEnde A, vanArkel GA: A host-vector system for gene cloning in the cyanobacterium Synechocystis PCC 6803. Mol Gen Genet 204: 185–191 (1986).
Chávez S: Glutamate dehydrogenase in cyanobacteria. Ph. D. thesis, University of Sevilla (1992).
Chen FL, Cullimore JV: Two isoenzymes of NADH-dependent glutamate synthase in root nodules of Phaseolus vulgaris. Plant Physiol 88: 1411–1417 (1988).
Chen FL, Cullimore JV: Localization of two isoenzymes of NADH-dependent glutamate synthase in root nodules of Phaseolus vulgaris. Planta 179: 441–447 (1989).
Devereux J, Haeberli P, Smithies O: A comprehensive set of sequence analysis programs for the VAX. Nucl Acids Res 12: 387–395 (1984).
Elhai J, Wolk CP: A versatile class of positive-selection vectors based on the nonviability of palindrome containing plasmids that allows cloning into long polylinkers. Gene 68: 119–138 (1988).
Florencio FJ, Marqués S, Candau P: Identification and characterization of glutamate dehydrogenase in the unicellular cyanobacterium Synechocystis PCC 6803. FEBS Lett 223: 37–41 (1987).
Galván F, Márquez AJ, Vega JM: Purification and molecular properties of ferredoxin-glutamate synthase from Chlamydomonas reinhardtii. Planta 162: 180–187 (1984).
Gregerson RG, Miller SS, Twary SN, Gantt JS, Vance CP: Molecular characterization of NADH-dependent glutamate synthase from alfalfa nodules. Plant Cell 5: 215–226 (1993).
Hayakawa T, Yamaya T, Kamachi K, Ojima K: Purification, characterization, and immunological properties of NADH-dependent glutamate synthase from rice cell cultures. Plant Physiol 98: 1317–1322 (1992).
Hemmilä IA, Mäntsälä PI: Purification and properties of glutamate synthase and glutamate dehydrogenase from Bacillus megaterium. Biochem J 173: 45–52 (1978).
Hirasawa M, Knaff DB: Ferredoxin-dependent chloroplast enzymes. Biochim Biophys Acta 1056: 93–125 (1991).
Hirasawa M, Robertson DE, Ameyibor E, Johnson MK, Knaff DB: Oxidation-reduction properties of the ferredoxin-linked glutamate synthase from spinach leaf. Biochim Biophys Acta 1100: 105–108 (1992).
Igeño MI, Caballero FJ, Castillo F: Molecular and kinetic characterization of glutamate synthase from the phototrophic bacterium Rhodobacter capsulatus E1F1. J Gen Microbiol 139: 2921–2929 (1993).
Johnson MK, Kowal AT, Morningstar JE, Oliver ME, Whittaker K, Gunsalus RP, Ackerll BAC, Cecchini G: Subunit location of the iron sulfur clusters in fumarate reductase from Escherichia coli. J Biol Chem 263: 14732–14738 (1989).
Karplus PA, Daniels MJ, Herriott JR: Atomic structure of ferredoxin-NADP+ reductase: prototype for a structurally novel flavoenzyme family. Science 251: 60–66 (1991).
Knaff DB, Hirasawa M, Ameyibor E, Fu W, Johnson MK: Spectroscopic evidence for a [3Fe-4S] cluster in spinach glutamate synthase. J Biol Chem 266: 15080–15084 (1991).
Laemmli UK: Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685 (1970).
Lea PJ, Miflin BJ: Alternative route for nitrogen assimilation in higher plants. Nature 251: 614–616 (1974).
Manodori A, Cecchini G, Schroder I, Gunsalus RP, Werth MT, Johson MK: [3Fe-4S] to [4Fe-4S] cluster conversion in Escherichia coli fumarate reductase by site-directed mutagenesis. Biochemistry 31: 2703–2712 (1992).
Marqués S, Florencio FJ, and Candau P: Purification and characterization of the ferredoxin-glutamate synthase from the unicellular cyanobacterium Synechococcus sp. PCC 6301. Eur J Biochem 206: 69–77 (1992).
Márquez AJ, Galván F, Vega JM: Purification and characterization of the NADH-glutamate synthase from Chlamydomonas reinhardtii. Plant Sci Lett 34: 305–314 (1984).
Mei B, Zalkin H: A cysteine-histidine-aspartate catalytic triad is involved in glutamine amide transfer function in purF-type glutamine amidotransferases. J Biol Chem 264: 16613–16619 (1989).
Mei B, Zalkin H: Amino-terminal deletions define a glutamine amide transfer domain in glutamine phosphoribosylpyrophosphate amidotransferase and other purF-type amidotransferases. J Bact 172: 3512–3514 (1990).
Mérida A, Flores E, Florencio FJ: Regulation of Anabaena sp. strain 7120 glutamine synthetase activity in a Synechocystis sp. strain PCC 6803 derivative strain bearing the Anabaena glnA gene and a mutated host glnA gene. J Bact 174: 6550–6554 (1992).
Miflin BJ, Lea PJ: Ammonia assimilation. In: Miflin BJ (ed) The Biochemistry of Plants, vol. 5, pp. 169–202. Academic Press, New York/London, (1980).
Miller RE, Stadtman ER: Glutamate synthase from Escherichia coli. J Biol Chem 247: 7407–7419 (1972).
Nalbantoglu B, Hirasawa M, Moomaw C, Nguyen H, Knaff DB, Allen R: Cloning and sequencing of the gene encoding spinach ferredoxin-dependent glutamate synthase. Biochim Biophys Acta 1183: 557–561 (1994).
Needleman SB, Wunsch CD: A general method applicable to the search for similarities in the amino acid sequence of two proteins. J Mol Biol 48: 443–453 (1970).
Oliver G, Gosset G, Sánchez-Pescador R, Lozoya E, Ku LM, Flores N, Becerril B, Valle F, Bolivar F: Determination of the nucleotide sequence for the glutamate synthase structural genes of Escherichia coli K-12. Gene 60: 1–11 (1987).
Pearson WR, Lipman DJ: Improved tools for biological sequence comparison. Proc Natl Acad Sci USA 85: 2444–2448 (1988).
Pelanda R, Vanoni MA, Perego M, Piubelli L, Galizzi A, Curti B, Zanetti G: Glutamate synthase genes of the diazotroph Azospirillum brasilense. J Biol Chem 268: 3099–3106 (1993).
Reith M, Munholland J: A high resolution gene map of the chloroplast genome of the red alga Porphyra purpurea. Plant Cell 5: 465–475 (1993).
Rippka R, Deruelles J, Waterbury JB, Herdman M, Stainer RY: Generic assignments, strain histories and properties of pure cultures of cyanobacteria. J Gen Microbiol 111: 1–61 (1979).
Sakakibara H, Watanabe M, Hase T, Sugiyama T: Molecular cloning and characterization of complementary DNA encoding for ferredoxin-dependent glutamate synthase in maize leaf. J Biol Chem 266: 2028–2035 (1991).
Sambrook J, Fritsch EF, Maniatis T: Molecular Cloning: A Laboratory Manual, 2nd ed. Cold Spring Harbor Laboratory Press, Cold Spring Harbor Laboratory, NY (1989).
Sanger F, Nicklen S, Coulson AR: DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA 74: 5463–5467 (1977).
Suzuki A, Gadal P: Glutamate synthase from rice leaves. Plant Physiol 69: 848–852 (1982).
Suzuki A, Gadal P: Glutamate synthase: physicochemical and functional properties of different forms in higher plants and other organisms. Physiol Vég 22: 471–486 (1984).
Suzuki A, Oaks A, Jacquot JP, Vidal J, Gadal P: An electron transport system in maize roots for reactions of glutamate synthase and nitrite reductase. Physiological and immunological properties of the electron carrier and pyridine nucleotide reductase. Plant Physiol 78: 374–378 (1984).
Trotta PP, Platzer KEB, Haschemeyer RH, Meister A: Glutamine binding subunits of glutamate synthase and partial reactions catalyzed by this glutamine amidotransferase. Proc Natl Acad Sci USA 71: 4607–4611 (1974).
Valentin K, Kostzewa M, Zetsche K: Glutamate synthase is plastid-encoded in a red alga: implications for the evolution of glutamate synthases. Plant Mol Biol 23: 77–85 (1993).
Vanoni MA, Accornero P, Carrera G, Curti B: The pH-dependent behavior of catalytic activities of Azospirillum brasilense glutamate synthase and iodoacetamide modification of the enzyme provide evidence for a catalytic Cys-His ion pair. Arch Biochem Biophys 309: 222–230 (1994).
Vanoni MA, Curti B, Zanetti G: Glutamate synthase. In: Muller F (ed) Chemistry and Biochemistry of Flayoproteins, vol III, pp. 309–317, Walter De Gruyter, Berlin (1991).
Wierenga RK, DeMaeyer MCH, Hol WGJ: Interaction of pyrophosphate moieties with α-helixes in dinucleotide binding protein. Biochemistry 24: 1346–1357 (1985).
Zehnacker C, Becker TW, Suzuki A, Carrayol E, Caboche M, Hirel B: Purification and properties of tobacco ferredoxin-depenent glutamate synthase, and isolation of corresponding cDNA clones. Light-inducibility and organ-specificity of gene transcription. Planta 187: 266–274 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Navarro, F., Chávez, S., Candau, P. et al. Existence of two ferredoxin-glutamate synthases in the cyanobacterium Synechocystis sp. PCC 6803. Isolation and insertional inactivation of gltB and gltS genes. Plant Mol Biol 27, 753–767 (1995). https://doi.org/10.1007/BF00020228
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00020228