Abstract
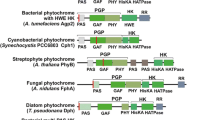
A cDNA clone encoding phytochrome (apoprotein) of the zygnematophycean green alga Mougeotia scalaris has been isolated and sequenced. The clone consisted of 3372 bp, encoded 1124 amino acids, and showed strain-specific nucleotide exchanges for M. scalaris, originating from different habitats. No indication was found of multiple phytochrome genes in Mougeotia. The 5′ non-coding region of the Mougeotia PHY cDNA harbours a striking stem-loop structure. Homologies with higher-plant phytochromes were 52–53% for PHYA and 57–59% for PHYB. Highest homology scores were found with lower-plant phytochromes, for example 67% for Selaginella (Lycopodiopsida), 64% for Physcomitrella (Bryopsida) and 73% for Mesotaenium (Zygnematophyceae). In an unrooted phylogenetic tree, the position of Mougeotia PHY appeared most distant to all other known PHYs. The amino acids Gly-Val in the chromophore-binding domain (-Arg-Gly-Val-His-Gly-Cys-) were characteristic of the zygnematophycean PHYs known to date. There was no indication of a transmembrane region in Mougeotia phytochrome in particular, but a carboxyl-terminal 16-mer three-fold repeat in both, Mougeotia and Mesotaenium PHYs may represent a microtubule-binding domain. Unexpected for a non-angiosperm phytochrome, its expression was autoregulated in Mougeotia in a red/far-red reversible manner: under Pr conditions, phytochrome mRNA levels were tenfold higher than under Pfr conditions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bonenberger J, Schendel R, Schneider-Poetsch HAW, Rüdiger W: Structural studies on the photoreceptor phytochrome: Reevaluation of the epitope for monoclonal antibody Z-3B1. Photochem Photobiol 56: 717–723 (1992).
Chou PY, Fasman GD: Prediction of the secondary structure of proteins from their amino acid sequence. Adv Enzymol 47: 45–148 (1978).
Cordonnier M-M: Monoclonal antibodies: molecular probes for the study of phytochrome. Photochem Photobiol 49: 821–831 (1989).
Cox KH, Goldberg RB: Analysis of plant gene expression. In: Shaw CH (ed) Plant Molecular Biology, pp. 2–8. IRL Press, Oxford (1988).
Danon A, Mayfield StP: ADP-dependent phosphorylation regulates RNA-binding in vitro: implications in light-modulated translation. EMBO J 13: 2227–2235 (1994).
Feinberg AB, Vogelstein B: A technique for radiolabelling DNA restriction endonuclease fragments to high specific activity. Anal Biochem 137: 6–13 (1983).
Feng DF, Doolittle RF: Progressive alignment and phylogenetic tree construction of protein sequences. In: Doolittle RF (ed) Molecular Evolution: Computer Analysis of Protein and Nucleic Acid Sequences. Methods in Enzymology, vol 183, pp. 375–383. Academic Press, San Diego, CA (1990).
Gibbs RA, Nguyen P-N, Caskey CT: Detection of single DNA base differences by competitive oligonucleotide priming. Nucl Acids Res. 17: 2437–2448 (1989).
Hanstein Ch, Grolig F, Wagner G: Immunolocalization of cytosolic phytochrome in the green alga Mougeotia. Bot Acta 105: 55–62 (1992).
Haupt W: Die Inversion der Schwachlichtbewegung des Mougeotia-chloroplasten: Versuche zur Kinetik der Phytochromumwandlung. Z Pflanzenphysiol 54: 151–160 (1966).
Haupt W: Light-mediated movement of chloroplasts. Annu Rev Plant Physiol 33: 205–233 (1982).
Haupt W, Weisenseel MH: Physiological evidence and some thoughts on localized responses, intracellular localization and action of phytochrome. In: Smith H (ed) Light and Plant Development, pp. 63–74. Butterworth, London (1976).
Hendricks SB, Borthwick HA: The function of phytochrome in regulation of plant growth. Proc Natl Acad Sci USA 58: 2125–2130 (1967).
Kidd DG, Lagarias JC: Phytochrome from the green alga Mesotaenium caldariorum. Purification and preliminary characterization. J Biol Chem 265: 7029–7035 (1990).
Kolukisaoglu HÜ, Marx St, Wiegmann C, Hanelt S, Schneider-Poetsch HAW: Divergence of the phytochrome family predates angiosperm evolution and suggests that Selaginella and Equisetum arose prior to Psilotum. J Mol Evol 41: 329–337 (1995).
Kraml M: Light direction and polarization. In: Kendrick RE, Kronenberg GHM (eds) Photomorphogenesis in Plants, vol 2, pp. 417–445. Kluwer Academic Publishers, Dordrecht, Netherlands (1993).
Kyte J, Doolittle RF: A simple method for displaying the hydropathic character of a protein. J Mol Biol 557: 105–132 (1982).
Lagarias DM, Wu S-H, Lagarias JC: Atypical phytochrome gene structure in the green alga Mesotaenium caldariorum. Plant Mol Biol 29: 1127–1142 (1995).
Morand LZ, Kidd DG, Lagarias JC: Phytochrome levels in the green alga Mesotaenium caldariorum are light regulated. Plant Physiol 101: 97–103 (1993).
Okamoto H, Hirano Y, Abe H, Tomozawa K, Furuya M, Wada M: The deduced amino acid sequence of phytochrome from Adiantum includes consensus motifs present in phytochrome B from seed plants. Plant Cell Physiol 34: 1329–1334 (1993).
Quail PH, Boylan MT, Parks BM, Short TW, Xu Y, Wagner D: Phytochromes: photosensory perception and signal transduction. Science 268: 675–680 (1995).
Russ U, Grolig F, Wagner G: Differentially adsorbed vital dyes inhibit chloroplast movement in Mougeotia scalaris. Protoplasma Suppl 1: 180–184 (1988).
Samaniego F, Chin J, Iwai K, Rouault TA, Klausner RD: Molecular characterization of a second iron-responsive element binding protein, iron regulatory protein 2: structure, function and post-translational regulation. J Biol Chem 269: 30904–30910 (1994).
Sambrook J, Fritsch EF, Maniatis T: Molecular Cloning: A Laboratory Manual, 2nd ed. Cold Spring Habor Laboratory Press, Cold Spring Harbor, NY (1989).
Sanger F, Nicklen S, Coulson AR: DNA sequencing with chainterminating inhibitors. Proc Natl Acad Sci USA 74: 5463 (1977).
Schlösser UG: SAG: Sammlung von Algenkulturen at the University of Göttingen: Catalogue of Strains 1994. Bot Acta 107: 113–186 (1994).
Schneider-Poetsch HAW: Signal transduction by phytochrome: Phytochromes have a module related to the transmitter modules of bacterial sensor proteins. Photochem Photobiol 56: 839–846 (1992).
Schneider-Poetsch HAW, Marx St, Kolukisaoglu HÜ, Hanelt S, Braun B: Phytochrome evolution: phytochrome genes in ferns and mosses. Physiol Plant 91: 241–250 (1994).
Smith H: Sensing the light environment. The functions of the phytochrome family. In: Kendrick RE, Kronenberg GHM (eds) Photomorphogenesis in Plants, vol 2, pp. 377–416. Kluwer Academic Publishers, Dordrecht, Netherlands (1993).
Stabenau H: Wachstum von Mougeotia in der Durchlüftungskultur. Ber Deut Bot Ges 91: 251–255 (1978).
Tretyn A, Kendrick RE, Wagner G: The role(s) of calcium ions in phytochrome action. Photochem Photobiol 54: 1135–1155 (1991).
Wada M, Grolig F, Haupt W: Light-oriented chloroplast positioning. Contribution to progress in photobiology. J Photochem Photobiol B: Biology 17: 3–25 (1993).
Wagner G: Intracellular movement. Progr Bot 57: 68–80 (1995).
Wiche G, Oberkanins Ch, Himmler A: Molecular structure and function of microtubule-associated proteins. Int Rev Cytol 124: 217–273 (1991).
Winands A, Wagner G, Marx St, Schneider-Poetsch HAW: Partial nucleotide sequence of phytochrome from the zygnematophycean green alga Mougeotia. Photochem Photobiol 56: 765–770 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Winands, A., Wagner, G. Phytochrome of the green alga Mougeotia: cDNA sequence, autoregulation and phylogenetic position. Plant Mol Biol 32, 589–597 (1996). https://doi.org/10.1007/BF00020200
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00020200