Abstract
In potato tubers two starch phosphorylase isozymes, types L and H, have been described and are believed to be responsible for the complete starch breakdown in this tissue. Type L has been localized in amyloplasts, whereas type H is located within the cytosol. In order to investigate whether the same isozymes are also present in potato leaf tissue a cDNA expression library from potato leaves was screened using a monoclonal antibody recognizing both isozyme forms. Besides the already described tuber L-type isozyme a cDNA clone encoding a second L-type isozyme was isolated. The 3171 nucleotide long cDNA clone contains an uninterrupted open reading frame of 2922 nucleotides which encodes a polypeptide of 974 amino acids. Sequence comparison between both L-type isozymes on the amino acid level showed that the polypeptides are highly homologous to each other, reaching 81–84% identity over most parts of the polypeptide. However the regions containing the transit peptide (amino acids 1–81) and the insertion sequence (amino acids 463–570) are highly diverse, reaching identities of only 22.0% and 29.0% respectively.
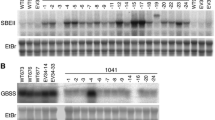
Northern analysis revealed that both forms are differentially expressed. The steady-state mRNA levels of the tuber L-type isozyme accumulates strongly in potato tubers and only weakly in leaf tissues, whereas the mRNA of the leaf L-type isozyme accumulates in both tissues to the same extent. Constitutive expression of an antisense RNA specific for the leaf L-type gene resulted in a strong reduction of starch phosphorylase L-type activity in leaf tissue, but had only sparse effects in potato tuber tissues. Determination of the leaf starch content revealed that antisense repression of the starch phosphorylase activity has no significant influence on starch accumulation in leaves of transgenic potato plants. This result indicated that different L-type genes are responsible for the starch phosphorylase activity in different tissues, but the function of the different enzymes remains unclear.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Amasino RM: Acceleration of nucleic acid hybridisation rate by polyethylene glycol. Anal Biochem 152: 304–307 (1986).
Bevan M: binary Agrobacterium vectors for plant transformation. Nucl Acids Res 12: 8711–8721 (1984).
Bradford MM: A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 243–254 (1976).
Brisson N, Giroux H, Zollinger M, Camirand A, Simard C: Maturation and subcellular compartmentation of potato starch phosphorylase. Plant Cell 1: 559–566 (1989).
Bullock WO, Fernandez JM, Short JM: XL-1 Blue: a high efficiency plasmid transforming recA Escherichia coli strain with beta-galactosidase selection. BioTechniques 5: 376 (1987).
Conrads J, van Berkel J, Schächtele C, Steup M: Nonchloroplast α-1,4-glucan phosphorylase from pea leaves: characterization and in-situ localization by indirect immunofluorescence. Biochim Biophys Acta 882: 452–463 (1986).
Debleare R, Bytebier B, DeGreve H, Debroek F, Schell J, Van Montagu M, Leemans J: Efficient octopine Ti plasmid-derived vectors of Agrobacterium mediated gene transfer to plants. Nucl Acids Res 13: 4777–4788 (1985).
Dickinson DB, Preiss J: Presence of ADP-glucose pyrophoshorylase in shrunken-2 and brittle-2 mutants of maize endosperm. Plant Physiol 44: 1058–1062 (1969).
Franck A, Guilley H, Jonrad G, Richards K, Hirth L: Nucleotide sequence of cauliflower mosaic virus DNA. Cell 21: 285–294 (1980).
Gielen J, De Beuckeleer M, Seurinck J, Debroeck H, DeGreve H, Lemmers M, Van Montagu M, Schell J: The complete nucleotide sequence of the TL-DNA of the Agrobacterium tumefaciens plasmid pTiAch5. EMBO J 3: 835–846 (1984).
Greve B: Immunochemische Analyse und submolekulare Charakterisierung pflanzlicher Heteropolymere mit Hilfe mono- und polyklonaler Antikörper. Dissertation, Universität Münster (1992).
Höfgen R, Willmitzer L: Storage of competent cells for Agrobacterium transformation. Nucl Acids Res 16: 9877 (1988).
Kossmann J, Müller-Röber B, Dyer TA, Raines CA, Sonnewald U, Willmitzer L: Cloning and expression analysis of plastidic fructose-1,6-bisphosphatase coding sequence from potato: circumstantial evidence for the import of hexoses into chloroplasts. Planta 188: 7–12 (1992).
Lin T-P, Caspar T, Sommerville C, Preiss J: Isolation and characterization of a starchless mutant of Arabidopsis thaliana (L.) Heynh lacking ADPglucose phosphorylase activity. Plant Physiol 86: 1131–1135 (1988).
Lin T-P, Caspar T, Sommerville C, Preiss J: A starch deficient mutant of Arabidopsis thaliana with low ADP-glucose pyrophosphorylase activity lacks one of the two subunits of the enzyme. Plant Physiol 88: 1175–1181 (1988).
Lin C-T, Yeh K-W, Lee PD, Su JC: Primary structure of sweet potato starch phosphorylase deduced from its cDNA sequence. Plant Physiol 95: 1250–1253 (1991).
Logemann J, Schell J, Willmitzer L: Improved method for the isolation of RNA from plant tissues. Anal Biochem 163: 21–26 (1987).
Maniatis T, Fritsch EF, Sambrook J: Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY (1982).
Mori H, Tanizawa K, Fukui T: Potato tuber type H phosphorylase isozyme. J Biochem 266: 18446–18453 (1991).
Müller-Röber B, Sonnewald U, Willmitzer L: Inhibition of the ADP-glucose pyrophosphorylase in transgenic potatoes leads to sugar-storing tubers and influences tuber formation and expression of tuber storage protein genes. EMBO J 11: 1229–1238 (1992).
Murashige T, Skoog F: A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15: 473–497 (1962).
Nakano K, Mori H, Fukui T: Molecular cloning of cDNA encoding potato amyloplast α-glucan phosphorylase and the structure of its transit peptide. J Biochem (Tokyo) 106: 691–695 (1989).
Preiss J: Regulation of the biosynthesis and degradation of starch. Annu Rev Plant Physiol 33: 431–454 (1982).
Proudfoot NJ, Brownlee GG: 3′ Non-coding region sequences in eukaryotic messenger RNA. Nature 263: 211–214 (1976).
Proudfoot NJ: The end of message and beyond. Nature 307: 412–413 (1984).
Shimomura S, Nagai M, Fukui T: Comparative glucan specificities of two types of spinach leaf phosphorylase. J Biochem 91: 703–717 (1982).
Rocha-Sosa M, Sonnewald U, Frommer W, Stratmann M, Schell J, Willmitzer L: Both developmental and metabolic signals activate the promoter of a class I patatin gene. EMBO J 8: 23–29 (1989).
Steup M, Latzko E: Intracellular localization of phosphorylases in spinach and pea leaves. Planta 145: 69–75 (1979).
Steup M: Starch degrading enzymes. Meth Plant Biochem 3: 103–128 (1990).
Stitt M, Wirtz W, Heldt HW: Pathway of starch break-down in photosynthetic tissue of Pisum sativum. Biochim Biophys Acta 544: 200–214 (1978).
Tsai CY, Nelson OE: Starch deficient maize mutant lacking adenosine diphosphate glucose pyrophosphorylase activity. Science 151: 341–343 (1966).
Vervliet G, Holsters M, Teuchy H, Van Montagu M, Schell J: Characterization of different plaque-forming and defective temperate phages in Agrobacterium strains. J Gen Virol 26: 33–48 (1975).
Yang Y, Steup M: Polysaccharide fraction from higher plants which strongly interacts with the cytosolic phosphorylase isozyme. Plant Physiol 94: 960–969 (1990).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sonnewald, U., Basner, A., Greve, B. et al. A second L-type isozyme of potato glucan phosphorylase: cloning, antisense inhibition and expression analysis. Plant Mol Biol 27, 567–576 (1995). https://doi.org/10.1007/BF00019322
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00019322