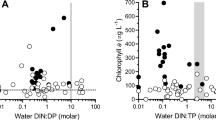
Abstract
Five treatments (replication n=2) were applied to mesocosms in an oligotrophic lake (TP=6–10 µg 1∼-1) to assess the effects of fish on planktonic communities. The treatments were: (1) high fish (30 kg ha−1 Lepomis auritus, Linnaeus), (2) low fish (10 kg ha−1), (3) high removal of zooplankton, (4) low removal of zooplankton and (5) control. Total phosphorus, chlorophyll a, zooplankton biomass, and species richness decreased from high fish > low fish > control > low removal > high removal treatments. The fish treatments were dominated by crustacean zooplankton, while rotifers outnumbered the other zooplankters in the removal treatments. Calculations of zooplankton grazing rates suggested that clearance rates seldom exceeded 2% of the enclosure volume d−1 and were unlikely to have had much influence on phytoplankton biomass. Calculations from a phosphorus bioenergetics model revealed that when fish were present, their excretion rates were higher than the rates ascribed to zooplankton. Diet analysis showed that the fish derived most of their energy from the benthos and periphyton, and that fish excretion and egestion made significant contributions to the very oligotrophic pelagic phosphorus pool. In the absence of fish, zooplankton excretion was highest in the control treatments and lowest in the zooplankton removal treatments. Our results suggest that in oligotrophic systems, planktivorous fish can be significant sources of phosphorus and that fish and zooplankton induced nutrient cycling have significant impacts on planktonic community structure.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
APRA (American Public Health Association), 1985. Standard methods for the examination of water and waste water, 16th edn. APHA, AWWA, WPCF Washington, DC.
Balcer, M. D., N. L. Korda & S. I. Dodson, 1984. Zooplankton of the Great Lakes: a guide to the identification and ecology of the common crustacean species. University of Wisconsin Press, Madison.
Bogdan, K. G., J. G. Gilbert & P. L. Starkweather, 1980. In situ clearance rates of planktonic rotifers. Hydrobiologia 73: 73–77.
Brooks, J. L. & S. I. Dodson, 1965. Predation, body size, and composition of zooplankton. Science 150: 28–35.
Carpenter, S. R. (ed.), 1988. Complex interactions in lake communities. Springer-Verlag, New York.
Carpenter, S. R. & J. F. Kitchell (ededs), 1993. The trophic cascade in lakes. Cambridge.
Carpenter, S. R., J. F. Kitchell & J. R. Hodgson, 1985. Cascading trophic interactions and lake productivity. BioScience 35: 634–638.
Carpenter, S. R., J. F. Kitchell, J. R. Hodgson, P. A. Cochran, J. J. Elser, M. M. Elser, D. M. Lodge, D. K. Kretchmer, X. He & C. N. von Ende, 1987. Regulation of lake primary productivity by food web structure. Ecology 68: 1863–1876.
Chow-Fraser, P, 1986. An empirical model to predict in situ grazing rates of Leptodiaptomus minutus Lilljeborg on small algal particles. Can. J. Fish. aquat. Sci. 43: 1065–1070.
Chow-Fraser, P. & R. Knoechel, 1985. Factors regulating in situ filtering rates of Cladocera. Can. J. Fish. aquat. Sci. 42: 567–576.
Edmondson, W. T. (ed.), 1959. Freshwater biology, 2nd edn. Wiley, New York.
Elliot, J. M. & L. Persson, 1978. The estimation of daily rates of food consumption for fish. J. anim. Ecol. 47: 977–991.
Hamilton, D. T. & W. D. Taylor, 1987. Short-term effects of zooplankton manipulations on phosphate uptaKe. Can. J. Fish. aquat. Sci. 44: 1038–1044.
Heady, H. F., 1942. Littoral vegetation on the lakes of the Huntington Forest. Roosevelt Wildlife Bull. 8. 33 pp.
Hewett, S. W. & B. L. Johnson, 1992. Fish bioenergetics model 2: an upgrade of a generalized bioenergetics model of fish growth for microcomputers. University of Wisconsin Sea Grant Institute, Madison.
Kerfoot, W. C. & A. Sih (eds), 1987. Predation: direct and indirect impacts on aquatic communities. University Press of New England, Hanover. 386 pp.
Kitchell, J. F., J. F. Koonce, R. V. O'Neill, H. H. Shugart Jr., J. J. Magnuson & R. S. Booth, 1974. Model of fish Biomass dynamics. Trans. am. Fish. Soc. 103: 786–798.
Knoechel, R. & L. B. Holtby, 1986. Construction and validation of a body-length-based model for the prediction of cladoceran community filtering rates. Limnol. Oceanogr. 31: 1–16.
Kraft, C. E., 1992. Estimates of phosphorus cycling by fish using a bioenergetics approach. Can. J. Fish. aquat. Sci. 49: 1–9.
Lehman, J. T., 1980. Release and cycling of nutrients between planktonic algae and herbivores. Limnol. Oceanogr. 25: 620–632.
Lorenzen, C. J., 1967. Determination of chlorophyll and pheopigments: spectrophotometric equations. Limnol. Oceanogr. 12: 343–346.
Lynch, M. & J. Shapiro, 1981. Predation, enrichment, and phytoplankton community structure. Limnol. Oceanogr. 26: 86–102.
Mazumder, A., D. J. McQueen, W. D. Taylor & D. R. S. Lean, 1988. Effects of fertilization and planktivorous fish (yellow perch) predation on size distribution of particulate phosphorus and assimilated phosphate: Large enclosure experiments. Limnol. Oceanogr. 33: 421–430.
McCauley, E. & F. Briand, 1979. Zooplankton grazing and phytoplankton species richness: field tests of the predation hypothesis. Limnol. Oceanogr. 24: 243–252.
McCauley, E. & J. Kalff, 1987. Effect of changes in zooplankton on orthophosphate dynamics of natural phytoplankton communities. Can. J. Fish. aquat. Sci. 44: 176–182.
McQueen, D. J., R. France & C. E. Kraft, 1992. Confounded impacts of planktivorous fish on freshwater biomanipulations. Arch. Hydrobiol. 125: 1–24.
McQueen, D. J., J. R. Post & E. L. Mills, 1986. Trophic interactions in freshwater pelagic ecosystems. Can. J. Fish. aquat. Sci. 43: 1571–1581.
Meijer, M. L. M. W. de Haan, A. W. Breukelaar & H. Buiteveld, 1990. Is reduction of the benthivorous fish an important cause of high transparency following biomanipulation in shallow lakes? Hydrobiologia 200/201 (Dev. Hydrobiol. 61): 303–315.
Meredith, M. P. & S. V. Stehman, 1991. Repeated measures experiments in forestry: focus on analysis of response curves. Can. J. For. Res. 21: 957–965.
Mills, E. L. & J. L. Confer, 1986. Computer processing of zooplankton/application in fisheries. Fisheries 11: 24–27.
Nakashima, B. S. & W. C. Legget, 1980. The role of fishes in the regulation of phosphorus availability in lakes. Can. J. Fish. aquat. Sci. 37: 1540–1549.
Pace, M. L. & E. Funke, 1991. Regulation of planktonic microbial communities by nutrients and herbivores. Ecology 72: 904–914.
Pace, M. L., G. B. McManus & S. E. G. Findlay, 1990. Planktonic community structure determines the fate of bacterial production in a temperate lake. Limnol. Oceanogr. 35: 795–808.
Pennak, R. W., 1989. Fresh-water invertebrates of the United States. Protozoa to Mollusca, 3rd edn. Wiley, New York.
Persson, L., G. Andersson, S. F. Hamrin & L. Johansson, 1988. Predator regulation and primary production along the productivity gradient of temperate lake ecosystems. In S. R. Carpenter (ed.), Complex interactions in lake communities, Springer-Verlag, New York: 45–46.
Peters, R. H. & J. A. Downing, 1984. Empirical analysis of zooplankton filtering and feeding rates. Limnol. Oceanogr. 29: 763–778.
Reynolds, C. S., 1984. The ecology of freshwater phytoplankton. Cambridge, 384 pp.
SAS Institute Inc., 1990. SAS user's guide: Statistics. SAS Institute, Cary, N.C.
Scavia, D. & G. A. Laird, 1987. Bacterioplankton in Lake Michigan: Dynamics, controls, and significance to carbon flux. Limnol. Oceanogr. 32: 1017–1032.
Scavia, D., D. A. Laird & G. L. Fahnenstiel, 1986. Production of planktonic bacteria in Lake Michigan. Limnol. Oceanogr. 31: 612–626.
Seaburg, K. G. & J. B. Moyle, 1964. Feeding habits of the warmwater fishes. Trans. am. Fish. Soc. 93: 280–285.
Shapiro, J. & D. I. Wright, 1984. Lake restoration by biomanipulation. Round Lake, Minnesota: the first two years. Freshwat. Biol. 14: 371–383.
Simberloff, D., 1972. Properties of the rarefaction diversity measurement. Am. Nat. 106: 414–418.
Taylor, W. D. & D. R. S. Lean, 1991. Phosphorus pool sizes and fluxes in the epilimnion of a mesotrophic lake. Can. J. Fish. aquat. Sci. 48: 1293–1301.
Vanni, M. J., 1987. Indirect effect of predators on age-structured prey populations: planktivorous fish and zooplankton. In W. C. Kerfoot & A. Sih (eds), Predation: direct and indirect impacts on aquatic communities, University Press of New England, Hanover: 149–160.
Vanni, M. J. & D. L. Findlay, 1990. Trophic cascades and phytoplankton community structure. Ecology 71: 921–937.
Wetzel, R. G. & G. E. Likens, 1991. Limnological analyses, 2nd edn. Springer-Verlag, New York.
Zaret, T. M., 1980. Predation and freshwater communities. Yale University Press, New Haven.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pérez-Fuentetaja, A., McQueen, D.J. & Ramcharan, C.W. Predator-induced bottom-up effects in oligotrophic systems. Hydrobiologia 317, 163–176 (1996). https://doi.org/10.1007/BF00018739
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00018739