Abstract
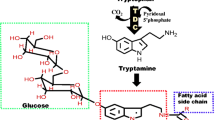
The purification of tryptophan decarboxylase from Catharanthus roseus (TDC, E.C.:4.1.1.27), to apparent homogeneity, is described. The enzyme represents a soluble protein with a molecular weight of 115 000±3 000, consisting of 2 identical subunits of 54 000±1 000. The pI was estimated to be 5.9 and the Km for L-tryptophan was found to be 7.5×10-5 M. Phenylalanine, tyrosine and DOPA were not decarboxylated by tryptophan decarboxylase from Catharanthus cells. Similar to the aromatic amino acid decarboxylase from hog kidney the enzyme does not appear to be obligatorily dependent on exogenously supplied pyridoxal phosphate, as it seems to contain a certain amount of this cofactor. The average percentage of TDC in the cells was found to be 0.002% in the growth medium while the level increased up to 0.03% when indole alkaloid biosynthesis was induced. The role of the protein as a bottleneck enzyme of indole alkaloid biosynthesis is discussed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adams E: Fluorometric determination of pyridoxal phosphate in enzymes. Anal Biochem 31: 118–122, 1969.
Baxter C, Slaytor M: Partial purification and some properties of tryptophan decarboxylase from Phalaris tuberosa. Phytochemistry 11: 2763–2766, 1972.
Berlin J, Forche E, Wray V, Hammer J, Hösel W: Formation of benzophenanthridine alkaloids by suspension cultures of Eschscholtzia california. Z Naturforsch 38c: 346–352, 1983.
Berlin J, Witte L, Hammer J, Kukoschke KG, Zimmer A, Pape D: Metabolism of p-fluorophenylalanine in p-fluorophenylalanine sensitive and resistant tobacco cell cultures. Planta 155:244–250, 1982.
Berlin J, Witte L: Metabolism of phenylalanine and cinnamic acid in tobacco cell cultures with high and low yields of cinnamoyl putrescines. J Natl Prod (Lloydia) 45: 88–93, 1982.
Bradford M: A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of proteindye binding. Anal Biochem 72: 2488, 1976.
Christenson JG, Dairman W, Udenfriend S: Preparation and properties of a homogenous aromatic L-amino acid decarboxylase from hog kidney. Arch Biochem Biophys 141:356–367, 1970.
Clark WC, Pass PS, Venkataraman B, Hodgetts RB: DOPA decarboxylase from Drosophila melanogaster. Purification, characterization and an analysis of mutants. Molec Gen Genet 162:287–297, 1978.
Gibson RA, Barret G, Wightman F: Biosynthesis and metabolism of indol-3yl-acetic acid: III. Partial purification and properties of a tryptamine-forming L-tryptophan decarboxylase from tomato shoots. J Exp Bot 23: 775–786, 1972.
Grosse W, Klapheck S: Enzymic studies on the biosynthesis of serotonin and the regulation in walnuts (Juglans regia L.). Z Pflanzenphysiol 93: 359–363, 1979.
Heller W, Egin-Bühler B, Gardiner S, Knobloch K-H, Matern U, Ebel J, Hahlbrock K: Enzymes of general phenylpropanoid metabolism and of flavonoid glycoside biosynthesis in parsley. Plant Physiol 64: 371–373, 1979.
Knobloch K-H, Berlin J: Influence of medium composition on the formation of secondary compounds in cell suspension cultures of Catharanthus roseus G. Don. Z Naturforsch 35c:551–556, 1980.
Knobloch K-H, Hansen B, Berlin J: Medium-induced formation of indole alkaloids and concomitant changes of interrelated enzyme activities in cell suspension cultures of Catharanthus roseus. Z Naturforsch 36c:40–43, 1981.
Laemmli UK: Cleavage of structural proteins during the assembly of the head of bacteriophage T 4. Nature 227:680–685, 1970.
Maizel JV: Polyacrylamide gel electrophoresis of viral proteins. Methods of Virol 5: 179–246, 1971.
Maurer HR: Disk-Elektrophorese. Verlag de Gruyter Berlin, 1968.
Merrill CR, Goldman D, Sedman SA, Ebert MH: Ultrasensitive stain for proteins in polyacrylamide gels shows regional variation in cerebrospinal fluid proteins. Science 211:1437–1438, 1981.
Sasse F, Buchholz M, Berlin J: Site of action of growth inhibitory tryptophan in analogues in Catharanthus roseus cell suspension cultures. Z Naturforsch, 1983 (in press).
Sasse F, Heckenberg U, Berlin J: Accumulation of β-carboline alkaloids and serotonin by cell cultures of Peganum harmala: II. Interrelationship between accumulation of serotonin and activities of related enzymes. Z Pflanzenphysiol 15:315–322, 1982a.
Scott AJ, Lee S-L: Biosynthesis of the indole alkaloids. A cell free system from Catharanthus roseus. J Amer Chem Soc 97:6906–6908, 1975.
Selby C, Turnbull A, Collin HA: Comparison of the onion plant (Allium cepa) and onion tissue cultures. II. Stimulation of flavour precursor synthesis in onion tissue cultures. New Phytol 84:307–312, 1980.
Waller GR, Dermer OC: Enzymology of alkaloids metabolism in plants and microorganisms. In: Conn EE (ed) The biochemistry of plants, Vol 7: Secondary plant products. Academic Press, New York, 1981, pp 317–373.
Wehrli A, Kovats E: Gas-chromatographische Charakterisierung organischer Verbindungen, Teil 3: Berechnung der Retentions-indices aliphatischer, alicyclischer und aromatischer Verbindungen 1/2. Helv Chim Acta 42:2709–2736, 1959.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Noé, W., Mollenschott, C. & Berlin, J. Tryptophan decarboxylase from Catharanthus roseus cell suspension cultures: purification, molecular and kinetic data of the homogenous protein. Plant Mol Biol 3, 281–288 (1984). https://doi.org/10.1007/BF00017782
Issue Date:
DOI: https://doi.org/10.1007/BF00017782