Abstract
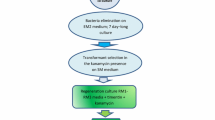
Regeneration of flax (Linum usitatissimum) following transformation by either Agrobacterium tumefaciens carrying a disarmed Ti-plasmid vector, or Agrobacterium rhizogenes carrying an unmodified Ri plasmid, was examined. Hypocotyl and cotyledon explants inoculated with A. tumefaciens formed transformed callus, but did not regenerate transformed shoots either directly or via callus. However, cotyledon explants inoculated with A. rhizogenes formed transformed roots which did regenerate transformed shoots. Ri T-DNA encoded opines were detected in the transformed plantlets and Southern hybridization analysis confirmed the presence of T-DNA from the Ri plasmid in their DNA. Transformed plantlets had curled leaves, short internodes and some had a more developed root system characterized by plagiotropic behaviour.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Barakat MN, Cocking EC: Plant regeneration from protoplast-derived tissues of Linum usitatissimum L. (flax). Plant Cell Rep 2: 314–317 (1983).
Basiran N, Armitage P, Scott RJ, Draper J: Genetic transformation of flax (Linum usitatissimum) by Agrobacterium tumefaciens: Regeneration of transformed shoots via a callus phase. Plant Cell Rep 6: 396–399 (1987).
Bolivar F, Rodriguez RL, Greene PJ, Betlach MC, Heyneker HL, Boyer HW, Croser JH, Falkow S: Construction and characterization of new cloning vehicles. II. A multipurpose cloning system. Gene 2: 95–113 (1977).
Chilton M-D, Drummond MH, Merlo DJ, Sciaky D, Montoya AL, Gordon MP, Nester EW: Stable incorporation of plasmid DNA into higher plant cells: The molecular basis of crown-gall tumorigenesis. Cell 11: 263–271 (1977).
Chilton M-D, Tepfer DA, Petit A, David C, Casse-Delbart F, Tempé J: Agrobacterium rhizogenes inserts T-DNA into the genome of the host root cells. Nature 295: 432–434 (1982).
Firoozabady E, DeBoer DL, Merlo DJ, Halk EL, Amerson LN, Rashka KE, Murray EE: Transformation of cotton (Gossypium hirsutum L.) by Agrobacterium tumefaciens and regeneration of transgenic plants. Plant Mol Biol 10: 105–116 (1987).
Flor HH: New physiologic races of flax rust. J Agric Res 60: 575–591 (1940).
Flor HH: Identification of races of flax rust by lines with single rust-conditioning genes. US Dept Agric Tech Bull 1087 (1954).
Flor HH: The complementary genic systems in flax and flax rust. Adv Genet 8: 29–54 (1956).
Gamborg OL, Shyluk JP: Tissue culture, protoplasts, and morphogenesis in flax. Bot Gaz 137: 301–306 (1976).
Green AG, Marshall DR: Isolation of induced mutants in linseed (Linum usitatissimum) having reduced linolenic acid content. Euphytica 33: 321–328 (1984).
Hain R, Stabel P, Czernilofsky AP, Steinbiß HH, Herrera-Estrella L, Schell J: Uptake, integration, expression and genetic transmission of a selectable chimaeric gene by plant protoplasts. Mol Gen Genet 199: 161–168 (1985).
Hepburn AG, Clarke LE, Pearson L, White J: The role of cytosine methylation in the control of nopaline synthase gene expression in a plant tumor. J Mol Appl Genet 2: 315–329 (1983).
Jouanin L: Restriction map of an agropine-type Ri plasmid and its homologies with Ti plasmids. Plasmid 12: 91–102 (1984).
Klee H, Horsch R, Rogers S: Agrobacterium-mediated plant transformation and its further application to plant biology. Ann Rev Plant Physiol 38: 467–486 (1987).
Link GKK, Eggers V: Mode, site, and time of initiation of hypocotyledonary bud primordia in Linum usitatissimum L. Bot Gaz 107: 441–454 (1946).
Maniatis T, Fritsch EF, Sambrook J: Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY (1982).
Mathews VH, Narayanaswamy S: Phytohormone control of regeneration in cultured tissues of flax. Z. Pflanzenphysiol 80: 436–442 (1976).
Miller JH: Experiments in Molecular Genetics. Cold Spring Harbor Laboratory. Cold Spring Harbor, NY (1972).
Murashige T, Skoog F: A revised medium for rapid growth and bioassay with tobacco tissue cultures. Physiol Plant 15: 473–497 (1962).
Petit A, David C, Dahl GA, Ellis JG, Guyon P, Casse-Delbart F, Tempé J: Further extention of the opine concept: Plasmids in Agrobacterium rhizogenes cooperate for opine degradation. Mol Gen Genet 190: 203–214 (1983).
Pomponi M, Spanò L, Sabbadini MG, Costantino P: Restriction endonuclease mapping of the root-inducing plasmid of Agrobacterium rhizogenes 1855. Plasmid 10: 119–129 (1983).
Pouwels PH, Enger-Valk BE, Brammar WJ: Cloning vectors: a laboratory manual. Elsevier, Amsterdam (1985).
Rogers SO, Bendich AJ: Extraction of DNA from milligram amounts of fresh, herbarium and mummified plant tissues. Plant Mol Biol 5: 69–76 (1985).
Rybczynski JJ: Callus formation and organogenesis of mature cotyledons of Linum usitatissimum L. var. Szokijskij in vitro culture. Genet Polon 16: 161–165 (1975).
Ryder MH, Tate ME, Kerr A: Virulence properties of strains of Agrobacterium on the apical and basal surfaces of carrot root discs. Plant Physiol 77: 215–221 (1985).
Spanò L, Pomponi M, Constantino P, Van Slogteren GMS, Tempé J: Identification of T-DNA in the root-inducing plasmid of the agropine type Agrobacterium rhizogenes 1855. Plant Mol Biol 1: 291–300 (1982).
Spanò L, Mariotti D, Pezzotti M, Damiani F, Arcioni S: Hairy root transformation in alfalfa (Medicago sativa L.) Theor Appl Genet 73: 523–530 (1982).
Stachel SE, Messens E, Van Montagu M, Zambryski P: Identification of signal molecules produced by wounded plant cells that activate T-DNA transfer in Agrobacterium tumefaciens. Nature 318: 624–629 (1985).
Tepfer D: Transformation of several species of higher plants by Agrobacterium rhizogenes: sexual transmission of the transformed genotype and phenotype. Cell 37: 959–967 (1984).
Tepfer M, Casse-Delbart F: Agrobacterium rhizogenes as a vector for transforming higher plants. Microbiol Sci 4: 24–28 (1987).
White FF, Ghidossi G, Gordon MP, Nester EW: Tumor induction by Agrobacterium rhizogenes involves the transfer of plasmid DNA to the plant genome. Proc Natl Acad Sci USA 79: 3193–3197 (1982).
White FF, Nester EW: Hairy root: Plasmid encodes virulence traits in Agrobacterium rhizogenes. J Bacteriol 141: 1134–1141 (1980).
Yamada S, Itano HA: Phenanthrenequinone as an analytical reagent for arginine and other monosubstituted guanidines. Biochim Biophys Acta 130: 538–540 (1966).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhan, Xc., Jones, D.A. & Kerr, A. Regeneration of flax plants transformed by Agrobacterium rhizogenes . Plant Mol Biol 11, 551–559 (1988). https://doi.org/10.1007/BF00017455
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00017455