Synopsis
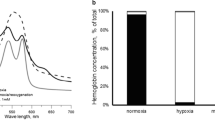
The oxygen transport characteristics and the acid-base status of carp blood was studied in vitro by equilibration of blood samples with and without addition of 5mmol l−1 of nitrite for 30 min at various Po2 values in combination with Pco2 of 1.5 and 5.7mmHg (0.2 and 0.76kPa). After equilibration pH, Po2, Pco2, and Co2 as well as methaemoglobin and HCO3 − concentration were determined and oxygen dissociation curves established. At Pco2 of 1.5mmHg (0.2kPa) oxygen affinity, expressed by a normal P50 of 3.3mmHg (0.44kPa) was unaffected by nitrite exposure, whereas at Pco2 5.7 (0.76kPa), nitrite exposure shifted P50 from 7.59mmHg (1.01kPa) to 21.9mmHg (2.92kPa). Methaemoglobin formation was greater at the higher Pco2 and increased with falling Po2. Erythrocyte shrinkage and rising plasma [HC03 −] during nitrite exposure indicated that the erythrocyte osmoregulation was significantly affected. The present results indicate significantly reduced oxygen affinity upon exposure of carp blood to nitrite. This result contrasts with findings in mammalian blood, where oxygen affinity is greatly enhanced.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References cited
Klein, D. & W. Lloyd. 1984. The history of glass Orbis London 288 pp Albers, C., R. Manz, D. Munster & G.M. Hughes. 1983. Effect of acclimation temperature on O2 transport in the blood of the carp,Cyprinus carpio. Resp. Physiol. 52: 165–179.
Chiodi, H. & J.G. Mohler, 1987. Nonautocatalytic methaemoglobin formation by sodium nitrite under aerobic and anaerobic conditions. Environ. Res. 44: 45–55.
Christoforides, C. & J. Hedley-Whyte. 1969. Effect of temperature and hemoglobin concentration on solubility of O2 in blood. J. Appl. Physiol. 27: 592–596.
Claiborne, J.B. & N. Heisler. 1984. Acid-base regulation and ion transfers in the carp (Cyprinus carpio) during and after exposure to environmental hypercapnia. J. Exp. Biol. 108: 25–43.
Darling, R.C. & F.J.W. Roughton. 1942. Effect of methaemoglobin on equilibrium between oxygen and hemoglobin. Amer. J. Physiol. 137: 56–58.
Eddy, F.B. & E.M. Williams. 1987. Nitrite and freshwater fish. Chem. Ecol. 3: 1–38.
Evelyn, K.A. & H.T. Malloy. 1938. Microdetermination of oxyhaemoglobin, methaemoglobin and sulphaemoglobin in a single sample of blood. J. Biol. Chem. 126: 655–662.
Freeman, L., T.L. Beitinger & D.W. Huey. 1983. Methemoglobin reductase activity in phylogenetically diverse piscine species. Comp. Biochem. Physiol. 75b: 27–30.
Glass, M.L., N.A. Andersen, M. Kruhøffer, E.M. Williams & N. Heisler. 1990. Combined effects of environmental P02 and temperature on ventilation and blood gases in the carp (Cyprinus carpio L.). J. Exp. Biol. 148: 1–17.
Harel, S., M.A. Salan & J. Kanner. 1988. Iron release from metmyoglobin, methaemoglobin and cytochrome c by a system generating hydrogen peroxide. Free Rad. Res. Comms. 5: 11–19.
Jaffé, E.R. 1981. Methaemoglobinaemia. Clin. Haematol. 10: 99–122.
Jensen, F.B. 1990. Nitrite and red cell function in carp: control factors for nitrite entry, membrane potassium ion permeation, oxygen affinity and methaemoglobin formation. J. Exp. Biol. 152: 149–166.
Jensen, F.B., N.A. Andersen & N. Heisler. 1987. Effects of nitrite exposure on blood respiratory properties acid-base and electrolyte regulation in the carp (Cyprinus carpio). J. Comp. Physiol. 157: 533–541.
Kosaka, H. & I. Tyuma. 1987. Mechanism of autocatalytic oxidation of oxyhaemoglobin by nitrite. Environ. Health Perspec. 73: 147–151.
Mansouri, A. 1989. Methemoglobin reduction under near physiological conditions. Biochem. Med. Metab. Biol. 42: 43–51.
Riggs, A.F. 1988. The Bohr effect. Ann. Rev. Physiol. 50: 181–204.
Rodkey, F.L. 1976. A mechanism for the conversion of oxyhaemoglobin to methaemoglobin by nitrite. Clin. Chem. 22/12: 1986–1990.
Spagnuolo, C., P. Rinelli, M. Coletta, E. Chiancone & F. Ascoli. 1987. Oxidation reaction of human oxyhemoglobin with nitrite: a re-examination. Biochem. Biophys. Acta 911: 59–65.
Thayer, J.R., J.H. Chasko, L.A. Swartz & N.J. Parxs. 1982. Gut reactions of radioactive nitrite after intratracheal administration in mice. Science 217: 151–153.
Tucker, C.S., R. Francis-Floyd & M.H. Beleau. 1989. Nitrite-induced anemia in channel catfish,Ictalurus punctatus Rafinesque. Bull. Environ. Contam. Tox. 43: 295–301.
Tucker, V.A. 1967. Method for oxygen content and dissociation curves on microliter blood samples. J. Appl. Physiol. 23: 410–414.
Ultsch, G.R., M.E. Ott & N. Heisler. 1981. Acid-base and electrolyte status in carp (Cyprinus carpio) exposed to low environmental pH. J. Exp. Biol. 93: 65–80.
Weber, R.E. & F.B. Jensen. 1988. Functional adaptations in hemoglobins from ectothermic vertebrates. Ann. Rev. Physiol. 50: 161–179.
Weber, R.E. & G. Lykkeboe. 1978. Respiratory adaptations in carp blood influences of hypoxia, red cell organic phosphates, divalent cations and CO2 on hemoglobin-O2 affinity. J. Comp. Physiol. 128: 127–137.
Williams, E.M. & F.B. Eddy. 1986. Chloride uptake in freshwater teleosts and its relationship to nitrite uptake and toxicity. J. Comp. Physiol. B. 156: 867–872.
Williams, E.M. & F.B. Eddy. 1988. Regulation of blood haemoglobin and electrolytes in rainbow troutSalmo gairdneri (Richardson) exposed to nitrite. Aquat. Tox. 13: 13–28.
Wolosin, J.M. & J.G. Forte 1985. Potassium and chloride conductances in the apical membrane from secreting oxyntic cells concurrently inhibited by divalent cations. J. Membr. Biol. 83: 261–272.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Williams, E.M., Glass, M.L. & Heisler, N. Effects of nitrite-induced methaemoglobinaemia on oxygen affinity of carp blood. Environ Biol Fish 37, 407–413 (1993). https://doi.org/10.1007/BF00005209
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00005209