Abstract
Reactive species are categorized into two broad sub-categories, reactive oxygen species (ROS) and reactive nitrogen species (RNS), possessing either a free radical with an unpaired electron in the valence shell or a neutral molecule. Free radicals are classified as one of the crucial steps in stress signaling which often cause unavoidable impairment to the essential biomolecules as a result of which the cell experiences various regulatory impairments. Redox homeostasis between the oxidants and antioxidants is the key to ensure normal cell functioning. As far as the current scientific evidence is concerned, almost all the plants possess natural antioxidants distributed throughout different parts of the plants. Different antioxidant assays show the potentiality of plant phenolics as an effective radical scavenger. Multiple hydroxyl and carbonyl groups of polyphenols help in the establishment of stable metal- and protein–polyphenol complex thereby restricting the development of free radicals. Phenols are known to work harmonious with other antioxidants thereby escalating the overall radical scavenging activity. A plentiful of research is evident on the propensity of plant phenolics as antioxidants but two things limit their current usability as food antioxidants. First, plant phenols are secondary metabolites and their continuous biotransformation inside the cells and tissues is an obvious event making their bioavailability difficult. Second, there are a few pieces of evidence on the toxicity and/or carcinogenicity of plant phenolics which restricts the considerations for the acceptability of such antioxidants as food additives. By considering the before-mentioned facts, this chapter compiles the emerging roles of phenolic compounds as antioxidant and impacts of multiple stressors on plant processes and stress managements through phenolic compounds.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
11.1 Introduction
The basic life functions of a typical plant involve production and utilization of various plant metabolites which are categorized into primary and secondary metabolites (SMs). The latter is a derivative of primary metabolite and often described as an intermediate or end product of metabolic pathway. A great proportion of organic compounds do not participate directly in growth and development. Unlike primary metabolites, the presence or absence of these metabolites does not strictly impair the organism’s established functionality. However, majority of the plant SMs are naturally constitutive and exist in their biologically active forms leaving few as inactive precursors. Damages to the plants caused by several biotic and abiotic factors trigger the activation and modulation of these inactive precursors. Complex metabolic pathway often involves overlapped intermediates of primary and secondary metabolism indicating common pathway being shared by primary and secondary metabolisms (Verpoorte et al. 2000; Yeoman and Yeoman 1996). There are three distinguished groups of SMs, includes terpenes, phenolics and nitrogen, sulphur compounds (Jamwal et al. 2018).
Phenolic compounds have one aromatic ring with one (phenol) or more (polyphenol) –OH− groups. These SMs often exist in the form of different functional derivatives (esters, methyl ethers, glycosides, etc.) synthesized either through the shikimic acid pathway or through the malonate/acetate pathway. Phenolics are broadly classified into simple phenolics (such as phenolic acids), polyphenols (such as tannins, flavonoids), lignans, coumarins, lignins, resveratrol, etc. on the basis of their carbon backbone (Harborne 1964; Cheynier et al. 2013). Solubility of simple phenolics can be changed from polar organic solvent to water through esterification and glycosylation with further facilitation through the increased number of hydroxyl groups in case of polyphenols. Even though phenolic compounds are abundant in plant kingdom but their allocated amount varies from one another depending on the selectionary advantage conferred to an individual plant during the course of evolution. Plant phenolics are divided in two classes such as preformed phenolics (synthesized during the normal development of plant) (Pridham 1960) and induced phenolics (elicitor mediated synthesis by plants during stressed environment) (Nicholson and Hammerschmidt 1992).
Plants being sessile experience multitudinous stresses ceaselessly throughout their growth and development due to the ever-changing environment (Singhal et al. 2017). Plants have no/little options to adapt with the changing conditions. One of the adaptive responses of plants is synthesize SMs, especially phenolic compounds. Phenolic compounds have been shown very effective under multiple abiotic and biotic stress and unfavorable climatic conditions and provide tolerance via antioxidant activity. This chapter compiles the overview of phenolic compounds and their role under multiple stressors.
11.2 Overview of Plant Phenolics and Its Role
11.2.1 Primary Function of Plant Phenolics as Antioxidants
Research in the field of plant ecology and plant physiology to explore the potential of plant phenolics has gained the pace by virtue of antioxidant and free radical scavenging properties of plant phenolics. Presence of more than one acidic hydroxyl groups in the phenyl ring of polyphenols makes them good donors of hydrogen or electron and hence the ability to confer free radical reduction (chain-breaking function) and termination of the Fenton reaction through the phenoxy radical intermediates. These phenoxy radical intermediates can also act as pro-oxidants at a higher pH in the presence of higher concentration of phenolic compounds and redox-active metals (copper, iron). Certain phenolics like flavonoids impede the membrane lipid peroxidation through altered membrane lipid packing. This alteration causes drastic loss of membrane fluidity and thus prevents free radical diffusion (Rice-Evans et al. 1997). Stimulatory role of phenolics cause amplification of cellular antioxidant capacity through host antioxidant enzyme responses (Shetty and Wahlqvist 2004). Phenolic coupling followed by oligomerization in case of complex polymeric phenolics (viz. tannins) escalates the antioxidant ability (Bors and Michel 2002).
11.2.2 Additional Functions of Phenolics in Plants
Phyllospheric and rhizospheric exudates containing polyphenols in the form of leachates can act as signaling compounds which when enter the soil cause alteration in the rates of decomposition and nutrient cycling through direct or indirect effects caused to soil microbial (decomposer) community (soil–microbe interaction). Flower and fruit pigmentation are also aided by phenolics (viz. flavonoids). This gives the plant a blessing in disguise for its own propagation through pollinator-mediated seed dispersal. Cross-linking among cell wall polymers ordinarily imparts the long-term maintenance of structural integrity. In this regard, hydroxycinnamate, a phenolic compound, plays a significant role making it difficult for the approaching pathogens to penetrate the cell wall. Among other physiological role of plant phenolics includes integral signal during plant–fungus interaction, maintain fruit quality and aroma, primary barrier during herbivores attack, synthesis of plant growth promoting substances, bioremediation, water and mineral absorption, chlorophyll content, pigment composition, and as allelochemicals (Zaprometov 1992; Sharma et al. 2019; Kumar et al. 2020a, b, c).
11.2.3 Impacts of Plant Phenolics on Plant Physiological, Biochemical, Reproductive, and Yield Traits
Wide distribution and as a part of key product of secondary metabolism, phenolics are known to influence various growth- and development-related physiological process in plants. Be it in normal or stress condition, plants get constant amelioration in the suboptimal conditions through phenolics-mediated tolerance and adaptation. As mentioned earlier, phenolics have diverse direct or indirect roles in nutrient mobilization through signal transduction (viz. flavonoid and salicylic acid signaling), allelopathy, plant growth promotion through plant growth promoting rhizobacteria (PGPR), pigmentation, etc. apart from their active role as antioxidants. Phenolics are involved in functional pollen development (Van Der Meer et al. 1992; Taylor and Grotewold 2005) and imbibition mediated boosting of seed germination rate (Shankar et al. 2009). Photosynthetic activity and biosynthesis of assimilatory pigments (chlorophyll a and b) can be amplified in C3 (sunflower) and C4 (maize) plants through polyphenols (Tanase et al. 2015).
11.2.3.1 Antioxidants: Definition, Classification, General Mechanism of Action
Antioxidants are the substances which stop the oxidation of any substance and counteract the free radicals at the same time generated during oxidative stress even with relatively low concentrations. Antioxidants discontinue these sequence reactions by eliminate free radical intermediates and obstruct other reactions by being oxidized themselves.
11.2.4 Classification of Antioxidants
Antioxidants can be grouped into natural and synthetic antioxidant system. Natural antioxidants are further categorized into enzymatic and non-enzymatic antioxidants. Enzymatic antioxidants include superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPX), glutathione reductase (GR), and glucose-6-phosphate dehydrogenase. Enzymatic antioxidants are produced endogenously where as non-enzymatic antioxidants are mostly from dietary origin. Non-enzymatic antioxidants include minerals (zinc, selenium), carotenoids (β-carotene, zeaxanthin, lycopene), vitamins (vit. A, C, E, K), some antioxidant cofactors (coenzyme Q10), and phenolics (Bunaciu et al. 2012) of which polyphenols is the largest class (Liu 2004). On the other hand, synthetic antioxidants (given in Fig. 11.1) are man-made compounds synthesized chemically. When stressed, plants generate singlet and triplet oxygen species, peroxides, harmful enzymes. The role of antioxidant system is to scavenge these toxic radicals as singlet and triplet oxygen quenchers, synergists, enzyme inhibitors, and peroxide decomposers (Manach et al. 1998). Some of the analytical methods to measure antioxidant content and capacity of plant extracts are based on assays involving hydrogen atom, assays involving electron transfer, chemiluminescence, etc. (Huang et al. 2005).
11.2.5 Stress-Mediated ROS Generation and Redox Signaling: Good or Bad?
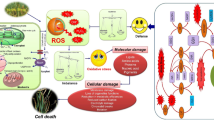
Exposure to various abiotic and biotic stresses leads to an imbalance in cellular redox homeostasis followed by generation of reactive oxygen species (ROS), which sooner or later leads to the oxidative stress and damage of plant cell organelles. For plant stress mitigation, optimal level of ROS acts as a priming step in the activation of antioxidant system through ROS-mediated redox signaling. On the contrary, over production of ROS leads to disruption of normal cell functions leading to the plant immune system failure (Hussain et al. 2019). Hence, generation of ROS can be considered as a necessary evil. Stress-mediated ROS generation has several impacts on plants both physiologically and biochemically through protein disruption, reduced CO2 fixation in chloroplasts, photooxidative damage to the photosynthetic complexes (principally PSII), membrane peroxidation ultimately leading to programmed cell death (PCD) (Amist et al. 2019).
ROS-mediated redox signaling is a cascade of event where redox-sensitive signaling proteins and metabolic enzymes undergo reversible oxidation/reduction to execute cellular functions via either regulating or amplifying the downstream signaling components, such as kinases, phosphatases, and transcription factors (Suzuki et al. 2012). Physiological events in the plants are regulated by phytohormones to a large extent. ROS acts as secondary messenger in the hormone signaling pathways and can impose hormonal regulation feedback or feedforward mechanism (Noctor et al. 2018). As a consequence of this cross-linked signaling between ROS and hormone, plants undergo modified root architecture triggering lateral root emergence with enhanced root hair growth (Du and Scheres 2018).
11.3 Abiotic Stress Response Through Phenolics
Challenges from ever transforming environment coupled with increased anthropogenic activities force plants to undergo a plethora of biotic and abiotic stresses throughout their life. These create a major setback in terms of their growth, development, and yield attributes.
Plants endure this unfavorable condition through different inherent mechanisms and acclimatize in the hostile environment surrounding them. In this regard, the chapter elaborates the role of antioxidant systems with a specific reference to phenolics (plant SM) to mitigate various abiotic and biotic stresses along with the basics of the mechanism underlying.
Plant SMs are believed to be considered as by-products until the scientific evidences regarding the active involvement of plant phenolics in maintaining a harmony between plant and its surrounded environment whether for stress mitigation or growth and developmental process have popped up. The fascinating fact about plant phenolics are they not only involved in the obliteration of harmful organisms associated with the abnormal growth and metabolism of plants but also playing a crucial role in maintaining the healthy population of beneficial symbiotic microorganisms around rhizosphere. The defensive action and expression of plant phenolics against biotic stresses (pathogenic microbes, insects, etc.) are something bestowed to plants during the natural evolution process. This built-in basal level of constitutive expression plus an induced expression of phenolics through stress-mediated signaling provides two-layered protection to the plants. Counteracting mechanism of phenolics against insects or pathogens can be direct or indirect (toxicity through induced changes in phenolic profiles) (Tyagi et al. 2020; Tak and Kumar 2020; Wagay et al. 2020). Among different schemes of the phenolics-mediated plant stress tolerance, the most noticeable is the activation of antioxidant systems which is synergistic to almost all the defense mechanisms in the plants. In fact, phenolics itself can act as an antioxidant to scavenge ROS (Amarowicz et al. 2010) and hinder the activities of specific oxidizing enzymes (Elavarthi and Martin 2010). Cross negotiation among various factors such as phytohormone production, biosynthetic pathway modulation occurs by virtue of the radical scavenging ability of antioxidant system and through optimized ROS signaling.
Physiological and molecular mechanisms concerning SMs production are deeply concatenated with the interactions between signaling molecules (salicylic acid, jasmonic acid, their derivatives, etc.) and enzymes for phenolics biosynthetic (phenylpropanoid) pathway (do Nascimento and Fett-Neto 2010; Cheynier et al. 2013). There is an absolute certitude that stress mitigation through phenolics is a resultant of one or more enzymatic (phenylalanine ammonia lyase; PAL, chalcone synthase; CHS, dihydroflavonol 4-reductase; DFR, cinnamate-4-hydroxylase; C4H, flavanone 3-hydroxylase; F3H, flavonol synthase; FLS, isoflavone synthase; IFS, isoflavone reductase; IFR, UDP flavonoid glycosyltransferase; UFGT) modulation(s) involved in phenolic biosynthetic pathway (Sharma et al. 2016, 2019; Ashraf et al. 2018; Naikoo et al. 2019).
11.3.1 Phenolics-Mediated Resistance Towards Salinity Stress
Phenylpropanoid-mediated synthesis of phenolics helps in scavenging ROS effectively in osmotic and ionic mismatches during salt stress (Rossi et al. 2016; Al-Ghamdi and Elansary 2018). Up-regulation of flavonoid biosynthetic genes like VvbHLH1, NtCHS1, GmFNSII-1, GmFNSII-2, etc. helps in accumulation of flavonoid thereby aiding phenolic-mediated ROS scavenging (Tanase et al. 2014; Wang et al. 2016; Zheng et al. 2017). Similarly, enhanced transcript levels of PAL, C4H, F3H, DFR, FLS facilitate tolerance mechanism to the plants under salt stress (Perin et al. 2019). In Brassicaceae (Chinese cabbage, white cabbage, and kale), short-term adaption to salinity stress is species specific and phenolic compound hydroxycinnamic acid is higher in tolerant species. In this case, phenolic compounds provide tolerance via maintaining of higher levels of SM, osmoprotectants, and suffer less from metabolic imbalance or disorders (Linić et al. 2019)
11.3.2 Phenolics-Mediated Resistance Towards Drought Stress
Reports suggest under water deficit conditions both the accumulation and synergism of phenolics (such as flavonoids) with other antioxidant systems go hand in hand resulting in plant resistance against drought stress (Sánchez-Rodríguez et al. 2011). Water scarcity induces closure of stomata resulting in less CO2 diffusion through stomatal pores. At this point, cell utilizes less reducing power (NADPH + H+) and ATP to synthesize carbohydrate as there is lower concentration of CO2 to be fixed. The lack of CO2 also leads to impaired Calvin cycle making it almost difficult for the conversion of NADPH + H+ and ATP to NADP+ and ADP + Pi. The non-utilization of vast number of free electrons possessed inside the reducing equivalent may then be able to break redox homeostasis through ROS generation inside the cell. Xanthophyll cycle play two key roles in drought stress. First, it helps in reverting the excess NADPH + H+ and ATP into NADP+ and ADP + Pi even in the absence of Calvin cycle. Second, it forms excess of SMs among which phenolics occupy the major proportion thus contributing towards the cellular redox homeostasis by scavenging ROS in the system if any (Gnanasekaran and Kalavathy 2017).
11.3.3 Phenolics-Mediated Resistance Towards Heavy Metal Stress
Transcriptional regulation of phenylpropanoid pathway enzymes (phenylalanine ammonia-lyase, chalcone synthase (CS), shikimate dehydrogenase, (SKDH) cinnamyl alcohol dehydrogenase (CAD), and polyphenol oxidase (PPO)) (Zafari et al. 2016; Chen et al. 2019) causes up-regulation in their activity under metal stress (Handa et al. 2019). This up-regulation is the main cause of phenolic (flavonoid)-mediated metal stress tolerance and their subsequent metal chelation process (Kisa et al. 2016) with an ultimatum of protecting the plants from oxidative stress and ROS.
11.3.4 Phenolics-Mediated Resistance Towards UV Stress
Light/UV stimulated flavonoid biosynthetic pathways and their corresponding gene transcript levels (Naikoo et al. 2019; Chen et al. 2019; Ghasemi et al. 2019) are the key to protect plant and its photosynthetic apparatus against high-intensity light and UV radiations by absorbing them (Lattanzio 2013; Landi et al. 2015). This modulation of the flavonoid biosynthetic pathway can either be a dependent or independent process on hormone (ABA) and/or jasmonic acid (Demkura et al. 2010; Berli et al. 2015).
Other abiotic factors like excess application of insecticides (Sharma et al. 2016), heat stress (Świeca 2015), chilling stress (Zhou et al. 2019; Wang et al. 2019), etc. invigorate cells’ ability to accumulate phenolic compounds by up-regulating biosynthetic pathway enzymes for quenching the reactive radicals. Plant cell wall thickening through lignification and suberization enhances freezing stress tolerance (Griffith and Yaish 2004).
11.4 Plant Phenolics and Biotic Stress: Beyond Antioxidant Role
The generalized scheme of plant stress mitigation through phenolics is depicted in Fig. 11.1. Like abiotic stresses, plants have to undergo multiple biotic stresses caused by diverse living micro or macroscopic organisms. Even though plants contain a diverse group of protective molecules such as preformed peptides, proteins, and SMs against these biotic factors but the concentrations of these molecules are often at basal levels. However, pathogenic attack causes genetic reprogramming followed by elevation to their levels through elicitor-induced expressions as documented in case of phenolics where plant accumulate higher levels of phenolics through elicitor-mediated up-regulation in the activities of biosynthetic enzymes such as phenylalanine ammonia lyase and chalcone synthase (Campos et al. 2003; Yedidia et al. 2003).
Detection of potential assailants can activate the plant defense response which is otherwise kept under tight genetic control in normal condition to maintain equilibrium between primary and secondary metabolism. The sudden surging in the SMs expression levels is due to the plant signal perception in the form of damages caused due to the toxins produced by pathogens and is evident from an increased levels of phenolics under fungal infection (Verma and Shukla 2015). In fact, preformed phenolic compounds are stored as antibiotic phenolics in inactive bound forms and get activated rapidly upon pathogen attack. The beauty of plant system having SMs (phenolics) is that there is no requirement of additional transcriptional activation for their expression but mere breakage of compartmentalization is enough to create a protective shield against biotic stresses (Osbourn 1996).
Phenolics-mediated oxidative reactions consume excess oxygen making a toxic environment for the pathogen survival. Presence of acidic hydroxyl groups in polyphenols has made them suitable candidates to penetrate biological membranes of fungus, while multiple hydroxyl groups help in uncoupling of oxidative phosphorylation (Siqueira et al. 1991; Parvez et al. 2004). Complex natural phenolics such as tannins and lignans can reduce the in vitro growth of fugal mycelium through their inhibitory action on the extracellular fungal enzymes which are essential for host tissue penetration (MacRae and Towers 1984). Mechanical penetration and survival of fungal pathogens are restricted via elicitor-mediated lignifications of plant cell wall which impede the movement of water and other diffusible molecules for pathogen survival (Garcia-Brugger et al. 2006; Houston et al. 2016). In this regard, flavonoid can also serve as low-molecular weight phytoalexins which are antimicrobial in nature (Samanta et al. 2011).
11.5 Factors Affecting the Regulation of Phenolic Biosynthesis
11.5.1 Proline Accumulation Under Stress
A key linkage between environmental stress and adaptive responses of plants towards stress is discussed here. Both biotic stresses and abiotic stresses induce the accumulation of a large quantity of free proline through reduced oxidation of the amino acids. This temporary proline accumulation through proline-D1 pyrroline-5-carboxylate (P5C) cycle generates a pool of NADP+ and regulates cellular redox potential (Kaur and Asthir 2015). Pentose phosphate pathway (PPP) is an established pathway for phenolic biosynthesis among others (Shetty and Wahlqvist 2004). Since NADP+ is the cofactor for the enzyme (glucose-6-phosphate dehydrogenase; G6PDH) catalyzing rate limiting step of PPP, proline-mediated high-NADP+/NADPH flux may augment phenolic biosynthesis (Cheynier et al. 2013) along with other phenolic precursor forming pathways like glycolysis (Maeda and Dudareva 2012) and calvin cycle (Lattanzio et al. 2009).
11.5.2 Cellular Compartmentalization
Synthesis of plant phenolics in thylakoids of chloroplasts and their subsequent storage (as inactive phenolic glycosides) in cell vacuoles manifest that the compartmentalization is a necessary physiological event to pile the reduced form of phenolics (Wink 1997). Stress-triggered de-compartmentalization (Beckman 2000) can cause mixing up of glycosidase enzyme with its inactive substrate (phenolic glycosides) resulting in clip off of glucose moiety thus making active phenolics ready for stress response (Dai et al. 1996).
11.5.3 Carbon Reallocation: As a Function of Growth vs Defense
Tolerance and resistance mechanisms of plants come with a high cost of resource allocation for the synthesis of defensive metabolites. Therefore, plants do not synthesize these metabolites unless they are threatened with potential harm above their threshold level. Unlike a stressed plant, a healthy plant only relies on the preformed or constitutive defenses (Morrissey and Osbourn 1999; Wittstock and Gershenzon 2002; Koricheva et al. 2004). Plant’s decision whether to grow or defend drives the quantitative (constitutive expression) and qualitative (de novo synthesis) synthesis phenolics through either carbon reallocation (production of defensive SMs instead of developmental primary metabolite like sucrose) or enzyme kinetics alteration at transcriptional level (Henkes et al. 2001; Nakane et al. 2003; Lloyd and Zakhleniuk 2004; Leser and Treutter 2005; Fritz et al. 2006; Walters and Heil 2007; Akula and Ravishankar 2011; Vos et al. 2013 represented in Fig. 11.2).
11.5.4 Theory of Photo-Protection and Co-evolution: Blessings in Disguise
Red to reddish-orange color of the deciduous tree leaves are due to the storing of anthocyanin, a class of flavonoids inside the acidic vacuole, a specialized leaf compartment. These color cues due to the presence of anthocyanin solve two major problems of the plants. First, it provides protection against photo-inhibition and photo-oxidation of photosynthetic pigments through light attenuation (green sunlight interception). Second, the red color might serve as a representation of plants poor nutrient status and hence not preferred by the insects and hence a low-insect load (Archetti 2009; Archetti et al. 2009; Nikiforou and Manetas 2010; Hughes 2011).
11.6 Plant Physiological, Genetical, Biochemical and Molecular Approaches to Improve Phenolic Compounds and Stress Mitigation
In terms of plant physiology, stress is defined as any abiotic or biotic factors which affect plant functioning and development. Nearly every plant has to undergo various forms of stress during their life cycle as a result of which it affects the growth and yield of the plant. Plants accumulate phenolic compounds as a defense strategy again, stress. Though it provides a defense to plants during adverse conditions but the trade-off between growth and defense has certain limitations. Since resources are limiting, the strategy of mitigating stress by improving the phenolic compound comes with certain restrictions. Three different approaches can achieve the engineering of improving phenolic content.
11.6.1 Biochemical Strategies of Phenolic Compounds to Mitigate Stress Tolerance
Growth regulators, nutrients, and fertilizers on phenolic development have crucial role in balancing of plant redox equilibrium. Treatment of grape leaf and fruit by sucrose and abscisic acid was found to be highly correlated with total phenolic content (Gambetta et al. 2010). Spayd et al. (1994) observed that nitrogen used to have an inhibitory effect on phenolic content. Under low nitrogen, the canopy was lighter and exposed for better light-enhancing phenolic accumulation. Phenolic content and metabolism have diversely been affected by growth regulators. Berhow (2000) studied that ABA had a little effect on the flavonon responsible for the bitterness of the grapefruit. Whereas GA reduced the flavonon content lowering the bitterness of grape. A plethora of literature suggests that the induction of biotic and abiotic stress response in plants is mediated by signaling molecules such as jasmonic acid, salicylic acid, and their derivatives. Such defensive phenolic compounds play a vital role when plants are infected with microbial pathogens. Lauvergeat et al. (2001) demonstrated that the application of salicylic acid in Arabidopsis resulting in accumulation of defense phenolic compounds. North et al. (2012) demonstrated that NAA and six BAP significantly increased phenolic exudation during micro-propagation. Several other studies showed that application of methyl jasmonate resulted in decreased phenolic content due to decreased gene expression of phenylalanine ammonia-lyase (PAL) important enzyme in chlorogenic acid biosynthesis. Ruiz et al. (2003) reported that calcium induces the activity of PAL. Due to the increase in total calcium content PAL activity increased, resulting in decreased phenolic content. Similarly, boron was found to have an inhibitory effect on phenols. Ruiz et al. (1998) reported that deficiency of boron leads to increase phenolic content. As provided from the information here, various biochemical factors play a significant role in increasing the accumulation in plants and hence crucial for mitigating abiotic stress.
11.6.2 Genetic/Metabolic Engineering of Phenolic Compounds to Mitigate Stress Tolerance
Flavonoids and carotenoids are one of the largest classes of plant phenolics. Both play an essential role in both abiotic and biotic stress response. Necessarily, the alteration in the biosynthetic pathway of phenolic compounds can optimize and regulate the production of flavonoids. The genetic and metabolic engineering of flavonoids and carotenoids can be achieved through following different approaches.
11.6.2.1 Blocking the Biosynthetic Pathway (RNAi Pathway)
In this technique, the RNA nucleotide, which is identical with the transcript sequence, is used. It is commonly known as the antisense RNA technique. Davuluri et al. (2005) reported that the operation of specific promoters using RNA technology increase the level of carotenoids. Casacuberta et al. (2015) reported that DET 1 gene was regulated using RNAi technology resulting in an enhanced level of flavonoids.
11.6.2.2 Endogenous Synthesis of Biochemical Compounds Using Structural and Regulatory Genes
The engineering of structural genes in the fruit peel of tomato resulted in a 70-fold elevation in flavonol (Bovy et al. 2007). CHS, CHI, F3H, and FLS are the four most essential enzymes flavonoid biosynthesis pathway. The overexpression of this for genes resulted in flavonoid production both in the peel (primary quercetin glycosides) and flesh (primary kampferol glycosides).
11.6.2.3 Inserting New Branches: The Pathway for the Production of Novel Flavonoids
Stilbene synthase is a key enzyme involved in trans-resveratrol biosynthesis. Trans-resveratrol is a phenolic compound with antioxidant properties. Bovy et al. (2007) reported that introduction of Stsy gene in lettuce enhanced the synthesis of revarstatol in transgenic lettuce. Shih et al. (2008) reported that engineering of soybean isoflavone synthase genes resulted in increased phenolic content. Through this genetic manipulation, neither the content of other phenolic compounds was altered or had any side effects.
11.6.3 Physiological Strategies of Phenolic Compounds to Mitigate Stress Tolerance
In the context of improving plant phenolics for abiotic stress tolerance, physiological strategy including screening based on mechanical wounding, chlorophyll pigment, osmolyte, nutrient stress, UV irradiation, and heat shock are essential (Godara et al. 2016). Among these various approaches, solar radiation plays a crucial role in increasing phenolic content in plants. Analysis of the metabolomics-based phenotyping of compounds, regulating physiological processes in plants, revealed that carotenoids and flavonoid content variation in different varieties could provide insight into an effective strategy for mitigating abiotic stress. For increasing the content of flavonoids and carotenoids, environmental factors play a crucial role. This happens due to the significant role of environment in modulating the physiological processes in plants. A list of environmental factors, which can be included in improving the phenolic content in the plant are enlisted below in Table 11.1.
11.7 Conclusion
In conclusion, phenolic compounds are ubiquitous in nature and have very crucial role in plant growth and developments. In the field conditions, plant faces a number of adverse situations like biotic and abiotic stresses at different growth stages and responsible for production of high ROS. These ROS are highly reactive, cause the membrane disruption of cell organelles, and disturb the functions of normal cell machinery. Phenolic compounds have important role in maintaining the cell functions under these situations by reducing ROS, provide structural integrity, signaling and change at biochemical and molecular levels. This chapter clearly concluded about the functions of phenolic compounds and impact of different stressors on physiological and biochemical levels of plants. Additionally, this chapter gives insight on the physiological, biochemical, and molecular mechanism of phenolic compounds under these circumstances. Therefore, a proper insight of the biochemical, physiological, and molecular understanding along with genomics, metabolomics, transcriptomics, and phenomics approach will enhance the opportunity of improving the phenolic content for mitigating multiple stress response in plants.
References
Akula R, Ravishankar GA (2011) Influence of abiotic stress signals on secondary metabolites in plants. Plant Signal Behav 6(11):1720–1731
Al-Ghamdi AA, Elansary HO (2018) Synergetic effects of 5-aminolevulinic acid and Ascophyllum nodosum seaweed extracts on Asparagus phenolics and stress related genes under saline irrigation. Plant Physiol Biochem 129:273–284
Amarowicz R, Weidner S, Wójtowicz I, Karmac M, Kosinska A, Rybarczyk A (2010) Influence of low-temperature stress on changes in the composition of grapevine leaf phenolic compounds and their antioxidant properties. Funct Plant Sci Biot 4:90–96
Amist N, Bano C, Singh NB (2019) Antioxidative machinery for redox homeostasis during abiotic stress. In: Molecular plant abiotic stress: biology and biotechnology, vol 12. Wiley, Hoboken, NJ, pp 65–90
Archetti M (2009) Decoupling vigour and quality in the autumn colours game: weak individuals can signal, cheating can pay. J Theor Biol 256(3):479–484
Archetti M, Döring TF, Hagen SB, Hughes NM, Leather SR, Lee DW, Lev-Yadun S, Manetas Y, Ougham HJ, Schaberg PG, Thomas H (2009) Unravelling the evolution of autumn colours: an interdisciplinary approach. Trends Ecol Evol 24(3):166–173
Ashraf MA, Iqbal M, Rasheed R, Hussain I, Riaz M, Arif MS (2018) Environmental stress and secondary metabolites in plants: an overview. In: Plant metabolites and regulation under environmental stress. Academic Press, London, pp 153–167
Beckman CH (2000) Phenolic-storing cells: keys to programmed cell death and periderm formation in wilt disease resistance and in general defence responses in plants? Physiol Mol Plant Pathol 57(3):101–110
Berhow MA (2000) Effects of early plant growth regulator treatments on flavonoid levels in grapefruit. Plant Growth Reg 30:225–232
Berli FJ, Alonso R, Beltrano J, Bottini R (2015) High-altitude solar UV-B and abscisic acid sprays increase grape berry antioxidant capacity. Am J Enol Vitic 66(1):65–72
Booij-James IS, Dube SK, Jansen MAK, Edelman M, Mattoo AK (2000) Ultraviolet-B radiation impacts light-mediated turnover of the photosystem II reaction center heterodimer in arabidopsis mutants altered in phenolic metabolism. Plant Physiol 124(3):1275–1283
Bors W, Michel C (2002) Chemistry of the antioxidant effect of polyphenols. Ann N Y Acad Sci 957(1):57–69
Bovy A, Schijlen E, Hall RD (2007) Metabolic engineering of flavonoids in tomato (Solanum lycopersicum): the potential for metabolomics. Metabolomics 3:399–412
Bunaciu AA, Aboul-Enein HY, Fleschin S (2012) FTIR spectrophotometric methods used for antioxidant activity assay in medicinal plants. Appl Spectrosc Rev 47(4):245–255
Campos ÂD, Ferreira AG, Hampe MM, Antunes IF, Brancão N, Silveira EP, Silva JB, Osório VA (2003) Induction of chalcone synthase and phenylalanine ammonia-lyase by salicylic acid and Colletotrichum lindemuthianum in common bean. Braz J Plant Physiol 15(3):129–134
Casacuberta JM, Devos Y, Jardin P, Ramon M, Vaucheret H, Nogue F (2015) Biotechnological uses of RNAi in plants: risk assessment considerations. Trends Biotechnol 33(3):145–147
Chen S, Wang Q, Lu H, Li J, Yang D, Liu J, Yan C (2019) Phenolic metabolism and related heavy metal tolerance mechanism in Kandelia Obovata under Cd and Zn stress. Ecotoxicol Environ Saf 169:134–143
Cheynier V, Comte G, Davies KM, Lattanzio V, Martens S (2013) Plant phenolics: recent advances on their biosynthesis, genetics, and ecophysiology. Plant Physiol Biochem 72:1–20
Dai GH, Nicole M, Andary C, Martinez C, Bresson E, Boher B, Daniel JF, Geiger JP (1996) Flavonoids accumulate in cell walls, middle lamellae and callose-rich papillae during an incompatible interaction between Xanthomonas campestris pv. malvacearum and cotton. Physiol Mol Plant Pathol 49(5):285–306
Davuluri GR, Tuinen A, van Fraser PD, Manfredonia A, Newman R, Burgess D, Brummell DA, King SR, Palys J, Uhlig J, Bramley PM, Pennings HMJ, Bowler C (2005) Fruit-specific RNAi-mediated suppression of DET1enhances carotenoid and flavonoid content in tomatoes. Nat Biotechnol 23(7):890–895
Demkura PV, Abdala G, Baldwin IT, Ballaré CL (2010) Jasmonate-dependent and-independent pathways mediate specific effects of solar ultraviolet B radiation on leaf phenolics and antiherbivore defense. Plant Physiol 152(2):1084–1095
do Nascimento NC, Fett-Neto AG (2010) Plant secondary metabolism and challenges in modifying its operation: an overview. In: Plant secondary metabolism engineering. Humana Press, Totowa, NJ, pp 1–13
Downey MO, Harvey JS, Robinson SP (2004) The effect of bunch shading on berry development and flavonoid accumulation in Shiraz grapes. Aust J Grape Wine Res 10(1):55–73
Du Y, Scheres B (2018) Lateral root formation and the multiple roles of auxin. J Exp Bot 69(2):155–167
Elavarthi S, Martin B (2010) Spectrophotometric assays for antioxidant enzymes in plants. In: Plant stress tolerance. Humana Press, New York, pp 273–280
Fletcher RS, Slimmon T, McAuley CY, Kott LS (2005) Heat stress reduces the accumulation of rosmarinic acid and the total antioxidant capacity in spearmint (Mentha spicata L). J Sci Food Agric 85(14):2429–2436
Fritz C, Palacios-Rojas N, Feil R, Stitt M (2006) Regulation of secondary metabolism by the carbon–nitrogen status in tobacco: nitrate inhibits large sectors of phenylpropanoid metabolism. Plant J 46(4):533–548
Gambetta GA, Matthews MA, Shaghasi TH, McElrone AJ, Castellarin SD (2010) Sugar and abscisic acid signaling orthologs are activated at the onset of ripening in grape. Planta 232(1):219–234
Garcia-Brugger A, Lamotte O, Vandelle E, Bourque S, Lecourieux D, Poinssot B, Wendehenne D, Pugin A (2006) Early signaling events induced by elicitors of plant defense. Mol Plant Microbe Interact 19(7):711–724
Ghasemi S, Kumleh HH, Kordrostami M (2019) Changes in the expression of some genes involved in the biosynthesis of secondary metabolites in Cuminum cyminum L. under UV stress. Protoplasma 256(1):279–290
Gnanasekaran N, Kalavathy S (2017) Drought stress signal promote the synthesis of more reduced phenolic compounds (chloroform insoluble fraction) in Tridax procumbens. Free Radic Antioxid 7(1). https://doi.org/10.5530/fra.2017.1.19
Godara OP, Kakralya BL, Kumar S, Kumar V, Singhal RK (2016) Influence of sowing time, varieties and salicylic acid application on different physiological parameters of Indian mustard (Brassica juncea L). J Pure Appl Microbiol 10(4):3063–3069
Griffith M, Yaish MW (2004) Antifreeze proteins in overwintering plants: a tale of two activities. Trends Plant Sci 9(8):399–405
Handa N, Kohli SK, Sharma A, Thukral AK, Bhardwaj R, Abd Allah EF, Alqarawi AA, Ahmad P (2019) Selenium modulates dynamics of antioxidative defence expression, photosynthetic attributes and secondary metabolites to mitigate chromium toxicity in Brassica juncea L. plants. Environ Exp Bot 161:180–192
Harborne JB (1964) Biochemistry of phenolic compounds. Academic Press, London
Henkes S, Sonnewald U, Badur R, Flachmann R, Stitt M (2001) A small decrease of plastid transketolase activity in antisense tobacco transformants has dramatic effects on photosynthesis and phenylpropanoid metabolism. Plant Cell 13(3):535–551
Houston K, Tucker MR, Chowdhury J, Shirley N, Little A (2016) The plant cell wall: a complex and dynamic structure as revealed by the responses of genes under stress conditions. Front Plant Sci 7:984
Huang D, Ou B, Prior RL (2005) The chemistry behind antioxidant capacity assays. J Agric Food Chem 53(6):1841–1856
Hughes NM (2011) Winter leaf reddening in ‘evergreen’ species. New Phytol 190(3):573–581
Hussain MB, Hassan S, Waheed M, Javed A, Farooq MA, Tahir A (2019) Plant physiological aspects of phenolic compounds. InTech, London
Jamwal K, Bhattacharya S, Puri S (2018) Plant growth regulator mediated consequences of secondary metabolites in medicinal plants. J Appl Res Med Aromatic Plants 9:26–38
Kaur G, Asthir BJ (2015) Proline: a key player in plant abiotic stress tolerance. Biol Plant 59(4):609–619
Kisa D, Elmastaş M, Öztürk L, Kayır Ö (2016) Responses of the phenolic compounds of Zea mays under heavy metal stress. Appl Biol Chem 59(6):813–820
Koricheva J, Nykänen H, Gianoli E (2004) Meta-analysis of trade-offs among plant antiherbivore defenses: are plants jacks-of-all-trades, masters of all? Am Nat 163(4):64–75
Kumar S, Abedin M, Singh AK, Das S (2020a) Role of phenolic compounds in plant-defensive mechanisms. In: Plant phenolics in sustainable agriculture. Springer, Singapore, pp 517–532
Kumar S, Bhushan B, Wakchaure GC, Meena KK, Kumar M, Meena NL, Rane J (2020b) Plant phenolics under water-deficit conditions: Biosynthesis, accumulation, and physiological roles in water stress alleviation. In: Plant phenolics in sustainable agriculture. Springer, Singapore, pp 451–465
Kumar M, Tak Y, Potkule J, Choyal P, Tomar M, Meena NL, Kaur C (2020c) Phenolics as plant protective companion against abiotic stress. In: Plant phenolics in sustainable agriculture. Springer, Singapore, pp 277–308
Landi M, Tattini M, Gould KS (2015) Multiple functional roles of anthocyanins in plant-environment interactions. Environ Exp Bot 119:4–17
Lattanzio V (2013) Phenolic compounds: introduction. In: Ramawat KG, Mérillon J-M (eds) Handbook of natural products. Springer, Berlin
Lattanzio V, Cardinali A, Ruta C, Fortunato IM, Lattanzio VM, Linsalata V, Cicco N (2009) Relationship of secondary metabolism to growth in oregano (Origanum vulgare L.) shoot cultures under nutritional stress. Environ Exp Bot 65(1):54–62
Lauvergeat V, Lacomme C, Lacombe E, Lasserre E, Roby D, Grima-Pettenati J (2001) Two cinnamoyl-CoA reductase (CCR) genes from Arabidopsis thaliana are differentially expressed during development and in response to infection with pathogenic bacteria. Phytochemistry 57(7):1187–1195
Leser C, Treutter D (2005) Effects of nitrogen supply on growth, contents of phenolic compounds and pathogen (scab) resistance of apple trees. Physiol Plant 123(1):49–56
Linić I, Šamec D, Grúz J, Vujčić Bok V, Strnad M, Salopek-Sondi B (2019) Involvement of phenolic acids in short-term adaptation to salinity stress is species-specific among Brassicaceae. Plan Theory 8(6):155
Liu RH (2004) Potential synergy of phytochemicals in cancer prevention: Mechanism of action. J Nutr 134:3479S–3485S
Lloyd JC, Zakhleniuk OV (2004) Responses of primary and secondary metabolism to sugar accumulation revealed by microarray expression analysis of the Arabidopsis mutant, pho3. J Exp Bot 55(400):1221–1230
MacRae WD, Towers GN (1984) Biological activities of lignans. Phytochemistry 23(6):1207–1220
Maeda H, Dudareva N (2012) The shikimate pathway and aromatic amino acid biosynthesis in plants. Annu Rev Plant Biol 63:73–105
Manach C, Morand C, Crespy V, Demigné C, Texier O, Régérat F, Rémésy C (1998) Quercetin is recovered in human plasma as conjugated derivatives which retain antioxidant properties. FEBS Lett 426(3):331–336
Marsilio V, d’Andria R, Lanza B, Russi F, Iannucci E, Lavini A, Morelli G (2006) Effect of irrigation and lactic acid bacteria inoculants on the phenolic fraction, fermentation and sensory characteristics of olive (Olea europaea L. cv. Ascolana tenera) fruits. J Sci Food Agric 86(6):1005–1013
Mori K, Sugaya S, Gemma H (2005) Decreased anthocyanin biosynthesis in grape berries grown under elevated night temperature condition. Sci Hortic 105(3):319–330
Morrissey JP, Osbourn AE (1999) Fungal resistance to plant antibiotics as a mechanism of pathogenesis. Microbiol Mol Biol Rev 63(3):708–724
Naikoo MI, Dar MI, Raghib F, Jaleel H, Ahmad B, Raina A, Khan FA, Naushin F (2019) Role and regulation of plants phenolics in abiotic stress tolerance: an overview. In: Plant signaling molecules. Woodhead Publishing, Duxford, pp 157–168
Nakane E, Kawakita K, Doke N, Yoshioka H (2003) Elicitation of primary and secondary metabolism during defense in the potato. J Gen Plant Pathol 69(6):378–384
Nicholson RL, Hammerschmidt R (1992) Phenolic compounds and their role in disease resistance. Annu Rev Phytopathol 30(1):369–389
Nikiforou C, Manetas Y (2010) Strength of winter leaf redness as an indicator of stress vulnerable individuals in Pistacia lentiscus. Flora Morphol Distrib Funct Ecol Plants 205(6):424–427
Noctor G, Reichheld JP, Foyer CH (2018) ROS-related redox regulation and signalling in plants. In: Seminars in cell and developmental biology, vol 80. Academic Press, London, pp 3–12
North JJ, Ndakidemi PA, Laubscher CP (2012) Effects of antioxidants, plant growth regulators and wounding on phenolic compound excretion during micropropagation of Strelitzia reginae. Int J Phys Sci 7(4):638–646
Osbourn AE (1996) Preformed antimicrobial compounds and plant defense against fungal attack. Plant Cell 8:1821
Parvez MM, Tomita-Yokotani K, Fujii Y, Konishi T, Iwashina T (2004) Effects of quercetin and its seven derivatives on the growth of Arabidopsis thaliana and Neurospora crassa. Biochem Syst Ecol 32(7):631–635
Perin EC, da Silva Messias R, Borowski JM, Crizel RL, Schott IB, Carvalho IR, Rombaldi CV, Galli V (2019) ABA-dependent salt and drought stress improve strawberry fruit quality. Food Chem 15(271):516–526
Pridham JB (1960) Phenolics in plants in health and disease. In: Proceedings of a plant phenolics group symposium held at Bristol, April 1959. Pergamon Press, Oxford, London
Reuber S, Bornman JF, Weissenbock G (1996) A flavonoid mutant of barley (Hordeum vulgare L.) exhibits increased sensitivity to UV-B radiation in the primary leaf. Plant Cell Environ 19(5):593–601
Rice-Evans C, Miller N, Paganga G (1997) Antioxidant properties of phenolic compounds. Trends Plant Sci 2(4):152–159
Rossi L, Borghi M, Francini A, Lin X, Xie DY, Sebastiani L (2016) Salt stress induces differential regulation of the phenylpropanoid pathway in Olea europaea cultivars Frantoio (salt-tolerant) and Leccino (salt-sensitive). J Plant Physiol 204:8–15
Ruiz JM, Bretones G, Baghour M, Ragala L, Belakbir A, Romero L (1998) Relationship between boron and phenolic metabolism in tobacco leaves. Phytochemistry 48(2):269–272
Ruiz JM, Rivero RM, Lopez-Cantarero I, Romero L (2003) Role of Ca2+ in the metabolism of phenolic compounds in tobacco leaves (Nicotiana tabacum L.). Plant Growth Regul 41(2):173–177
Samanta A, Das G, Das SK (2011) Roles of flavonoids in plants. Carbon 100(6):12–35
Sánchez-Rodríguez E, Moreno DA, Ferreres F, del Mar Rubio-Wilhelmi M, Ruiz JM (2011) Differential responses of five cherry tomato varieties to water stress: changes on phenolic metabolites and related enzymes. Phytochemistry 72(8):723–729
Santos TP, Lopes CM, Rodrigues ML, De Souza CR, Ricardo-Da-Silva JM, Maroco JP, Pereira JS, Chaves MM (2005) Effects of partial root-zone drying irrigation on cluster microclimate and fruit composition of field-grown Castelao grapevines. Vitis 44(3):117–125
Shankar SR, Girish R, Karthik N, Rajendran R, Mahendran VS (2009) Allelopathic effects of phenolics and terpenoids extracted from Gimelina arborea on germination of Black gram (Vigna mungo) and Green gram (Vigna radiata). Allelopath J 23(2):323–332
Sharma M, Sandhir R, Singh A, Kumar P, Mishra A, Jachak S, Singh SP, Singh J, Roy J (2016) Comparative analysis of phenolic compound characterization and their biosynthesis genes between two diverse bread wheat (Triticum aestivum) varieties differing for chapatti (Unleavened Flat Bread) quality. Front Plant Sci 15(7):1870
Sharma A, Shahzad B, Rehman A, Bhardwaj R, Landi M, Zheng B (2019) Response of phenylpropanoid pathway and the role of polyphenols in plants under abiotic stress. Molecules 24(13):2452
Shetty K, Wahlqvist M (2004) A model for the role of the proline-linked pentose-phosphate pathway in phenolic phytochemical bio-synthesis and mechanism of action for human health and environmental applications. Asia Pac J Clin Nutr 13(1):1–24
Shih CH, Chen Y, Wang M, Chu IK, Lo C (2008) Accumulation of isoflavonegenistin in transgenic tomato plants overexpressing a soybean isoflavone synthase gene. J Agric Food Chem 56:5655–5661
Singhal RK, Sodani R, Chauhan J, Sharma MK, Yashu BR (2017) Physiological adaptation and tolerance mechanism of rice (Oryza sativa L.) in multiple abiotic stresses. Int J Pure Appl Biosci 5(3):459–466
Siqueira JO, Nair MG, Hammerschmidt R, Safir GR, Putnam AR (1991) Significance of phenolic compounds in plant-soil-microbial systems. Crit Rev Plant Sci 10(1):63–121
Spayd SE, Wample RL, Evans RG, Stevens RG, Seymour BJ, Nagel CW (1994) Nitrogen fertilization of white Riesling grapes in Washington. Must and wine composition. Am J Enol Vitic 45(1):34–42
Spayd SE, Tarara JM, Mee DL, Ferguson JC (2002) Separation of sunlight and temperature effects on the composition of Vitis vinifera cv. Merlot berries. Am J Enol Vitic 53(3):171–182
Suzuki N, Koussevitzky SH, Mittler RO, Miller GA (2012) ROS and redox signalling in the response of plants to abiotic stress. Plant Cell Environ 35(2):259–270
Świeca M (2015) Elicitation with abiotic stresses improves pro-health constituents, antioxidant potential and nutritional quality of lentil sprouts. Saudi J Biol Sci 22(4):409–416
Tak Y, Kumar M (2020) Phenolics: a key defence secondary metabolite to counter biotic stress. In: Plant phenolics in sustainable agriculture. Springer, Singapore, pp 309–329
Tanase C, Boz I, Stingu A, Volf I, Popa VI (2014) Physiological and biochemical responses induced by spruce bark aqueous extract and deuterium depleted water with synergistic action in sunflower (Helianthus annuus L.) plants. Ind Crop Prod 60:160–167
Tanase C, Bara CI, Popa VI (2015) Cytogenetical effect of some polyphenol compounds separated from industrial by-products on maize (Zea mays L.) plants. Cell Chem Technol 49(9–10):799–805
Taylor LP, Grotewold E (2005) Flavonoids as developmental regulators. Curr Opin Plant Biol 8(3):317–323
Tyagi K, Shukla P, Rohela GK, Shabnam AA, Gautam R (2020) Plant phenolics: their biosynthesis, regulation, evolutionary significance, and role in senescence. In: Plant phenolics in sustainable agriculture. Springer, Singapore, pp 431–449
Van Der Meer IM, Stam ME, van Tunen AJ, Mol JN, Stuitje AR (1992) Antisense inhibition of flavonoid biosynthesis in petunia anthers results in male sterility. Plant Cell 4(3):253–262
Verma N, Shukla S (2015) Impact of various factors responsible for fluctuation in plant secondary metabolites. J Appl Res Med Aromatic Plants 2(4):105–113
Verpoorte R, van der Heijden R, Memelink J (2000) Engineering the plant cell factory for secondary metabolite production. Transgenic Res 9(4–5):323–343
Vos IA, Pieterse CM, Van Wees SC (2013) Costs and benefits of hormone-regulated plant defences. Plant Pathol 62:43–55
Wagay NA, Lone R, Rafiq S, Bashir SU (2020) Phenolics: a game changer in the life cycle of plants. In: Plant phenolics in sustainable agriculture. Springer, Singapore, pp 241–275
Walters D, Heil M (2007) Costs and trade-offs associated with induced resistance. Physiol Mol Plant Pathol 71(1–3):3–17
Wang LY, Liu JL, Wang WX, Sun Y (2016) Exogenous melatonin improves growth and photosynthetic capacity of cucumber under salinity-induced stress. Photosynthetica 54(1):19–27
Wang L, Shan T, Xie B, Ling C, Shao S, Jin P, Zheng Y (2019) Glycine betaine reduces chilling injury in peach fruit by enhancing phenolic and sugar metabolisms. Food Chem 272:530–538
Wink M (1997) Compartmentation of secondary metabolites and xenobiotics in plant vacuoles. In: Advances in botanical research, vol 25. Academic Press, San Diego, pp 141–169
Wittstock U, Gershenzon J (2002) Constitutive plant toxins and their role in defense against herbivores and pathogens. Curr Opin Plant Biol 5(4):300–307
Yedidia I, Shoresh M, Kerem Z, Benhamou N, Kapulnik Y, Chet I (2003) Concomitant induction of systemic resistance to Pseudomonas syringae pv. lachrymans in cucumber by Trichoderma asperellum (T-203) and accumulation of phytoalexins. Appl Environ Microbiol 69(12):7343–7353
Yeoman MM, Yeoman CL (1996) Tansley Review No. 90. Manipulating secondary metabolism in cultured plant cells. New Phytol 1:553–569
Zafari S, Sharifi M, Chashmi NA, Mur LA (2016) Modulation of Pb-induced stress in Prosopis shoots through an interconnected network of signaling molecules, phenolic compounds and amino acids. Plant Physiol Biochem 99:11–20
Zaprometov MN (1992) On the functional role of phenolic compounds in plants. Fiziologiya rastenij (Russian Federation)
Zheng X, Tan DX, Allan AC, Zuo B, Zhao Y, Reiter RJ, Wang L, Wang Z, Guo Y, Zhou J, Shan D (2017) Chloroplastic biosynthesis of melatonin and its involvement in protection of plants from salt stress. Sci Rep 7(1):1–2
Zhou P, Li Q, Liu G, Xu N, Yang Y, Zeng W, Chen A, Wang S (2019) Integrated analysis of transcriptomic and metabolomic data reveals critical metabolic pathways involved in polyphenol biosynthesis in Nicotiana tabacum under chilling stress. Funct Plant Biol 46(1):30–43
Zucker M (1965) Induction of phenylalanine deaminase by light and its relation to chlorogenic acid synthesis in potato tuber tissue. Plant Physiol 40(5):779–784
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Mishra, U.N. et al. (2023). Plant Phenolics: As Antioxidants and Potent Compounds Under Multiple Stresses. In: Lone, R., Khan, S., Mohammed Al-Sadi, A. (eds) Plant Phenolics in Abiotic Stress Management. Springer, Singapore. https://doi.org/10.1007/978-981-19-6426-8_11
Download citation
DOI: https://doi.org/10.1007/978-981-19-6426-8_11
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-19-6425-1
Online ISBN: 978-981-19-6426-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)