Abstract
The regulation of plant metabolism, development, and growth depends upon different nutrient requirements, and phosphorus (P) plays an essential role in it. Though there is a huge presence of P in soil, the bioavailability of the element is very less for plants due to its chemical fixation in soil environment. Application of rock phosphate in cooperation with solubilization by microbes may help to enhance the phosphate level in soil. This technology not only remunerate the expensive industrial manufactured fertilizers but also helps to mobilize the fertilizers added to the soil. The phosphate-solubilizing microorganisms predominantly belongs to the genera of Aspergillus, Actinomycetes, Aereobacter, Agrobacterium, Bacillus, Enterobacter, Erwinia, Micrococcus, Penicillium, Pseudomonas, Rhizobium, and Trichoderma. The solubilization mechanisms used by these PSMs were several such as (i) lower pH by acid production, (ii) chelation of ion, and (iii) exchanging reactions within the broth. These qualities put froth an ideal technology for the development of potent microbial inoculum for ultimate utilization in agricultural systems. The potentiality of PSMs as rock phosphate solubilizers to enhance crop production and the detailed mechanisms are discussed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
17.1 Introduction
Phosphorus (P) is considered an essential macronutrient for plant growth and improvement after nitrogen (N). It plays an essential role in plant metabolism, i.e., cell cycle regulation, division of the cell, plant development, photosynthesis, breaking of sugars, transportation of nutrient within the plant, regulation of metabolic pathways, and genetic character transfer from one generation to another (Hamdali et al. 2008; Sharma et al. 2013). Although there is considerable level of P in the soil system, a large portion of this is not readily available to plants due to complex formation with cations like iron, aluminum, and calcium (Son et al. 2006). In order to fulfil the P requirement of plants, the commercially available chemical fertilizers can be applied to increase the crop productivity. However, the high-cost production and the fertilizers being chemically processed with insoluble mineral phosphate (high-grade ore) with the treatment of sulfuric acid in high temperature makes it a polluting constituent (Shigaki et al. 2006; Vassilev et al. 2006). With the application of phosphorus-based fertilizers to the soil, the initiation of reactions between P, soil, and nonphosphatic fertilizer components takes place, which results in the formation of insoluble P and makes it unavailable for plant growth regulation (Ivanova et al. 2006).
Furthermore, the soluble P gets rapidly washed away, responsible for the eutrophication of freshwater and pollution in groundwater used for drinking (Shigaki et al. 2006). Recently the focus is shifted toward the microbial-mediated rock phosphate solubilization techniques and its application in agricultural field. The phosphate-solubilizing microorganisms (PSM) converts the insoluble phosphates to a soluble form by acidification, chelation, and exchange reaction (Coutinho et al. 2012). Microbial activities play a prime role in the increment of bioavailability of minerals that are the main part of plant growth regulations and eventually reduce the demand of unsustainable chemical fertilizers (Kennedy et al. 2004). Therefore, this process not only seems a cheap alternative to the use of expensive chemical fertilizer but also enhances the mobilization of fertilizers added to the soil.
17.2 Availability of Rock Phosphate
The distribution of rock phosphate reserves can be found in sedimentary and ingenious deposits around the world. The phosphate reserves around the world were approximate 68 billion metric tons according to the US Geological Survey of 2017 (Kohn et al. 2018). Morocco contained approximately 70% of the total reserves of phosphate rock which is 50 billion tons. After that China comes in the second position, having 5% of the total reserves, which indicates 3.1 billion tons. However China is the largest producer of phosphate at 138 million metric tons. The third largest reserves of phosphate are Syria and Algeria which contributes 3% to the world’s phosphate reserves, i.e., 1.8 and 2.2 billion metric tons, respectively. Russia, South Africa, the USA, Egypt, and Jordan each contribute to 2% of world’s phosphate reserves. The 1% of world’s phosphate reserve belongs to Peru, Saudi Arabia, Senegal, Australia, and Iran, respectively, and other countries account for less than 1% of the total reserves which contribute to the global phosphate production.
In comparison to other producers all over the world, the contribution of India in the world market doesn’t show any profitable numbers. According to the NMI data based on the UNFC system, India provides 312.67 million tonnes of phosphate, while 266.87 million tons of phosphate comes from the remaining resource categories. The reserves constitute 45.80 million ton only. The states like Jharkhand, Rajasthan, Madhya Pradesh, Uttar Pradesh, and Uttarakhand contribute 34%, 31%, 19%, and 8% each, respectively, to the total reserves in India. Meghalaya and Gujarat also constitute less quantities of resources. According to the Indian Minerals Yearbook (2016), if the gradation of phosphorus is taken in account, the low-grade phosphorus is 37%, followed by beneficial (29%), blendable (11%), chemical fertilizer and soil reclamation (8% each), and the remaining unclassified and not-known grades (about 7%) (Indian Minerals Yearbook 2016).
The major mineral of rock phosphate is apatite, and the most common is fluorapatite-Ca5(PO4)3F or Ca10(PO4)6(F,OH)2. As the mineral is insoluble in nature, it cannot be used as fertilizer directly. Some of the soil’s physicochemical factors such as pH, reaction time, and Ca2+ or H2PO4− absorption ability may affect the rock phosphate solubility. The level of solubility is determined from the degrees of fitness and the origin of phosphate. Compared to phosphates of igneous or metamorphic origin, the sedimentary RP origin may be more soluble (Nahas 1996). The most important factor is the soil pH which affects the efficacy of the RP (Ma and Rao 1999), as the increment in soil acidity results in higher rate of dissolution and effectiveness. The immobilization of Pb in Pb-contaminated soil has been reported by several studies after the application of phosphoric acid to RP (Yang et al. 2001; Cao et al. 2003; Yoon et al. 2007; Cao et al. 2009). However, the acidic water solubilization of P source exhibits a great disadvantage due to the low pH results in potential leaching of both the applied P and Pb in the soil. Therefore the application of P-solubilizing microorganisms to act on RP to enhance solubilization could be a potential approach than acidification.
17.3 Phosphate-Solubilizing Microorganisms (PSMs) Accountable for RP Solubilization
The solubilization of insoluble mineral phosphate into ionic forms could be possible by using soil microorganisms from different ecological niches which can be utilized by the crop plants. From the soil and rhizosphere samples, many PSMs like bacterial, fungal, yeast, and actinomycete species have been isolated, identified, and characterized (Kucey et al. 1989). Bacterial species acts as a potent culture in comparison to fungus to solubilize phosphorus (Alam et al. 2002). PSMs are present ubiquitously in nature, and their broad range of variation is observed in different soil environments (López-Arredondo and Herrera-Estrella 2012). In the whole microbial population, the presence of PSB is 1–50%, and PSF are found to be 0.1–0.5%, showing higher rate of P solubilization.
17.3.1 Bacteria
The potent bacterial strain can be isolated by a series of experiments on bacterial species that help to solubilize efficiently the insoluble inorganic phosphate compounds, like tricalcium phosphate, dicalcium phosphate, hydroxyapatite, and rock phosphate (Goldstein 1986). The bacterial genera having solubilizing properties are found to be Achromobacter, Aereobacter, Agrobacterium, Bacillus, Burkholderia, Flavobacterium, Micrococcus, Pseudomonas, and Rhizobium, and they increase the accessibility of soluble phosphate and enhance biological nitrogen fixation, resulting in enhanced plant growth (Kucey et al. 1989; Ponmurugan and Gopi 2006). With the incorporation of Pseudomonas spp., the nodule number, its dry weight, yield components, grain yield, nutrient availability, and uptake in soybean crop have been enhanced (Son et al. 2006). PSB supports the enhancement in seedling length of Cicer arietinum (Sharma et al. 2007), while the inoculation of PSM along with PGPR showed reduction in P application by 50%, and the corn yield has not been affected (Yazdani et al. 2009). Below is the list of a few bacterial species studied for their P-solubilizing potential:
Gram-positive bacteria: Bacillus brevis, B. cereus var. albolactis, B. circulans, B. coagulans, B. firmus, B. megaterium, B. megaterium var. phosphaticum, B. mesentricum, B. mycoides, B. polymyxa, B. pumilus, B. pulvifaciens, B. sphaericus, B. subtilis, Clostridium sp., B. licheniformis, B. amyloliquefaciens, and A. atrophaeus.
Gram-negative bacteria: Acetobacter diazotrophicus, Achromobacter sp., Aerobacter aerogenes, Agrobacterium radiobacter, Agrobacterium sp., Alcaligenes sp., Arthrobacter mysorens, Bradyrhizobium sp., Brevibacterium sp., Burkholderia cepacia, Citrobacter freundii, Enterobacter aerogenes, Enterobacter agglomerans, Enterobacter asburiae, Enterobacter cloacae, Escherichia freundii, Escherichia intermedia, Erwinia herbicola, Flavobacterium sp., Gluconacetobacter diazotrophicus, Micrococcus sp., Mycobacterium sp., Nitrosomonas sp., Pseudomonas calcis, P. cepacia, P. fluorescens, P. putida, P. rathonia, P. striata, P. syringae, Serratia marcescens, S. phosphaticum, Thiobacillus ferrooxidans, T. thiooxidans, Rahnella aquatilis, Rhizobium meliloti, Xanthomonas sp., Azotobacter chroococcum, Kluyvera ascorbata, Azospirillum brasilense, A. lipoferum, and Acinetobacter calcoaceticus.
17.3.2 Fungi
After bacterial species, fungal strains such as Aspergillus and Penicillium are recognized to be the most potent P solubilizers (Whitelaw 2000). Mycorrhizal fungi and phosphate-solubilizing microorganisms are identified to process the bioavailable P (Fankem et al. 2006). To solubilize the phosphate rocks, a nematophagous fungus Arthrobotrys oligospora has also been identified as a potent species (Duponnois et al. 2006). These fungal species have the capability in converting the insoluble P into its soluble form, produce plant growth promoting substances, and also protect plants from soil pathogens. A fungus does not lose its solubilizing capability with repeated subculturing under laboratory conditions like that of the PSB (Sperber 1958; Kucey 1983). Moreover, the fungus has the capability to traverse longer distance in the soil than bacterial species making them suitable for P solubilization (Kucey 1983). Generally in PSF the rate of acid production is higher in comparison to bacteria, which indicates the higher P-solubilizing activity (Venkateswarlu et al. 1984). Duponnois et al. (2006) have also reported nematophagous fungus, i.e., Arthrobotrys oligospora, which shows a significant rate of RP solubilization (Meena et al. 2013, 2016; Bahadur et al. 2014; Maurya et al. 2014; Jat et al. 2013; Kumar et al. 2015; Ahmad et al. 2016; Parewa et al. 2014; Prakash and Verma 2016). Mycorrhizal fungi are another group of microorganisms playing an important role in P acquisition (Fankem et al. 2006). Below are the list of fungi studied for having potential P dissolution phenotype.
PSF: Achrothecium sp., Alternaria tenuis, Aspergillus aculeatus, A. awamori, A. carborundum, A. flavus, A. foetidus, A. fumigatus, A. japonicus, A. nidulans, A. nidulans var. acristatus, A. niger, A. rugulosus, A. terreus, A. wentii, Cephalosporium sp., Chaetomium globosum, Cladosporium herbarum, Cunninghamella sp., C. elegans, Curvularia lunata, Fusarium oxysporum, Helminthosporium sp., Humicola lanuginosa, H. inslens, Mortierella sp., Micromonospora sp., Mucor sp., Myrothecium roridum, Oidiodendron sp., Paecilomyces lilacinus, P. fusisporus, Penicillium aurantiogriseum, P. bilaji, P. digitatum, P. funiculosum, P. lilacinum, P. oxalicum, P. pinophilum, P. rubrum, P. rugulosum, P. simplicissimum, P. variabile, Phoma sp., Populospora mytilina, Pythium sp., Rhizoctonia solani, Rhizopus sp., Sclerotium rolfsii, Torulaspora globosa, Torula thermophila, Trichoderma harzianum, T. viridae, Schwanniomyces occidentalis, Emericella rugulosa, Penicillium camemberti, and Colletotrichum sp.
Yeast: Pichia fermentans, Schizosaccharomyces pombe, and Yarrowia lipolytica.
17.3.3 Other Soil Microorganisms
17.3.3.1 Mycorrhizae
The most essential component for sustainable soil-plant systems among the microbial community is identified to be the Arbuscular mycorrhizal fungi (AMF) (Schreiner et al. 2003). It has a positive response on nitrogen (Barea et al. 1991), phosphorus (Bolan 1991), and other micronutrient (Bürkert and Robson 1994) uptake; responsible for soil aggregation (Tisdall 1994) for the plant system; and has also analogist impacts against some plant pathogens (Duponnois et al. 2005). Moreover, research in this field has shown that in comparison to non-inoculated plant, the AMF, in symbiotic association with the host, uses additional soluble phosphate from rock phosphate (Antunes and Cardoso 1991; Guissou et al. 2001). The insoluble mineral phosphate solubilization by organic acid produced by AMF has been proved by many evidences (Lapeyrie 1988).
17.3.3.2 Actinomycetes
In recent years the actinomycetes attracted the research interest globally due to its P-solubilizing property, because this community of soil microbes has better tolerance toward extreme environment like drought, temperature, etc. and also has the ability to produce benefits such as antibiotic and compounds like phytohormone production which is ultimately responsible for plant growth regulation (Fabre et al. 1988; Hamdali et al. 2008). The major area of rhizoplane and rhizosphere are occupied by the actinomycetes (Solans and Vobis 2003; Frioni 2006) and have a key role in the regulation of soil nutrient cycle (Elliott and Lynch 1995). Below are the list of few actinomycetes known to have an active role in P solubilization.
Actinomyces coelicolor, Actinomyces sp., Agromyces soli, Angustibacter luteus, Isoptericola hypogeus, Isoptericola variabilis, Kocuria flava, Kocuria palustris, M. kitamiense, Microbacterium yannicii, Microbacterium aurantiacum, Micromonospora sp., Nocardia sp., S. cinereorectus, S. cinnabarinus, Streptomyces sp., Streptomyces violascens, S. noboritoensis, Streptoverticillium sp., and Thermoactinomycetes sp.
17.3.3.3 Cyanobacteria
Similar to bacteria, the Cyanobacteria sp. has also the property to mobilize bound phosphates like FePO4, Ca3(PO4)2, AlPO4, and (Ca5(PO4)3OH). The bound P seems to be released by different mechanisms like organic acid production, chelation, dissimilatory reduction, and enzymatic solubilization or might be involving more than one mechanism (N. Kishore et al. 2015). These are few Cyanobacteria being studied for their ability to solubilize bound P: Anabaena, Calothrix braunii, Hapalosiphon fontinalis, Nostoc sp., Scytonema, Scytonema cincinnatom, Tolypothrix, Tolypothrix tenuis, Tolypothrix ceylonica, Westiellopsis prolifica, and Phormidium sp.
17.3.3.4 Protozoa and Other Mesofauna
Protozoa present in the soil are dependent upon the ingestion of some bacterial species like Aerobacter, Bacillus, Agrobacterium, Escherichia, Pseudomonas, and Micrococcus and to their protoplasm. They are well known to have the capability to assimilate soluble minerals and increase P bioavailability. They represent a major mechanism for regulating fungal and bacterial members in the soil and ultimately influences the soil P cycle (Alphei et al. 1996).
Mesofauna are responsible for enhancing P cycling and availability in a range of soils; however, they are difficult to handle practically; therefore, they are applied in agricultural soil directly for nutrient availability (Lopez-Hernandez et al. 1993). Several PSMs present in the soil don’t have enough population to compete with other bacterial community that are already established in rhizospheric zone. In consequence, the amount of P released by them is generally inadequate for proper in situ plant growth. Therefore, the application of potential microorganism at higher doses than that of the normally available microbial community can be improve methods for plant growth improvement.
17.4 The Mechanism of Rock Phosphate Solubilization
Phosphate-solubilizing microorganisms (PSMs) are able to solubilize insoluble inorganic phosphate compounds, like tricalcium phosphate, dicalcium phosphate, hydroxyapatite, and rock phosphate through various mechanisms (Goldstein 1986).
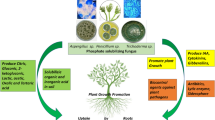
In general the accepted major mechanism for mineral phosphate solubilization is the synthesis and secretion of organic acids by soil microorganisms (Halder et al. 1990) either by (i) decreasing the level of pH, (ii) by enhancing the cation chelation that are bound to P, (iii) by competing with P for adsorption sites on the soil, or (iv) by the formation of soluble complexes with metal ions association with insoluble P (Ca, Al, Fe) and thus releasing P (Omar 1998). The conceptual diagram of RP solubilization is presented in Fig. 17.1.
The mechanism of rock phosphate solubilization (Arcand and Schneider 2006)
Many studies have been done to investigate the ability of phosphate in microbes in solubilizing insoluble rock phosphate in a pure liquid culture medium (Gupta et al. 2010; Bhattacharjya et al. 2019). The bio-solubilization of inorganic rock phosphate in liquid medium by microbial species is often due to the excretion of organic acids (Table 17.1). Microorganism-secreted organic acids are rich sources of H+ ion; this ion dissolves the mineral phosphate and converts it into bioavailable phosphate for the utilization of green plant. Panhwar et al. (2013) and Gomes et al. (2014) found a significant negative correlation between the final culture medium pH and the concentration of soluble phosphate. This negative correlation indicates that rock phosphate solubilization is a consequence of the decrease in pH of the medium due to the production of organic acids. Yadav et al. (2017) also reported that organic acids could be generated in the cellular level to facilitate P solubilization from mineral phosphates in elevated quantity by the supplementation of both protons and metal-complexing organic acid anions, which might influence the microbial community in turn.
17.5 Agronomic Implications of Microbial-Mediated RP Solubilization
Due to the insolubility of rock phosphate, it becomes insufficient for the crop to uptake required the amount of soluble P. The PSM utilization can improve the crop yields up to 70% (Verma 1993). Several authors reported enhanced plant growth and higher uptake of P through the application of rock phosphate with PSMs to soils (Rachewad et al. 1992a, b). It has also been reported that the combined incorporation of PSB and arbuscular mycorrhiza results in higher uptake of both the native P from soil and the P from rock phosphate (Goenadi and Sugiarto 2000). In alfalfa plant the accumulation of N and P with increased biomass was seen, with the inoculation of AM fungi added with RP, in field conditions (Barea et al. 2002a, b). The application of potent PSMs increases the accessibility of soluble P and enhances plant growth by improving the biological nitrogen fixation (Kucey et al. 1989; Ponmurugan and Gopi 2006). Abbasi et al. (2013) stated that the combination of organic waste, PSB with RP, has shown significant increase in agronomic effectiveness of RP and improved soil fertility. The application of RP with these combinations would display favorable results, which will minimize the utilization of high-cost P fertilizers.
17.6 Future Prospects
The application of RP as a P fertilizer shows greater potential for becoming extensive once the bioavailability of P from RP sources can be enhanced to a range, in which there should be a significant quantity of crop yields and plant tissue P content. In the establishment of sustainable soil management systems, there is every reason to believe that the PSM as biofertilizers will likely to improve their utilization. The main focus of the agricultural produce is the consumers’ health and the quality and nutritional value of those products. So in order to reach the consumer requirements, the employment of PSM as biofertilizers can be a preferable option to increase food production without imposing any health hazard and also to conserve the environment. Further research is required for the comprehensive study of in vitro RP solubilization to elucidate the exact biochemical mechanism involved in the release of P from insoluble RP in liquid broth, continue gaining an understanding about PSM, and transfer this knowledge into a form that can easily be utilized by farmers.
17.7 Conclusion
Phosphorus is one of the essential mineral macronutrient for the maintenance of plant growth regulation. Farmers need to apply chemical phosphorus fertilizer in a timely manner for the proper growth and to avoid P deficiency of plants. The efficacy of phosphate fertilizers is scanty due to their poor mechanism of fixation in both acidic and alkaline soils, and it requires a greater input which can’t be afforded by the farmers of the developing nations. The appliance of RP as a phosphatic fertilizer has the probability to become well-known once the bioavailability of P from RP sources can be improved to an extent so that the total crop yields and P content in plant tissue are increased significantly. The biological mechanism for microbe-induced RP solubilization represents innovative solutions for creating P from RP highly accessible to plant. Technologies show the potential to enhance dissolution of indigenous sources of RP and transform these resources into more agronomically effective P fertilizers. Principal mechanisms of RP solubilization is the lowering of pH by organic acids produced from microbes. More study is required to enhance the performance of phosphate-solubilizing microbes (PSMs) as microbial inoculants to increase the plant’s nutrition. PSM technology incorporated with RP can contribute to low-energy input farming systems and a cleaner environment. Although there an undoubtable potential clearly exists for developing such kind of inoculants with RP, their widespread application remains limited by a poor understanding of population dynamics and microbial ecology in the soil and by the inconsistent performance over an extent of environments. Therefore, there is a need to develop microbial-mediated RP solubilization technologies specific to various regions, and approach should be taken to make the farmers understand in a short duration of time.
References
Abbasi MK, Mansha S, Rahim N, Ali A (2013) Agronomic effectiveness and phosphorus utilization efficiency of rock phosphate applied to winter wheat. Agron J 6:1606–1612
Ahmad M, Nadeem SM, Naveed M et al (2016) Potassium-solubilizing bacteria and their application in agriculture. Springer, New Delhi, pp 293–313. https://doi.org/10.1007/978-81-322-2776-2_21
Akintokun AK, Akande GA, Akintokun PO et al (2007) Solubilization of insoluble phosphate by organic acid-producing fungi isolated from Nigerian soil. Int J Soil Sci 2:301–307
Alam S, Khalil S, Ayub N (2002) In vitro solubilization of inorganic phosphate by phosphate solubilizing microorganism (PSM) from maize rhizosphere. Intl J Agric Biol 4:454–458
Alphei J, Bonkowski M, Scheu S (1996) Protozoa, Nematoda and Lumbricidae in the rhizosphere of Hordelymus Europaeus (Poaceae): faunal interactions, response of microorganisms and effects on plant growth. Oecologia 1:111–126
Antunes V, Cardoso EJBN (1991) Growth and nutrient status of citrus plants as influenced by mycorrhiza and phosphorus application. Plant Soil 1:11–19
Arcand MM, Schneider KD (2006) Plant-and microbial-based mechanisms to improve the agronomic effectiveness of phosphate rock: a review. An Acad Bras Cienc 4:791–807
Bahadur I, Meena VS, Kumar S (2014) Importance and application of potassic biofertilizer in Indian agriculture. Int Res J Biol Sci 3:80–85
Barea JM, El-Atrach F, Azcon R (1991) The role of VA mycorrhizas in improving plant N acquisition from soil as assessed with 15N. The use of stable isotopes in plant nutrition. In: Fitton C (ed) Soil fertility and environmental studies. Joint AIEA, FAO, Division, Vienna, pp 677–808
Barea J, Toro M, Orozco M et al (2002a) The application of isotopic (32P and 15N) dilution techniques to evaluate the interactive effect of phosphate-solubilizing rhizobacteria, mycorrhizal fungi and Rhizobium to improve the agronomic efficiency of rock phosphate for legume crops. Nutr Cycl Agroecosyst 63:35–42
Barea JM, Azcón R, Azcón-Aguilar C (2002b) Mycorrhizosphere interactions to improve plant fitness and soil quality. Antonie Van Leeuwenhoek 1-4:343–351
Bhattacharjya S, Adhikari T, Kundu S et al (2019) Evaluation of microbial solubilisation of nano rock phosphate. Int J Curr Microbiol App Sci 1:1055–1069
Bolan NS (1991) A critical review on the role of mycorrhizal fungi in the uptake of phosphorus by plants. Plant Soil 2:189–207
Bürkert B, Robson A (1994) 65Zn uptake in subterranean clover (Trifolium subterraneum L.) by three vesicular-arbuscular mycorrhizal fungi in a root-free sandy soil. Soil Biol Biochem 9:1117–1124
Cao RX, Ma LQ, Chen M et al (2003) Phosphate-induced metal immobilization in a contaminated site. Environ Pollut 1:19–28
Cao X, Wahbi A, Ma L et al (2009) Immobilization of Zn, Cu and Pb in contaminated soils using phosphate rock and phosphoric acid. J Haz Mat 164:555–564
Coutinho FP, Felix WP, Yano-Melo AM (2012) Solubilization of phosphates in vitro by Aspergillus spp. and Penicillium spp. Ecol Eng 42:85–89
Cunningham JE, Kuiack C (1992) Production of citric and oxalic acids and solubilization of calcium phosphate by Penicillium bilaii. Appl Environ Microbiol 58(5):1451–1458
Duponnois R, Colombet A, Hien V et al (2005) The mycorrhizal fungus glomus intraradices and rock phosphate amendment influence plant growth and microbial activity in the rhizosphere of Acacia holosericea. Soil Biol Biochem 8:1460–1468
Duponnois R, Kisa M, Plenchette C (2006) Phosphate-solubilizing potential of the nematophagous fungus Arthrobotrys oligospora. J Plant Nutr Soil Sci 2:280–282
Elliott LF, Lynch JM (1995) Am J Altern Agric 2:50–73
Fabre B, Armau E, Etienne G et al (1988) A simple screening method for insecticidal substances from actinomycetes. J Antibiot 41:212–219
Fankem H, Nwaga D, Deubel A et al (2006) Occurrence and functioning of phosphate solubilizing microorganisms from oil palm tree (Elaeis guineensis) rhizosphere in Cameroon. Afr J Biotechnol 5:24
Frioni L (2006) Microbiologı’a ba’sica, ambiental y agrı’cola. Universidfad de la Repu’blica, Facultad de Agronomı’a. Montevideo, Uruguay
Goenadi DH, Sugiarto Y (2000) Bioactivation of poorly soluble phosphate rocks with a phosphorus-solubilizing fungus. Soil Sci Soc Am J 3:927–932
Goldstein AH (1986) Bacterial solubilization of mineral phosphates: historical perspective and future prospects. Am J Altern Agric:51–57
Gomes EA, Silva UC, Marriel IE, Oliveira CA et al (2014) Rock phosphate solubilizing microorganisms isolated from maize rhizosphere soil. Rev Bras Milho Sorgo 13:69–81
Guissou T, Guinko S, Plenchette C et al (2001) Mobilisation des phosphates naturels de Kodjari par des jujubiers (Ziziphus mauritiana Lam.) mycorhize’s dans un sol acidifie´ avec de la tourbe. Fruits 56:261–269
Gupta N, Das S, Basak UC (2010) TCP and rock phosphate solubilization by mangrove fungi grown under different pH and temperature in liquid culture. J Agri Technol 3:421–428
Halder AK, Mishra AK, Bhattacharyya P et al (1990) Solubilization of rock phosphate by rhizobium and Bradyrhizobium. J Gen Appl Microbiol 2:81–92
Hamdali H, Hafidi M, Virolle MJ et al (2008) Rock phosphate-solubilizing Actinomycetes: screening for plant growth-promoting activities. World J Microbiol Biotechnol 11:2565–2575
Indian Minerals Yearbook (2016) Mineral reviews 55th edition, marble, Government of India, Ministry of Mines, Indian Bureau of Mines (Part III)
Ivanova RP, Bojinova DY, Gruncharov IN, Damgaliev DL (2006) The solubilization of rock phosphate by organic acids. Phosphorous Silicon Relat Elem 181(11):2541–2554
Jat SR, Patel BJ, Shivran AC et al (2013) Effect of phosphorus and Sulphur levels on growth and yield of cowpea under rainfed conditions. Ann Plant Soil Res 2:114–117
Kennedy IR, Choudhury ATMA, Kecskés ML (2004) Non-symbiotic bacterial diazotrophs in crop-farming systems: can their potential for plant growth promotion be better exploited? Soil Biol Biochem 8:1229–1244
Kishore N, Pindi PK Reddy SR (2015) Phosphate-solubilizing microorganisms: a critical review. Plant biology and biotechnology. pp. 307–333
Kohn MJ, Rakovan J, Hughes JM (2018) Phosphates: geochemical, geobiological and materials importance, vol 8. Walter de Gruyter GmbH & Co KG
Kucey RMN (1983) Phosphate-solubilizing bacteria and fungi in various cultivated and virgin Alberta soils. Can J Soil Sci 4:671–678
Kucey RMN, Janzen HH, Leggett ME (1989) Microbially mediated increases in plant-available phosphorus. Adv Agron 42:199–228
Kumar A, Bahadur I, Maurya BR et al (2015) Does a plant growth-promoting rhizobacteria enhance agricultural sustainability? J Pure Appl Microbiol 9:715–724
Lapeyrie F (1988) Oxalate synthesis from soil bicarbonate by the mycorrhizal fungus Paxillus involutus. Plant Soil 110:3–8
López-Arredondo DL, Herrera-Estrella L (2012) Engineering phosphorus metabolism in plants to produce a dual fertilization and weed control system. Nat Biotechnol 9:889–893
Lopez-Hernandez D, Lavelle P, Fardeau JC et al (1993) Phosphorus transformations in two P-sorption contrasting tropical soils during transit through Pontoscolex corethrurus (Glossoscolecidae: Oligochaeta). Soil Biol Biochem 6:789–792
Ma LQ, Rao GN (1999) Aqueous Pb reduction in Pb-contaminated soils by Florida phosphate rocks. Water Air Soil Pollut 1-2:1–16
Maurya BR, Meena VS, Meena OP (2014) Influence of Inceptisol and Alfisol’s potassium solubilizing bacteria (KSB) isolates on release of K from waste mica. Vegetos 27:181–187
Meena OP, Maurya BR, Meena VS (2013) Influence of K-solubilizing bacteria on release of potassium from waste mica. Agric Sust Dev 1:53–56
Meena RK, Singh RK, Singh NP, Meena SK, Meena VS (2016) Isolation of low temperature surviving plant growth-promoting rhizobacteria (PGPR) from pea (Pisum sativum L.) and documentation of their plant growth promoting traits. Biocatal Agric Biotechnol 4:806–811
Nahas E (1996) Factors determining rock phosphate solubilization by microorganisms isolated from soil. World J Microbiol Biotechnol 6:567–572
Omar SA (1998) The role of rock-phosphate-solubilizing fungi and vesicular - arbuscular – mycorrhiza (VAM) in growth of wheat plants fertilized with rock phosphate. World J Microb Biot 14:211–218
Ouahmane L, Thioulouse J, Hafidi M et al (2007) Soil functional diversity and P solubilization from rock phosphate after inoculation with native or allochthonous arbuscular mycorrhizal fungi. For Ecol Manag 1-3:200–208
Panhwar QA, Jusop S, Naher UA (2013) Application of potential phosphate-solubilizing bacteria and organic acids on phosphate solubilization from phosphate rock in aerobic rice. Sci World J
Parewa HP, Yadav J, Rakshit A (2014) Plant growth promoting rhizobacteria enhance growth and nutrient uptake of crops. Agric Sustain Dev 2:101–116
Patel DK, Archana G, Kumar GN (2008) Variation in the nature of organic acid secretion and mineral phosphate solubilization by Citrobacter sp. DHRSS in the presence of different sugars. Curr Microbiol 2:168–174
Ponmurugan P, Gopi C (2006) Distribution pattern and screening of phosphate solubilizing bacteria isolated from different food and forage crops. J Agron 4:600–604
Prakash S, Verma JP (2016) Global perspective of potash for fertilizer production. Potassium solubilizing microorganisms for sustainable agriculture. Springer, New Delhi, pp 327–331
Rachewad SN, Raut RS, Malewar GU et al (1992a) Effects of phosphate solubilizing biofertilizer on biomass production and uptake of phosphorous by sunflower. J Maharashtra Agric Univ 17:480–481
Rachewad SN, Raut RS, Malewar GU et al (1992b) Effects of phosphate solubilizing biofertilizer on biomass production and uptake of phosphorus by sunflower. J Maharashtra Agric Univ 17:480–480
Schreiner RP, Mihara KL, Daniel M et al (2003) Mycorrhizal fungi influence plant and soil functions and interactions. Plant Soil 188:199–209
Sharma K, Dak G, Agrawal A et al (2007) Effect of phosphate solubilizing bacteria on the germination of Cicer arietinum seeds and seedling growth. J Herbal Med Toxicol 1:61–63
Sharma SB, Sayyed RZ, Trivedi MH et al (2013) Phosphate solubilizing microbes: sustainable approach for managing phosphorus deficiency in agricultural soils. Springerplus 1:587
Shigaki F, Sharpley AN, Prochnow LI (2006) Animal-based agriculture, phosphorus and management and water quality in Brazil: options for the future. Sci Agric 63:194–209
Solans M, Vobis G (2003) Actinomycetes saprofíticos asociados a la rizósfera y rizoplano de Discaria trinervis. Ecol Austral 1:97–107
Son HJ, Park GT, Cha MS, Heo MS (2006) Solubilization of insoluble inorganic phosphates by a novel salt-and pH-tolerant Pantoea agglomerans R-42 isolated from soybean rhizosphere. Bioresour Technol 2:204–210
Sperber JI (1958) Solution of apatite by soil microorganisms producing organic acids. Aust J Agric Res 6:782–787
Tisdall JM (1994) Possible role of soil microorganisms in aggregation in soils. Plant Soil 1:115–121
Vassilev N, Vassileva M, Nikolaeva I (2006) Simultaneous P-solubilizing and biocontrol activity of microorganisms: potentials and future trends. Appl Microbiol Biotechnol 2:137–144
Venkateswarlu B, Rao AV, Raina P (1984) Evaluation of phosphorus solubilisation by microorganisms isolated from Aridisols. J Indian Soc Soil Sci 2:273–277
Verma LN (1993) Organics in soil health and crop production. In: Thampan PK (ed) Tree Crop Development Foundation, Cochin, pp 151–184
Vyas P, Gulati A (2009) Organic acid production in vitro and plant growth promotion in maize under controlled environment by phosphate-solubilizing fluorescent pseudomonas. BMC Microbiol 1:174
Whitelaw MA (2000) Growth promotion of plants inoculated with phosphate solubilizing fungi. Adv Agron 69:99–151
Yadav H, Fatima R, Sharma A (2017) Enhancement of applicability of rock phosphate in alkaline soils by organic compost. Appl Soil Ecol 113:80–85
Yang J, Mosby DE, Casteel SW et al (2001) Lead immobilization using phosphoric acid in a smelter-contaminated urban soil. Environ Sci Technol 17:3553–3559
Yazdani M, Bahmanyar MA, Pirdashti H et al (2009) Effect of phosphate solubilization microorganisms (PSM) and plant growth promoting rhizobacteria (PGPR) on yield and yield components of corn (Zea mays L.). World Acad Sci Eng Technol 49:90–92
Yoon JK, Cao X, Ma LQ (2007) Application methods affect phosphorus-induced lead immobilization from a contaminated soil. J Environ Qual 2:373–378
Acknowledgments
The INSPIRE fellowship (DST/INSPIRE/03/2015/000438, IF160155) to AcSIR PhD student, Ms. Rojali Maharana for her PhD work from the Department of Science and Technology, and the Government of India are highly acknowledged. The authors were also thankful to Prof. S. Basu, the Director of CSIR-IMMT, Bhubaneswar, Odisha, India, for providing necessary lab facilities to carry out the work.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Maharana, R., Singh, B.S.M., Mandal, K., Dhal, N.K. (2022). Microbial-Mediated Mechanism to Improve Rock Phosphate Solubilization and Its Agronomic Implications. In: Mulla, S.I., Bharagava, R.N. (eds) Enzymes for Pollutant Degradation . Microorganisms for Sustainability, vol 30. Springer, Singapore. https://doi.org/10.1007/978-981-16-4574-7_17
Download citation
DOI: https://doi.org/10.1007/978-981-16-4574-7_17
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-4573-0
Online ISBN: 978-981-16-4574-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)