Abstract
Obesity is multifactorial and complex. Remarkable progress has been made recently in search for polygenic obesity through genome-wide association study (GWAS), but biology of polygenic effects on obesity is largely poor. This review summarizes the available evidence and provides an overview of the links between ALDH2 variants and adiposity, which were firstly and mainly derived from studies of polygenic obesity and also indirectly investigated by using cell lines and mice. The genetic association studies have observed consistent associations of ALDH2 variants with obesity-related traits including BMI, waist circumference (WC), waist-to-hip ratio (WHR), and visceral fat accumulation. In consideration of ALDH2 variants with enzyme activity and alcohol consumption behavior in physiological mechanism studies, we proposed a model by which the physiological and behavioral consequences of alcohol consumption serve as an intermediary process between polymorphisms in ALDH2 and obesity.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Obesity, defined as an excessive fat accumulation, has increased at an alarming pace around the world. For the years 2013–2014, more than one-third (37.7%) of US adults have obesity, and the number is projected to 51% in 2030 [1]. Not only that, obesity is also a major heath concern in developing countries, where almost two in three of the world’s people live with obesity. The rate of obesity has tripled since 1980 in the Middle East, the Pacific Islands, and China [2]. Obesity-related diseases such as type 2 diabetes, stroke, coronary heart disease, and some cancers have been the leading cause of disability and death. Clinically, overweight and obesity are quantified through the surrogate measure of body mass index (BMI), calculated as weight divided by the square of height. Individuals with a BMI of ≥28 kg/m2 are considered obese and those with a BMI of 25 kg/m2 to 29.9 kg/m2 considered overweight according to World Health Organization (WHO) criteria [3]. The given BMI values indicate a higher percentage of body fat and metabolic disease risk for Asian populations than European populations; therefore, some Asian countries have revised the definition of obesity to adjust for ethnic differences [4]. In China, overweight and obesity are, respectively, identified as 24 kg/m2≤ BMI <28 kg/m2 and BMI ≥28 kg/m2 [5]. Central obesity (also known as visceral, android, apple-shaped or upper body obesity, abdominal obesity) can be clinically assessed by waist circumference (WC) which is highly associated with intra-abdominal fat. International Diabetes Federation (IDF) consensus defined central obesity in Europeans as a WC of ≥94 cm in males and ≥ 80 cm in females. Lower WC cutoffs are proposed for some ethnic groups (e.g., Chinese, Southeast Asians) [6]. Some indices corresponding to fat distribution, including waist-to-hip ratio (WHR), visceral fat are also used to assess central obesity.
Obesity is a product of the interaction between genetic factors and lifestyle risk factors (like smoking, drinking, lack of physical activity, sedentary, and Western diet). Even though surrounded by “obesogenic environment,” a certain number of individuals could maintain their weight, suggesting genetic factors play a critical role in regulation of obesity. The advent of genome technology innovation (e.g., microarray and sequencing) has allowed far more detailed investigation of genetic factors than previously known. Approximately 150 variants linked to obesity (e.g., BMI, WC, and WHR) have been identified through genome-wide association studies (GWAS) [7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24]. To be frustrated, little is known about the underlying mechanism by which those surprisingly high numbers of obesity-related variants imposed risk to obesity or body fat distribution. The interpretation of genomics to clinical care and public health is not enough, which requires the knowledge of genetic basis for obesity and interactions with health behaviors. Population research on the gene-environment interaction, which means that the sensitivity to environmental influences is regulated modulated by genetic factors, was still lacking.
The p.Glu504Lys (c.1510G>A, rs671) ALDH2 missense variant is among numerous variants linked to obesity which reached the genome-wide significance. Aldehyde dehydrogenase 2 encoded by ALDH2 is well known for most efficient enzyme of alcohol oxidation in the liver and other organs. Since alcohol consumption is also a public health concern, we focused on aldehyde dehydrogenases (ALDHs) genetic polymorphisms in this review and elucidated a genetic link to a common behavior and health.
2 Aldehyde Dehydrogenases
Human aldehyde dehydrogenases superfamily is a group of oxidizing enzymes responsible for the metabolism of endogenous and exogenous aldehydes. There are 19 functional isoforms with a wide range of tissue distribution, and some of them display specific subscapular compartments [25, 26]. Amino acid sequence similarities are about 40% between families and 60% or higher between the subfamilies. While some aldehydes functioned by ALDHs play key roles in normal physiological processes including vision, embryonic development, and neurotransmission, most of aldehydes can lead to cytotoxic damage. In this case, ALDHs are regarded as detoxification enzymes and serve as an important shield from the cytotoxic damage of aldehydes by converting them to their respective carboxylic acids.
Mitochondrial ALDH2 emerges as a particularly important enzyme for the oxidation of acetaldehyde in vivo, an immediate metabolite of alcohol. Alcohol-detoxifying pathway, specifically, consists of two-step enzymatic reaction. The first step is catalyzed by enzyme alcohol dehydrogenase (ADH), which converts alcohol to acetaldehyde. The second step is mainly catalyzed by ALDH2, dehydrogenating acetaldehyde into acetate to keep low levels of circulating acetaldehyde under normal condition. Any abnormal endogenous (i.e., mutations or liver disease) and exogenous (i.e., drugs) conditions which influence the enzyme activity could lead to the accumulation of acetaldehyde, manifested by a variety of unpleasant effects such as alcohol-flushing responses, nausea, vomiting, hypotension, or rapid heart rate. These unpleasant effects may prevent individuals from consuming more alcohol. Besides, it can also serve to remove lipid peroxidation-derived aldehydes and other reactive aldehydes to protect from the damage of excessive oxidative stress. Compelling evidence indicates that ALDH2 is a key mediator of endogenous cytoprotection against ischemia injury [27, 28], gastrointestinal cancers [29, 30], late-onset Alzheimer’s disease [31, 32], and a variety of human diseases.
The importance of ALDH2 proteins in physiological or pathological processes might be best evidenced by the associations between ALDH2 functional variants and distinct disease phenotypes in humans. The p.Glu504Lys (c.1510G>A, rs671) ALDH2 is the most common single point mutation in humans. This single point mutation occurs 35–45% among East Asians (approximately 560 million) but very rare in European populations [33]. The ALDH2*2 (termed A allele) carriers have a lower ALDH2 enzymatic activity. ALDH2*1/*2 heterozygotes are expected to have dramatically lower than 50% of the wild-type’s enzymatic activity and ALDH2*2/*2 homozygotes have <1–4% of the wild-type activity. Several large meta-analyses identified that carriers of the highly active ALDH2*1 allele (or G allele) had an increased risk of alcoholism. rs886205 is another variant in the promoter region of ALDH2 gene and was linked to ALDH2 activity through changing transcriptional activity in European populations.
3 The Associations Between Obesity and ALDH2 Variants
3.1 What’s for the GWAS of Obesity?
Multiple measurements including BMI, WC, WHR, and visceral fat area (VFA) are applied to quantify the degree of obesity. BMI, is a simple but standard measurement for overall obesity. Great advances in identification of variants linked to obesity can be largely attributed to the strategy of GWAS. The attempts to identify BMI-related variants are considered to facilitate some patterns of discovery for neuronal regulation in overall obesity [12, 16]. WC and WHR are considered as simple and commonly used markers for central obesity. Besides, other indices corresponding to fat distribution imaged by MRI or CT technology are superior to WC and WHR in terms of distinguishing between visceral fat and abdominal subcutaneous fat. The important thing for the central obesity-related variants has proven vital to elucidate the signals either shared with overall obesity or specific to central obesity.
3.2 The Link Between Obesity and ALDH2 Variants
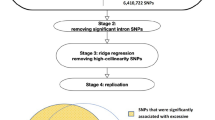
The involvement of ALDH2 in obesity and fat distribution was first suggested by GWAS in East Asian populations. So far, a total of three large-scale GWAS analyses and one replication study were performed among East Asian populations (as shown in Table 1) [21, 22, 34, 35]. The first GWAS analysis, published in Nature Genetics in 2009, uncovered a novel locus rs2074356 affecting WHR that reached genome-wide significance in Korean populations (8842 and 7861 samples in stages 1 and 2, respectively) [34]. This locus is mapped to chromosome 12q24 in the 24th intron of the C12orf51 and in moderate linkage disequilibrium with the rs671 at ALDH2 in East Asian populations (r2 = 0.58 in JPT and CHB). Then, two enlarged GWAS of obesity among East Asian populations were performed. The latter GWAS was conducted by Wen et al. to test the association of BMI with 2.5 million genotyped or imputed SNPs in Asian population in 2014 [21]. The significant associations of the two related SNPs in 12q24 region (rs671 at ALDH2, rs12229654 at MYL2, r2 = 0.58) with BMI has been primarily identified in a population of 86,757 individuals and replicated in an independent sample of 11,233 and 23,454 individuals, respectively. The carriers of G allele (namely, highly active ALDH2*1 allele) conferred higher BMI compared with non-carriers. There was substantial overlapping between overall obesity and central obesity and previously reported locus near ALDH2 for WHR was not adjusted for BMI. Therefore, Wen et al. conducted a new round of meta-analyses to test the associations of WC and WHR with 2.5 million SNPs among individuals of East Asian ancestry in 2016 (n = 53,052 and 48,312 for WC and WHR, respectively) [22]. They confirmed the effects of rs671 in ALDH2 on WC and WHR before or after adjusting for BMI. Even though WC and WHR are considered good markers for central obesity, they cannot distinguish between visceral fat and abdominal subcutaneous fat directly. The genetic study for more accurate proxy of central obesity may reveal novel variants that are not necessarily discovered when WC and WHR are used as the outcomes. The study by Wang et al. in 2016, consisted of 2958 subjects in FAt Distribution and diseasE (FADE) cohort from Chinese Han populations with refined visceral fat area (VFA) and subcutaneous fat area (SFA) imaged by MRI, explored whether ALDH2 variants directly imposed effects on visceral fat or subcutaneous fat deposit [35]. They demonstrated that rs671 at ALDH2 was associated with visceral fat accumulation specifically. The carriers of G allele (highly active ALDH2*1 allele) confers more visceral fat accumulation compared with non-carriers. All of studies mentioned above indicated that ALDH2 variants have substantial influence on obesity, especially for visceral fat accumulation.
3.3 Subgroup Analysis Stratified by Alcohol Composition
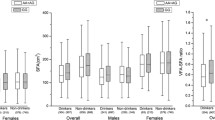
rs671 in ALDH2 was previously demonstrated a robust association with alcohol consumption in genetic and functional studies. To evaluate the underlying effect of alcohol consumption on the association between ALDH2 variants and obesity-related traits, the subgroup analyses were conducted but produced inconsistent results (as shown in Table 2). Two studies mentioned above by Wen et al. mainly involved a total of 6918 Chinese individuals with data of alcohol consumption available from Shanghai Genome-Wide Association Studies (SGWAS). The information about alcohol consumption was collected using a standard questionnaire. The association of rs671 with BMI was mainly observed among nondrinkers in SGWAS cohort, suggesting an antagonistic effect of alcohol consumption on the ALDH2-BMI association. Nonetheless, WC-increasing effect or WHR-increasing effect conferred by rs671 G allele failed to be replicated among drinkers or nondrinkers separately. In FADE cohort, Wang et al. had access to individual data of alcohol consumption among a total of 2937 Chinese Han individuals. Drinkers were defined if subjects had ever consumed alcohol in their lifetime including chance drinkers (less than three times in every week) and regular drinkers (equal or more than three times in every week), whereas nondrinkers were those who never drank in their lifetime. The effects of rs671 on BMI, WC, and WHR with or without adjustment of BMI were mainly observed among drinkers. More importantly, the effect of ALDH2 rs671 on VFA and VFA/SFA was also observed among drinkers, indicating association of rs671 with obesity-related traits mediated by alcohol consumption. Wang et al. also conducted subgroup analysis which draws a distinction between chance drinkers and regular drinkers to strengthen their findings. The results showed that the nominal associations of ALDH2 variant with WC and VFA after adjustment of BMI were restricted to regular drinkers specifically but did not observe associations in chance drinkers. The mixed results between SGWAS and FADE study might be explained by the difference in study design. Although the definition of alcohol consumption in SGWAS and FADE study was approximately similar, the proportion of alcohol consumption was different (drinkers vs nondrinkers = 1:13 in SGWAS and 2:3 in FADE study). The possibility that BMI- or WC- or WHR-increasing effects conferred by rs671 G allele were not observed because of small sample size of drinkers in SGWAS cannot be excluded (Table 3).
3.4 Gender Difference
Generally, males have more visceral fat deposit, whereas females have more subcutaneous fat deposit before menopause. Taking into account this heterogeneity in fat distribution in both genders, the subgroup analyses in males and females were performed separately. The results showed more pronounced associations in males than in females, and there is evident heterogeneity in both gender for obesity-related traits (Table 4). Additionally, Wang et al. also tested sex difference among drinkers and nondrinkers separately. The nominal associations between the ALDH2 variant and visceral fat accumulation were restricted to male drinkers specifically, and the effects of rs671 in ALDH2 on VFA and SFA revealed a borderline sex-related significance among overall drinkers. Note that the male to female ratio was not balanced between drinkers and nondrinkers, that is, both of SGWAS and FADE study have more male drinkers than female drinkers. We believe that the statistical power of rs671 with central obesity-related traits in female drinkers was inadequate due to the relatively small sample size of female drinkers. The studies with comparable amount of males and females regarding to alcohol consumption to test whether alcohol consumption affect obesity or visceral fat accumulation in a sex-dependent manner are warranted.
3.5 Ethnicity
Varied ancestry populations differed in fat distribution and underlying genetic background [36, 37]. For a given amount of BMI, Asian populations seem to be prone to the accumulation of visceral fat compared to European populations [38]. An indisputable fact is that large-scale obesity GWAS that include Asian and other non-European populations are more likely to provide insight into different genetic architectures and identify evidence for specific causal genes [19, 20]. 12q24 region is polymorphic only in East Asians, and it is still unknown to what extent ALDH2 gene contributed to the risk of visceral fat accumulation among East Asian populations exactly.
4 A Possible Model for ALDH2-Induced Obesity
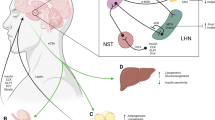
The ALDH2 gene encodes the mitochondrial ALDH2, a critical enzyme not only for ethanol oxidation during alcohol ingestion but also for several endogenous aldehydes such as propionaldehyde, butyraldehyde, and 4-hydroxykenals (4-HNE) originated from mitochondrial production of reactive oxygen species (ROS). Therefore, two plausible mechanisms have been postulated for the link between ALDH2 variants and obesity including ethanol/acetaldehyde metabolism and endogenous bioactive aldehyde metabolism.
4.1 Proposed Mechanism One: Ethanol/Acetaldehyde Metabolism
ALDH2 encodes a functionally ALDH enzyme subunit that leads to impaired the removal of acetaldehyde, a toxic byproduct of ethanol metabolism. The A allele (also ALDH2*2 allele), common in 30–50% of individuals of northeast Asian descent, is associated with a significantly reduced likelihood of heavy drinking and alcohol dependence due to a unpleasant symptoms like flushing, tachycardia, and nausea. The physiological and behavioral consequences of alcohol consumption serve as an intermediary process between ALDH2 genetics and obesity. In other words, rs671 in ALDH2 may influence the obesity and visceral fat accumulation by affecting alcohol consumption behavior, with A allele carriers having lower BMI and visceral fat depots due to lower alcohol consumption. A similar conclusion was drawn from a Mendelian randomization analysis among Korean population regarding rs671 in ALDH2 as an instrumental variable, which indicated the marked positive effects of alcohol intake (as indexed by the absence of alcohol flushing and the ALDH2 rs671 GG genotype) on blood pressure [39].
Recent evidence indicated that excess drinking was consistently associated with weight gain or increased waist circumference, whereas light-to-moderate alcohol consumption is not linked to adiposity gain [40,41,42]. Further, Molenaar et al. [43] reported that intake of large amounts of alcohol was associated with decrease of subcutaneous adiposity in females and increase of visceral adiposity in males from Framingham Offspring Study. A common trend appears to be that it is multifactorial and involves cross talk among various organs and tissues.
In food, the energy content is derived from macronutrients (carbohydrate, fat, protein, and alcohol). Both of carbohydrate and protein provide 4 kcal per gram and fat provides 9 kcal per gram. Alcohol is a calorically dense substance and produces 7.1 kcal (29 kJ) per gram [44], which should theoretically play a critical role in energy balance. The findings about the effects of alcohol-derived energy on body mass and fat deposit are debatable. A line of evidence indicates that alcohol-derived calorie consumption seems to supplement food-derived energy [45, 46], and individuals with excessive drinking appear to increase adiposity indices among varied populations [47,48,49,50]. A diet recall study in 951 healthy males from Koreans, which recorded the dietary intake of energy from food and alcohol, showed that total energy intake increased with higher alcohol consumption and further observed that there was an increase in visceral fat accumulation with either decrease or no change in subcutaneous fat accumulation [51]. Another study, however, found that chronic and moderate alcohol intake was likely to lead to a decrease in macronutrients intake to compensate for ethanol calories [52]. A certain number of studies have examined the short-term effect of alcohol consumption on appetite control and feeding behavior. In these studies, alcohol may amplify individuals’ perception of appetite in response to food stimuli but fail to produce sufficient signals on satiety or enhance the rewarding effects [53,54,55]. Besides, several neurotransmitters including γ-aminobutyric acid (GABA), opioid and dopaminergic system were considered to be vital for motivational effect of alcohol on stimulation of appetite [56, 57]. Genomic-based evidence showed that compared to individuals with the GG genotype (ALDH2*1/*1 homozygotes), those with the inactive A allele (ALDH2*2) reported greater negative alcohol expectancies, and lower risk of alcohol abuse, indicating that differences in alcohol metabolism were reported to influence how drinking events are experienced, interpreted, and stored in memory in central nervous system [58].
Abundance of data showed that higher amounts of daily alcohol intake were positively associated with visceral adiposity [43, 59, 60]. The correlation between alcohol intake and fat distribution was likely mediated by plasma androgens at least in part or fatty liver, which can result in hepatic insulin resistance and subsequent weight gain. Several studies, however, have been rather inconsistent, reporting no association between alcohol consumption and visceral fat [61, 62]. Genomic-based evidence indicated that ALDH2 variants strongly correlated with obesity, especially for visceral fat accumulation. Further studies are required to validate the association and get understanding of the mechanism process of ALDH2 which manifested as visceral adiposity.
Moreover, increasing studies showed that excess alcohol consumption were often associated with chronic systemic inflammation status and high circulating proinflammatory cytokine levels [63,64,65] as well as high circulating cortisol levels [66, 67]. Alcohol intake may enhance cortisol secretion which changed the pattern of fat distribution, together with an increase in abdominal and hepatic fat deposition and subcutaneous adiposity lipolysis [66].
Obesity is a combination of genetic and environmental factors. These factors may act independently or they may interact with each other. Gene-environment interaction, in a statistical sense, refers to a situation in which the impacts of genes depend on the environment or the impacts of the environment depend on genotype [68]. For example, a study found that GNB3 variation interacts with physical activity to influence obesity. They reported that carriers of 825 T allele in physical active group had a 20% lower prevalence of obesity for each additional T allele, while those with the same genotype who were not physically active had a 23% greater prevalence of obesity [69]. The integration of gene-environment information is crucial to move genomic discoveries in obesity to actual behavioral interventions or medications that reduce the burden of obesity. Previous genomic-based evidence indicated the association of ALDH2 variants with obesity-related indices, which were mediated with alcohol consumption. However, the knowledge on how ALDH2 and environment interact at a biological level is crucial in fully understanding the processes of obesity or visceral fat accumulation but remains unclear currently. Research on the advance of a wide range of biological responses (e.g., energy intake, appetite control, systemic inflammation, or some hormones) after alcohol consumption in the internal environment among individuals with certain genotype is needed.
4.2 Proposed Mechanism Two: Endogenous Bioactive Aldehyde Metabolism
In the body, several degradation reactions are known to form endogenous acetaldehyde during the oxidative stress, many of which are highly reactive and toxic. Apart from alcohol metabolism, ALDH2 is also responsible for oxidizing several bioactive aldehydes (i.e., propionaldehyde, butyraldehyde, and 4-HNE). It is suggested that ALDH2 could protect against oxidative stress-related diseases such as atherosclerosis, tumors, diabetes, and acute lung injury and pulmonary arterial hypertension [70,71,72]. Recent evidence indicated that reactive oxygen species (ROS) balance was required for the physiology adipocyte function and differentiation. Yu et al. reported that ALDH2 overexpression or ALDH2 agonist Alda1 was correlated with adipocyte differentiation, mediated by signaling pathways downstream of peroxisome proliferator-activated receptor (PPAR)-γ [72]. As oxidative stress is consolidated in obesity complications such as cardiomyopathy, Wang et al. reported that ALDH2 can help preserve high-fat diet-induced obesity cardiomyopathy through a mechanism related to modulation of autophagy and SUV39H-Sirt1-dependent PGC-1α deacetylation [73].
5 Ways Ahead and Conclusions
Great advance has been made through GWAS in different ethnic groups for the discovery the susceptibility of obesity. Findings of this research in large-scale population-based studies have proven significant to advancing our knowledge of the pathways by which obesity development is modulated. Currently, evidence on quantifying the role of gene-environment interactions in the development of obesity, which are crucial for clinical or public health practice, is still lacking. Along this review, we illustrated the available evidence on ALDH2 variants and obesity and then proposed a model by which the physiological and behavioral consequences of alcohol consumption are considered to be an intermediary process between ALDH2 genetics and obesity. This integration of ALDH2 variants and environment or personal behavior is of great value: (1) shedding new light on the role of aldehyde dehydrogenases in biology process in humans, (2) improving the integration of currently uncertain data on alcohol consumption in etiology of obesity, and (3) regarded as an important way to understand the functional diversity of the numerous genetic polymorphisms for obesity and any other serious, chronic pathologies and ultimately to improve population health.
However, to explore the interpretation of genetic findings to environmental factors for obesity or fat distribution is currently in the early stage, and much of that should be validated. First, to obtain the better assessments of gene-environment interactions, alcohol type as well as consumption pattern including frequency and amount of alcohol intake should be taken into account in quantitative models. Measurement of environmental factors including behavioral and lifestyle factors appears to be less certain and complete rather than measurement of the genomics in epidemiological studies. Therefore, the advances in genomics on intervention and management of obesity will depend on the reliability of evidence obtained from epidemiological studies. Second, whether alcohol-derived metabolites per se or other endogenous reactive dehydrates play a key role in regulation of obesity should be studied since the cytoprotection of ALDH2 is considered to be a prominent function for a variety of human diseases. Another concern is that the usefulness of ALDH2-variants findings should be determined through longitudinal studies or intervention studies. Such longitudinal studies in which ALDH2 variants could be of predictive value or serve as markers to identify individuals who are at high risk of obesity are warranted. Additional research on behavioral interventions targeted to subgroups with varied genotypes is needed to understand behavioral responses to genetic information. For instance, researchers might as well examine whether health guidance based on genetic testing would be more beneficial in framing behavior of certain genotype subgroup.
Abbreviations
- 4-HNE:
-
4-hydroxykenals
- ADH:
-
alcohol dehydrogenase
- ALDHs:
-
aldehyde dehydrogenases
- BMI:
-
body mass index
- FADE:
-
FAt Distribution and diseasE
- GABA:
-
γ-aminobutyric acid
- GWAS:
-
genome-wide association studies
- IDF:
-
International Diabetes Federation
- PPAR-γ:
-
peroxisome proliferator-activated receptor γ
- ROS:
-
reactive oxygen species
- SFA:
-
subcutaneous fat area
- SGWAS:
-
Shanghai Genome-Wide Association Studies
- VFA:
-
visceral fat area
- WC:
-
waist circumference
- WHO:
-
World Health Organization
- WHR:
-
waist-to-hip ratio
References
Flegal KM et al (2016) Trends in obesity among adults in the United States, 2005 to 2014. JAMA 315(21):2284–2291
Ellulu M et al (2014) Epidemiology of obesity in developing countries: challenges and prevention. Global Epidem Obes 2(1):2
James WP (2008) The epidemiology of obesity: the size of the problem. J Intern Med 263(4):336–352
Misra A et al (2009) Consensus statement for diagnosis of obesity, abdominal obesity and the metabolic syndrome for Asian Indians and recommendations for physical activity, medical and surgical management. J Assoc Physicians India 57:163–170
Zhou BF (2002) Predictive values of body mass index and waist circumference for risk factors of certain related diseases in Chinese adults—study on optimal cut-off points of body mass index and waist circumference in Chinese adults. Biomed Environ Sci 15(1):83–96
Alberti KG, Zimmet P, Shaw J (2005) The metabolic syndrome—a new worldwide definition. Lancet 366(9491):1059–1062
Frayling TM et al (2007) A common variant in the FTO gene is associated with body mass index and predisposes to childhood and adult obesity. Science 316(5826):889–894
Loos RJ et al (2008) Common variants near MC4R are associated with fat mass, weight and risk of obesity. Nat Genet 40(6):768–775
Heard-Costa NL et al (2009) NRXN3 is a novel locus for waist circumference: a genome-wide association study from the CHARGE Consortium. PLoS Genet 5(6):e1000539
Lindgren CM et al (2009) Genome-wide association scan meta-analysis identifies three loci influencing adiposity and fat distribution. PLoS Genet 5(6):e1000508
Thorleifsson G et al (2009) Genome-wide association yields new sequence variants at seven loci that associate with measures of obesity. Nat Genet 41(1):18–24
Willer CJ et al (2009) Six new loci associated with body mass index highlight a neuronal influence on body weight regulation. Nat Genet 41(1):25–34
Heid IM et al (2010) Meta-analysis identifies 13 new loci associated with waist-hip ratio and reveals sexual dimorphism in the genetic basis of fat distribution. Nat Genet 42(11):949–960
Speliotes EK et al (2010) Association analyses of 249,796 individuals reveal 18 new loci associated with body mass index. Nat Genet 42(11):937–948
Fox CS et al (2012) Genome-wide association for abdominal subcutaneous and visceral adipose reveals a novel locus for visceral fat in women. PLoS Genet 8(5):e1002695
Locke AE et al (2015) Genetic studies of body mass index yield new insights for obesity biology. Nature 518(7538):197–206
Shungin D et al (2015) New genetic loci link adipose and insulin biology to body fat distribution. Nature 518(7538):187–196
Chambers JC et al (2008) Common genetic variation near MC4R is associated with waist circumference and insulin resistance. Nat Genet 40(6):716–718
Okada Y et al (2012) Common variants at CDKAL1 and KLF9 are associated with body mass index in east Asian populations. Nat Genet 44(3):302–306
Wen W et al (2012) Meta-analysis identifies common variants associated with body mass index in east Asians. Nat Genet 44(3):307–311
Wen W et al (2014) Meta-analysis of genome-wide association studies in East Asian-ancestry populations identifies four new loci for body mass index. Hum Mol Genet 23(20):5492–5504
Wen W et al (2016) Genome-wide association studies in East Asians identify new loci for waist-hip ratio and waist circumference. Sci Rep 6:17958
Ng MC et al (2012) Genome-wide association of BMI in African Americans. Obesity (Silver Spring) 20(3):622–627
Monda KL et al (2013) A meta-analysis identifies new loci associated with body mass index in individuals of African ancestry. Nat Genet 45(6):690–696
Marchitti SA et al (2008) Non-P450 aldehyde oxidizing enzymes: the aldehyde dehydrogenase superfamily. Expert Opin Drug Metab Toxicol 4(6):697–720
Vasiliou V et al (2013) Aldehyde dehydrogenases: from eye crystallins to metabolic disease and cancer stem cells. Chem Biol Interact 202(1–3):2–10
Zhang Y, Ren J (2010) Autophagy in ALDH2-elicited cardioprotection against ischemic heart disease: slayer or savior? Autophagy 6(8):1212–1213
Sun L, Ferreira JC, Mochly-Rosen D (2011) ALDH2 activator inhibits increased myocardial infarction injury by nitroglycerin tolerance. Sci Transl Med 3(107):107–111
Lee CH et al (2009) Genetic modulation of ADH1B and ALDH2 polymorphisms with regard to alcohol and tobacco consumption for younger aged esophageal squamous cell carcinoma diagnosis. Int J Cancer 125(5):1134–1142
Toh Y et al (2010) Alcohol drinking, cigarette smoking, and the development of squamous cell carcinoma of the esophagus: molecular mechanisms of carcinogenesis. Int J Clin Oncol 15(2):135–144
Hao PP et al (2011) Meta-analysis of aldehyde dehydrogenase 2 gene polymorphism and Alzheimer's disease in East Asians. Can J Neurol Sci 38(3):500–506
Wang B et al (2008) The association of mitochondrial aldehyde dehydrogenase gene (ALDH2) polymorphism with susceptibility to late-onset Alzheimer's disease in Chinese. J Neurol Sci 268(1–2):172–175
Brooks PJ et al (2009) The alcohol flushing response: an unrecognized risk factor for esophageal cancer from alcohol consumption. PLoS Med 6(3):e50
Cho YS et al (2009) A large-scale genome-wide association study of Asian populations uncovers genetic factors influencing eight quantitative traits. Nat Genet 41(5):527–534
Wang T et al (2016) Effects of obesity related genetic variations on visceral and subcutaneous fat distribution in a Chinese population. Sci Rep 6:20691
Lear SA et al (2009) Ethnic variation in fat and lean body mass and the association with insulin resistance. J Clin Endocrinol Metab 94(12):4696–4702
Lear SA et al (2007) Visceral adipose tissue accumulation differs according to ethnic background: results of the Multicultural Community Health Assessment Trial (M-CHAT). Am J Clin Nutr 86(2):353–359
Deurenberg P, Deurenberg-Yap M, Guricci S (2002) Asians are different from Caucasians and from each other in their body mass index/body fat per cent relationship. Obes Rev 3(3):141–146
Cho Y et al (2018) Exploring the utility of alcohol flushing as an instrumental variable for alcohol intake in Koreans. Sci Rep 8(1):458
MacInnis RJ et al (2014) Predictors of increased body weight and waist circumference for middle-aged adults. Public Health Nutr 17(5):1087–1097
Sayon-Orea C et al (2011) Type of alcoholic beverage and incidence of overweight/obesity in a Mediterranean cohort: the SUN project. Nutrition 27(7–8):802–808
Schutze M et al (2009) Beer consumption and the 'beer belly': scientific basis or common belief? Eur J Clin Nutr 63(9):1143–1149
Molenaar EA et al (2009) Association of lifestyle factors with abdominal subcutaneous and visceral adiposity: the Framingham Heart Study. Diabetes Care 32(3):505–510
Yeomans MR (2010) Alcohol, appetite and energy balance: is alcohol intake a risk factor for obesity? Physiol Behav 100(1):82–89
Austin GL, Krueger PM (2013) Increasing the percentage of energy from dietary sugar, fats, and alcohol in adults is associated with increased energy intake but has minimal association with biomarkers of cardiovascular risk. J Nutr 143(10):1651–1658
Yeomans MR (2004) Effects of alcohol on food and energy intake in human subjects: evidence for passive and active over-consumption of energy. Br J Nutr 92(Suppl 1):S31–S34
Coulson CE et al (2013) Alcohol consumption and body composition in a population-based sample of elderly Australian men. Aging Clin Exp Res 25(2):183–192
French MT et al (2010) Alcohol consumption and body weight. Health Econ 19(7):814–832
Mozaffarian D et al (2011) Changes in diet and lifestyle and long-term weight gain in women and men. N Engl J Med 364(25):2392–2404
Wannamethee SG, Shaper AG, Whincup PH (2005) Alcohol and adiposity: effects of quantity and type of drink and time relation with meals. Int J Obes 29(12):1436–1444
Kim KH et al (2012) Alcohol consumption and its relation to visceral and subcutaneous adipose tissues in healthy male Koreans. Ann Nutr Metab 60(1):52–61
Cornier MA, Gayles EC, Bessesen DH (2002) The effects of chronic ethanol consumption on energy balance in rats. Metabolism 51(6):787–791
Yeomans MR, Phillips MF (2002) Failure to reduce short-term appetite following alcohol is independent of beliefs about the presence of alcohol. Nutr Neurosci 5(2):131–139
Yeomans MR (2010) Short term effects of alcohol on appetite in humans. Effects of context and restrained eating. Appetite 55(3):565–573
Jerlhag E et al (2009) Requirement of central ghrelin signaling for alcohol reward. Proc Natl Acad Sci U S A 106(27):11318–11323
Yeomans MR, Caton S, Hetherington MM (2003) Alcohol and food intake. Curr Opin Clin Nutr Metab Care 6(6):639–644
Rodrigue FD, Covenas R (2011) Targeting opioid and neurokinin-1 receptors to treat alcoholism. Curr Med Chem 18(28):4321–4334
Hendershot CS et al (2009) ALDH2, ADH1B and alcohol expectancies: integrating genetic and learning perspectives. Psychol Addict Behav 23(3):452–463
Komiya H et al (2006) Smoking as a risk factor for visceral fat accumulation in Japanese men. Tohoku J Exp Med 208(2):123–132
Cigolini M et al (1996) Moderate alcohol consumption and its relation to visceral fat and plasma androgens in healthy women. Int J Obes Relat Metab Disord 20(3):206–212
Halkjaer J et al (2004) Food and drinking patterns as predictors of 6-year BMI-adjusted changes in waist circumference. Br J Nutr 92(4):735–748
Koppes LL et al (2005) Cross-sectional and longitudinal relationships between alcohol consumption and lipids, blood pressure and body weight indices. J Stud Alcohol 66(6):713–721
Gonzalez-Reimers E et al (2014) Alcoholism: a systemic proinflammatory condition. World J Gastroenterol 20(40):14660–14671
Kang L et al (2007) Chronic ethanol-induced insulin resistance is associated with macrophage infiltration into adipose tissue and altered expression of adipocytokines. Alcohol Clin Exp Res 31(9):1581–1588
Sun X et al (2012) Activation of peroxisome proliferator-activated receptor-gamma by rosiglitazone improves lipid homeostasis at the adipose tissue-liver axis in ethanol-fed mice. Am J Physiol Gastrointest Liver Physiol 302(5):G548–G557
Leggio L et al (2009) Is cortisol involved in the alcohol-related fat mass impairment? A longitudinal clinical study. Alcohol Alcohol 44(2):211–215
Brkic S, Soderpalm B, Gordh AS (2016) High cortisol responders to stress show increased sedation to alcohol compared to low cortisol responders: An alcohol dose-response study. Pharmacol Biochem Behav 143:65–72
Dick DM (2011) Gene-environment interaction in psychological traits and disorders. Annu Rev Clin Psychol 7:383–409
Grove ML et al (2007) Gene-environment interaction and the GNB3 gene in the Atherosclerosis Risk in Communities study. Int J Obes 31(6):919–926
Yin H, Xu L, Porter NA (2011) Free radical lipid peroxidation: mechanisms and analysis. Chem Rev 111(10):5944–5972
Xu T et al (2017) Aldehyde dehydrogenase 2 protects against oxidative stress associated with pulmonary arterial hypertension. Redox Biol 11:286–296
Voulgaridou GP et al (2011) DNA damage induced by endogenous aldehydes: current state of knowledge. Mutat Res 711(1–2):13–27
Wang S et al (2018) ALDH2 protects against high fat diet-induced obesity cardiomyopathy and defective autophagy: role of CaM kinase II, histone H3K9 methyltransferase SUV39H, Sirt1, and PGC-1alpha deacetylation. Int J Obes 42(5):1073–1087
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Hu, C. (2019). Aldehyde Dehydrogenases Genetic Polymorphism and Obesity: From Genomics to Behavior and Health. In: Ren, J., Zhang, Y., Ge, J. (eds) Aldehyde Dehydrogenases. Advances in Experimental Medicine and Biology, vol 1193. Springer, Singapore. https://doi.org/10.1007/978-981-13-6260-6_8
Download citation
DOI: https://doi.org/10.1007/978-981-13-6260-6_8
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-6259-0
Online ISBN: 978-981-13-6260-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)