Abstract
Coral-reef ecosystems are declining worldwide, compromising their capacity to provide ecosystem services that include feeding hundreds of millions of people and protecting shorelines from erosion. The anthropogenic causes of reef degradation are complex and operate over a broad range of scales and hierarchical levels, but accelerating climate change and its collateral impacts are currently the strongest drivers. Deleterious trends in local-scale, ecological processes that occur within reef communities, such as declining herbivory and increasing eutrophication, generally play a subsidiary role at present, because their effects are overwhelmed by the impacts of climate change on many reefs. That does not mean local-scale ecology is irrelevant. Solving environmental problems at one scale or level will by default leave problems at the other scale as the new primary problems. If humanity is able to control climate change at the global level, then community-level processes will in general become limiting. Both local and global impacts must be mitigated and reversed if we are to save coral reefs.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Acropora
- Caribbean
- Climate change
- Coral bleaching
- Coral disease
- Marine protected areas
- MPAs
- White-band disease
11.1 Introduction
Marine ecosystems throughout the world ocean have been damaged by human activities, and coral reefs have suffered especially severe impacts (Halpern et al. 2008). The challenge for coral-reef scientists is to determine the strongest causal pathways to degradation. Measuring the relative contributions of proximate and ultimate candidate-causes is not merely an academic exercise; the prescriptions for mitigating and reversing reef degradation differ depending on the scales, hierarchical levels, and identities of those causes. With limited resources available to conserve coral reefs, it is imperative that time, labor, and funds be devoted to corrective measures that will yield the maximum benefits .
Because reefs are geological as well as ecological entities, the physical sciences have been integral to their study from the start. Our understanding of how coral reefs operate strongly emphasizes physical drivers (e.g., Roberts et al. 1992; Hubbard 1997; Montaggioni and Braithwaite 2009). A few basic examples highlight the physical control of biological processes: (1) reef development is limited to latitudes warmer than the 18 °C winter-minimum isotherm (Dana 1843; Johannes et al. 1983; Kleypas et al. 2001); (2) upwelling driven by oceanic gyres restricts reef development off the west coasts of continents (Birkeland 1997; Hubbard 1997), and inimical waters suppress reef development on smaller scales (Neumann and Macintyre 1985; Hallock and Schlager 1986; Ginsburg and Shinn 1994); (3) antecedent topography and fluctuating sea level determine the growth and form of reefs and their scope for vertical accretion (Darwin 1842; Neumann and Macintyre 1985); (4) light and wave exposure combine with topography to create the biological zonation of reefs (Adey and Burke 1977; Geister 1977; Woodley et al. 1981; Hallock and Schlager 1986; Hubbard 1988; Acevedo et al. 1989; Graus and Macintyre 1989; Murdoch 2007); and (5) climatic fluctuations set the tempo and mode of reef development (Precht and Aronson 2004; Precht and Miller 2007; Toth et al. 2012, 2015). Today geology and paleobiology are helping us to distinguish natural from anthropogenic perturbations of coral reefs and to understand the scales at which those perturbations occur (Aronson 2007). An emerging theme—and the subject of this review—is the overriding influence of physical forcing in the recent, worldwide degradation of reefs.
What is signal to an ecologist is largely noise to a paleontologist. Fossil deposits from shallow, soft-bottom facies are often temporally and spatially averaged, obscuring the record of short-term variability on which the science of ecology still nourishes itself, for better or worse (but see Kidwell 2001, 2007). Time averaging and transport are generally less problematic in interpreting coral-reef deposits than biotas buried in soft sediments, because for coral reefs the benthic assemblages themselves construct their sedimentary fabrics . Coral colonies have longer lifespans than most soft-sediment invertebrates, and their skeleton s are to some extent resistant to taphonomic degradation. Furthermore, because coral skeletons are made of calcium carbonate and, in most cases, the skeletal framework and entombing sediments are cemented, corals are often buried and preserved in place and in sequence , or at most subject only to minor transport. Even where reefs are uncemented, the coral assemblages are often autochthonous and sequential (Aronson and Precht 1997; Aronson et al. 2002, 2004, 2005; Wapnick et al. 2004; Greer et al. 2009). Fossil and subfossil reef deposits, therefore, provide an excellent record from which to understand the ecology of coral reefs in times past and thereby discern the time frame and effects of natural and anthropogenic perturbations on modern reefs (Pandolfi 1996; Greenstein et al. 1998; Pandolfi and Jackson 2001, 2006; Precht and Aronson 2006; Greenstein and Pandolfi 2008; Lescinsky et al. 2012; Toth et al. 2012; and many others).
In this chapter we use evidence from fossil and modern coral reefs to review and critically evaluate three related propositions that have attained enormous popularity among coral-reef ecologists: (1) localized human activity, specifically overfishing , has been the primary cause of the decline of coral populations; (2) the cascading, top-down effects of overfishing are currently limiting the recovery of coral assemblages; and, therefore, (3) local management actions are capable of promoting the resilience of reefs to climate change. We contrast these notions with the idea that physical drivers are the primary determinants of reef dynamics at scales not much larger than the scale of the reef or reef system and should be important considerations in management and conservation. We focus on the reefs of Florida , the Bahamas , and the Caribbean (henceforth collectively termed ‘the Caribbean’), which are significantly altered (Gardner et al. 2003; Schutte et al. 2010) and for which the historical, ecological, and paleobiological data are complete enough to draw reasonably firm conclusions.
11.2 Causal Connections in the Degradation of Caribbean Reefs
Aronson and Precht (2001a, 2001b, 2006) suggested that larger-scale factors, specifically climate change and coral disease (which is related to climate change ), were the primary causes of reef degradation throughout the Caribbean region over the preceding three decades. The elkhorn coral Acropora palmata had dominated the reef-crest and shallow fore-reef habitats at 0–5 m depth on windward -facing Caribbean reefs, whereas the staghorn coral A. cervicornis had dominated intermediate, fore-reef depths of 5–25 m and some back-reef and lagoon al habitats. A regional outbreak of white-band disease (WBD : Fig. 11.1) was the primary cause of the Caribbean-wide mass mortality of these congeneric corals from the late 1970s to the early 1990s. WBD is an infectious, bacterial syndrome that appears only to affect the acroporids (Gil-Agudelo et al. 2006; Weil et al. 2006; Vollmer and Kline 2008; Kline and Vollmer 2011; Gignoux-Wolfsohn et al. 2012; Sweet et al. 2014). Because in many locations A. cervicornis and A. palmata were the dominant occupants of reef substratum and the dominant constructors of framework , Aronson and Precht (2001a, 2001b, 2006) concluded that WBD had been the most important cause of coral mortality in the Caribbean in recent decades. Hurricanes , coral bleaching from anomalously high sea temperatures, and additional factors such as corallivory had played subsidiary roles in killing the Caribbean acroporids (ABRT 2005; Gardner et al. 2005). Bleaching and other diseases later killed massive corals, including the formerly abundant, framework-building Orbicella annularis species complex (McWilliams et al. 2005; Aronson and Precht 2006; Eakin et al. 2010; Toth et al. 2014; see also Rogers 2008; Miller et al. 2009; Rogers and Miller 2013).
Meta-analyses subsequent to Aronson and Precht (2001a, 2001b) have supported their conclusions (Côté et al. 2005; Alvarez-Filip et al. 2009; Schutte et al. 2010). Furthermore, paleoecological studies of reefs in several locations around the Caribbean have demonstrated that the recent mass mortality of acroporids was a novel event in at least the last three millennia (Aronson and Precht 1997; Greenstein et al. 1998; Aronson et al. 2002, 2005; Wapnick et al. 2004; Lescinsky 2012). Epidemiological work has suggested, albeit obliquely, that rising sea temperatures were responsible for the devastating outbreak of WBD in the Caribbean (Kline and Vollmer 2011), providing a link to physical processes. More recently, Randall and van Woesik (2015) linked outbreaks of WBD to increased thermal stress associated with climate change. Outbreaks of some other coral diseases have also been tied to rising temperatures (Rosenberg and Ben-Haim 2002; Selig et al. 2006; Bruno et al. 2007; but see Lafferty et al. 2004).
Jackson et al. (2001), in contrast, asserted in a highly publicized review that the disruption of trophic cascades by overfishing was the most important cause of ecological degradation in shallow-marine environments worldwide. For coral reefs, the scenario was that overfishing reduced herbivory, releasing macroalgae , or seaweeds, to overgrow and otherwise outcompete corals for space (see also Pandolfi et al. 2003). The review by Jackson et al. (2001) was immediately welcomed by conservation groups and the popular media, who touted it as a visionary breakthrough in our understanding of human threats to marine life. With equal rapidity Jackson et al. (2001) drew fire from scientists who pointed out that overfishing was neither the only human assault on marine ecosystems nor necessarily the most significant one. Jackson and colleagues responded that they had never intended to imply a negligible role for other drivers of ecosystem degradation (Peterson et al. 2001). In reality, Jackson et al. (2001) had acknowledged the existence of other factors but had downplayed them.
This group of authors later moderated their stance on overfishing to a more pluralistic view of causality by including sedimentation and nutrient loading from terrestrial sources as another major threat to coral reefs (Bellwood et al. 2004; Kuntz et al. 2005; Jackson 2008). Some of them have recently regressed to their initial stance that overfishing of herbivores , specifically parrotfish , is far and away the primary cause (Jackson et al. 2014).
Terrigenous input has certainly been an important cause of degradation in some situations (Rogers 1990; Cortés 1994; Aronson et al. 2004, 2014; De’ath and Fabricius 2010). On the other hand, one top-down scenario of the impacts of overfishing on coral reefs included speculation of a strong, cascading trophic connection between shark-fishing and a high prevalence of infectious diseases in coral populations (Sandin et al. 2008). That causal chain has not been demonstrated. More convincing are data showing that populations of the corallivorous seastar Acanthaster planci are reduced on Pacific reefs that support more intact stocks of predatory fish (Dulvy et al. 2004; McCook et al. 2010).
Claims about the primacy of overfishing are a step backward from Hughes (1994), who argued that overfishing, the regional mass mortality in 1983–1984 of the herbivorous echinoid Diadema antillarum from an infectious disease , and direct coral mortality from a hurricane had combined to drive a phase shift from coral to macroalgal dominance on Jamaican reefs. The loss of herbivores has been particularly egregious in Jamaica (Aronson 1990; Hughes et al. 1999; Aronson and Precht 2000), so overfishing was considered an important ingredient in the transition to dominance by macroalgae . Whether or not the construct for Jamaica can be generalized to the rest of the Caribbean is an important question (Côté et al. 2013). In fact, mass coral mortalities permitted macroalgae to rise to dominance opportunistically even in some locations with reasonably intact fish assemblages. It also turns out, surprisingly in hindsight, that only a minority of reefs in the Caribbean actually became dominated by macroalgae (Aronson and Precht 2006; Precht and Aronson 2006; Bruno et al. 2009; Dudgeon et al. 2010; Schutte et al. 2010; Bruno et al. 2014).
There is no doubt that much of the world is overfished and that in some situations overfishing can have drastic, cascading impacts on marine ecosystems. The model of Jackson et al. (2001), that the loss of top predators to overfishing fundamentally alters marine food webs and is the primary impact of human activity, works well for ecosystems with strong top-down trophic connections, such as kelp forests (Estes and Duggins 1995; Shears and Babcock 2003; Estes et al. 2011), but its applicability is far from universal. The overfishing hypothesis has been questioned or refuted for seagrass beds, oyster reefs, pelagic ecosystems, and some kelp forests, as well as coral reefs. In these cases, top-down trophic connections play a minor role in community structure , are too weak to respond substantially to the restoration of fisheries, or are complicated by bottom-up effects and other causes (Boesch et al. 2001; MacKenzie 2007; Waycott et al. 2009; Foster and Schiel 2010; Condon et al. 2012). Like the issue of terrigenous input, the question is not whether overfishing can be important on some coral reefs under some circumstances—because clearly it can—but what is its relative contribution to the overall decline of coral populations and coral reefs, and on what spatio-temporal scales?
Because the two Caribbean species of Acropora are now rare to the point of being threatened or endangered (Precht et al. 2004; ABRT 2005; Hogarth 2006; Carpenter et al. 2008; Aronson et al. 2009a, 2009b), accurate knowledge of the timing and causes of their decline is critical to their effective management and conservation (National Marine Fisheries Service 2015). Hughes et al. (2010) presented a history of the causes of decline of the acroporid corals in the Caribbean, which included the following language:
[T]wo meta-analyses of the loss of structural complexity of Caribbean reefs between 1969 and 2008 [Alvarez-Filip et al. 2009] and of coral cover from 1971 to 2006 [Schutte et al. 2010] have proposed that an unreported epidemic of white band disease [emphasis ours] killed off most branching staghorn and elkhorn corals across the region in the1970s. In reality, the loss of coral cover has been highly asynchronous, and disease is only one of many causes of the decline . For instance, cold water killed >90 % of staghorn corals in the Dry Tortugas, Florida in the winter of 1976–77 [Davis 1982]. The collapse of branching acroporids in Jamaica was overwhelmingly because of Hurricane Allen in 1980 [Woodley et al. 1981]. There is only one report of a significant outbreak of white band disease in the Caribbean before 1980, a localized die-off affecting 5 hectares of shallow reef in St. Croix , US Virgin Islands in 1976–1979 [Gladfelter 1982]. In contrast, hurricanes and coral disease were dismissed as causes of the steep decline in coral cover in the Dutch Antilles from 1973 to 1992 [Bak and Nieuwland 1995].
This passage raises several issues. First, Hughes et al. (2010) may be correct that a cold-water event was responsible for the mortality of vast fields of A. cervicornis in the Dry Tortugas in 1977. Porter et al. (1982) used photographs of permanent quadrats taken 6 months before and 6 months after the event as evidence. The photographs, unfortunately, do not establish causality and WBD cannot be ruled out as the cause of mortality of A. cervicornis. In fact, the before-and-after photographs look suspiciously as though they are displaying mortality from WBD (see especially Fig. 11.2 in the paper) and not bleaching from cold-exposure. Most of the losses of A. palmata and A. cervicornis throughout the Florida reef tract were from WBD , especially after 1978 (Precht and Miller 2007; references therein).
Approximate ranges of annual sea temperatures within shallow-water habitats from polar to tropical latitudes. Narrow ranges have driven the evolution of stenothermy in ectotherms at the poles and in the tropics, whereas temperate ectotherms tend to be eurythermal. The polar range is for Antarctica (Data are from Clarke and Crame 2010 and other sources. Modified from Aronson et al. 2011)
Second, Hughes et al. (2010) pointed out that mortality from Hurricane Allen in 1980 was the principal cause of the collapse of Acropora populations at Discovery Bay and elsewhere along the north coast of Jamaica . Knowlton et al. (1981), however, also noted, “Unusual amounts of tissue exfoliation, resembling that termed ‘white band disease ’ were observed in some colonies of A. cervicornis before the hurricane . This exfoliation continued after the storm …,” and within 5 months there was a 100-fold decrease in the abundance of living colonies of A. cervicornis compared to the population immediately after the storm. Hurricane Allen was clearly a catastrophic disturbance on Jamaican reefs, but it is equally apparent that WBD was critical to the decline of A. cervicornis on these reefs. In fact, lagoonal populations of A. cervicornis at Discovery Bay were killed outright by WBD , not by Hurricane Allen (Wapnick et al. 2004).
Third, Gladfelter (1982) recognized the devastating effects of WBD on acroporids in St. Croix , spanning the years 1976–1979, prior to Hurricane Allen. Hughes et al. (2010) minimized Gladfelter’s work by asserting that the outbreak of WBD was localized to a small area in St. Croix. In contrast, Gladfelter (1982) stated the following:
Throughout much of its range, A. palmata is subject to a necrosis which can cause extensive local mortality of the coral. The author has observed this necrosis (= “white band disease ”) in the northeastern Caribbean Sea (Virgin Islands , St. Marten, Antigua), Curaçao, [and] Nicaragua (Miskito Cays), and it has been observed in Panama (P. Glynn, pers. comm.) and south Florida (A. Antonius, pers. comm.).
The outbreak of WBD in St. Croix was clearly part of an epidemic that was well underway throughout the Caribbean in the late 1970s, and it was recognized by Gladfelter as a regional phenomenon at the time.
Finally, citing Bak and Nieuwland (1995), Hughes et al. (2010) stated that coral disease was not responsible for the observed mortality of corals in the Netherlands Antilles. Although Bak and Nieuwland (1995) noted that factors such as diseases were unlikely to have been important in structuring the reefs in question, their study was confined to water depths of 10–40 m and their quadrats contained no Acropora species. They were, however, careful to note, “White-band disease is practically limited to the Acropora species and these are only common at depths shallower than 10 m along these coasts (Bak and Criens 1981; van Duyl 1985).” The two latter references were the same ones Aronson and Precht (2001a, 2001b) cited to describe the strong impact of WBD in the Netherlands Antilles.
Using data from a paleoecological study, Cramer et al. (2012) attempted to dispute the conclusions of Aronson and Precht (2001a, 2001b, 2006): (1) that the most significant losses of acroporid corals in the Caribbean occurred beginning in the late 1970s; and, related, (2) that WBD was the primary cause of the decline of acroporids . Aronson and Precht drew these conclusions based on their compilation of direct observations by a large number of scientists and other informed observers from 31 areas distributed among 16 countries or territories throughout the tropical and subtropical western Atlantic . Cramer et al. (2012) suggested instead that A. cervicornis had begun to decline in the Caribbean because of anthropogenic pressure decades before the outbreak of WBD and the frequent occurrence of coral-bleaching events. They based their alternative interpretation on the stratigraphic distribution of subfossil A. cervicornis in 18 circular trenches, each 60 cm in diameter and 60–80 cm deep, which they excavated at six stations in one area in one country: Bocas del Toro, Panamá .
Cramer dug three trenches at each of the six stations at Bocas del Toro. Half the sites (nine trenches) were dug in a lagoonal environment and the other half (another nine trenches) were situated ‘offshore ,’ in a near-coastal environment. At the lagoonal stations, which were located in Bahía Almirante and the adjacent Laguna de Chiriquí, three of the nine trenches exhibited a decline of A. cervicornis before the regional WBD outbreak (based on radiocarbon dates of Porites furcata—not A. cervicornis—derived from accelerator mass spectrometry, or AMS), a pattern consistent with the preferred scenario of Cramer et al. (2012). Five lagoonal trenches had little or no A. cervicornis at any level, ranging in proportional weight from 0 to 8 % of the total coral material. Radiocarbon dates from the ninth lagoonal trench showed reversals that indicated significant stratigraphic mixing. In the offshore environment, none of the trenches showed evidence of an early decline of A. cervicornis. One station, consisting of three trenches, had negligible quantities of A. cervicornis. At each of the other two stations, two trenches showed declines of A. cervicornis in the 1970s or later (again based on AMS dates from P. furcata rather than the focal species), whereas there was no clear pattern in the third trench at any of the stations due to mixing. Not only are the temporal patterns of coral dominance inconsistent, but AMS is inaccurate at the young ages of the corals that were dated, calling into question the age models on which the conclusions were based (Aronson et al. 2014). The claim that the decline of A. cervicornis at Bocas del Toro began prior to the outbreak of WBD and subsequent bleaching events is not supported by the data in Cramer et al. (2012), nor by more accurate chronologies from the same area based on 210Pb dating (Aronson et al. 2014).
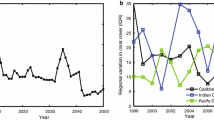
There can be little doubt that the largest decline in coral cover on Caribbean reefs that occurred in recent decades resulted from the regional mass mortality of Acropora spp. from WBD (Gladfelter 1982; Bythell and Sheppard 1993; Aronson and Precht 2001a, 2001b; Schutte et al. 2010), subsequent mass mortalities of massive corals notwithstanding. The disease -induced mass mortality of acroporids that occurred from the late 1970s through the early 1990s was not demonstrably connected to overfishing or changes in land use, and it was more than likely related to warming sea temperatures (Kline and Vollmer 2011; Randall and van Woesik 2015). Why is this important? Cramer et al. (2012) stated, “[Our] results, coupled with increasing evidence that protection from local disturbances may increase reef resilience to climate change (Hughes et al. 2007; Knowlton and Jackson 2008), highlight the importance of managing local impacts such as fishing and land clearing to stem the tide of reef decline .” In fact, the primary causes of the decline of Acropora, and other Caribbean corals for that matter, operated—and still operate—on larger spatial scales, highlighting the importance of confronting regional and global impacts if we are to save coral reefs.
Below, we evaluate whether local management can promote resilience under current conditions. We emphatically agree that local problems should be addressed, for reasons spelled out at the end of this chapter. Contrary to the accusation of Knowlton and Jackson (2008) that we have been monolithic in our view of the importance of WBD , we have always explicitly subscribed to the pluralistic view of Quinn and Dunham (1983) that ecology seeks to evaluate the relative importance of the many causes underlying an observed pattern. Overestimating and overvaluing our capacity to promote resilience through local action, however, diverts attention and resources from the issue of climate change.
11.3 Indicators of Degradation
McClanahan, Graham et al. (2011) showed that, as reefs in the Indian Ocean degraded, corals were the most resistant components and the last to decline . Planes et al. (2005) reported that when shock waves from nuclear testing extirpated the reef-fish assemblages of Mururoa Atoll in French Polynesia , the living coral assemblages remained intact and the habitat they provided facilitated recolonization of the fish. Likewise, the early losses of reef components in the Caribbean other than corals, such as fish stocks (Jackson and Johnson 2000; Jackson 2008), are not incompatible with the recent loss of acroporids and other coral species to diseases and other causes (cf. Woodley 1992). On reefs where living coral cover has recently declined, however, the loss of that coral and concomitant loss of physical structure have resulted in significant declines in reef fish, independent of any impacts of fishing pressure (Jones et al. 2004; Alvarez-Filip et al. 2009; Paddack et al. 2009).
Our perception of the extent of reef degradation clearly depends on how degradation is defined and which components are considered important from ecological, societal, or other viewpoints. Significant functional degradation is perceived to have occurred earlier if fish stocks are considered most important than if corals are considered to be the signal components of the reefs named for them (Jackson and Johnson 2000; Pandolfi et al. 2003; McClanahan, Graham et al. 2011). The first view, in which the ‘health’ of a reef hinges on the state of its fish assemblage, implies long periods of latent degradation at the ecosystem level. Undetected losses of resilience are expressed latterly by threshold phenomena in the coral assemblages, including mass mortalities and poor recovery from bleaching events and disease outbreaks. The second view, which focuses on the corals themselves as the bellwethers of reef condition, implies the alternative hypothesis that fish—especially herbivorous fish—are less important to maintaining coral dominance than was previously thought. Herbivory could be critical to recovery in situations in which macroalgae have the potential to monopolize the substratum and suppress populations of juvenile corals (Sammarco 1982; Hughes and Tanner 2000; Mumby 2006; Box and Mumby 2007; Mumby et al. 2007a; Idjadi et al. 2010; Adam et al. 2015). Even in those cases, however, fish are not necessarily the most important herbivores . Echinoids, especially Diadema antillarum, are often far more potent herbivores on Caribbean reefs (Sammarco 1982; Edmunds and Carpenter 2001; Idjadi et al. 2006; Idjadi et al. 2010). The latter observation is independent of whether or not overfishing artificially enhanced the abundance of Diadema prior to their regional mass mortality (Hay 1984; Carpenter 1986; Precht and Aronson 2006; Sandin and McNamara 2012).
If macroalgae pose a threat to the recovery of coral populations on most reefs, there is little difference in the two views beyond the semantic issue of the point at which a reef is said to be degrading or degraded. The two views differ markedly, however, if macroalgae generally do not threaten coral recovery. Although, as stated above, high abundances of macroalgae can suppress coral recruitment , Bruno et al. (2009, 2014) questioned the proposition that macroalgae dominate most Caribbean reefs in their current state of low coral cover (see also Côté et al. 2005, 2013). The implication is that the decline and recovery of coral populations are largely decoupled from fishing pressure, as has been demonstrated explicitly for several reef systems in the Caribbean (Aronson et al. 2012; Edmunds 2013).
11.4 The Role of Marine Protected Areas
The overfishing hypothesis in its extreme form leads directly to the idea that ecological problems in the sea would largely be solved if only we would control fishing pressure (e.g., Jackson et al. 2014). This presumption provides a clear rationale for continuing to set aside marine protected areas (MPAs) and continuing the protections afforded by existing MPAs, which are designed to control the exploitation of fish stocks. By virtue of their current design, however, MPAs are less effective or wholly ineffective at controlling terrigenous inputs of nutrients and sediments. And by virtue of their scale they do not address the root-causes of climate change: the size of the human population and greenhouse-gas emissions. Climate change is expressed on coral reefs through the direct impacts of increasing sea temperatures, decreasing carbonate saturation states, and rising sea levels, as well as ancillary effects that may include outbreaks of coral disease and increasing intensities of hurricanes (Kleypas et al. 2001; Gardner et al. 2005; Hoegh-Guldberg et al. 2007; Harvell et al. 2009; Anthony et al. 2011; and many others).
Although Mora et al. (2006) concluded that MPAs in their current form do not preserve the trophic cascades of predation and herbivory that putatively maintain coral populations, there are benefits to coral cover of protection from fishing and terrigenous input, as well as benefits of protection from fishing pressure alone (Houk et al. 2010; McCook et al. 2010; Selig and Bruno 2010). Evidence is rapidly mounting, however, that overfishing is not the primary threat to benthic assemblages on coral reefs. Protecting fish stocks does not necessarily reduce the cover of macroalgae , increase coral populations, or preserve or increase the topographic heterogeneity that is critical to maintaining and increasing those fish stocks (McClanahan et al. 2001, 2005; Aronson and Precht 2006; Bood 2006; Idjadi et al. 2006; Vroom et al. 2006; Coelho and Manfrino 2007; Kramer and Heck 2007; Bruno et al. 2009; Myers and Ambrose 2009; Stockwell et al. 2009; Dudgeon et al. 2010; Alvarez-Filip et al. 2011; Lowe et al. 2011; McClanahan, Huntington et al. 2011; Żychaluk et al. 2012; Bégin et al. 2016). The threats of continuing climate change and its collateral impacts loom large, raising questions about the potential of local management alone, or the phenotypic or evolutionary responses of corals and their zooxanthellae (Baker et al. 2008; Sammarco and Strychar 2009; Pandolfi et al. 2011; van Woesik and Jordán-Garza 2011), to reverse or even delay significantly the hemmorhagic damage that is already well underway (Donner et al. 2005; Hoegh-Guldberg et al. 2007; Donner 2009; Hoegh-Guldberg et al. 2011; Toth et al. 2014).
The claim that restoring herbivores will save coral populations by reducing the cover of macroalgae (Aronson 1990; Jackson et al. 2001; Pandolfi et al. 2003; Mumby 2006; Jackson et al. 2014) has been ‘augmented’ with the idea that MPAs will maintain the resilience of reefs, ‘buying time’ while we address climate change (Hughes et al. 2003; Bellwood et al. 2004; Hughes et al. 2007; Mumby et al. 2007a, 2007b; Hughes et al. 2010; Edwards et al. 2011). Even the latter concept is of questionable validity (McClanahan et al. 2005; Bood 2006; Graham et al. 2008; McClanahan 2008; Graham et al. 2011; Huntington et al. 2011). Existing MPAs do not enhance the resistance or resilience of reef assemblages to thermal stress (Hoegh-Guldberg and Bruno 2010; Selig et al. 2012). Previous exposure of corals to high-temperature conditions is a far better predictor of the persistence of coral populations during positive thermal anomalies than their status of protection (Thompson and van Woesik 2009; Selig et al. 2012; Grottoli et al. 2014).
Côté and Darling (2010) pointed out that disturbed reef assemblages, which replace more pristine (or less degraded) assemblages following perturbations , are by default more resilient because ‘recovery ’ to those early successional states requires little time and meets with little or no systemic resistance . It is cold comfort to be reminded of the inescapable, thermodynamic reality that the end of marine life will be the most stable state of all. As we labor to prevent that dreadful eventuality from accelerating into the present century, it is well to remember that the most resistant or resilient configurations are not necessarily the most desirable (Rogers 2013).
11.5 Parsimonious Explanations
Millennial-scale physical drivers, including natural trends in climate , often explain the historical limits to the growth and composition of Holocene coral assemblages more simply and more completely than hypotheses of human exploitation and other forms of interference. Fishing and terrigenous input are regional issues, but they are perpetrated and controlled locally. Climate change occurs at the largest spatial scales, but the resultant changes in parameters such as sea temperature and pH act at very small scales. They influence the coral holobiont, its physiological rates, and the microenvironment in which it lays down aragonite crystals, as well as rates of carbonate precipitation and submarine cementation from other biotic and abiotic processes (Kleypas et al. 1999; Macintyre and Aronson 2006; Manzello et al. 2008). Those microscale processes scale up to the level of the reef system and beyond, interacting with such second-order rates as the flux of nutrients into the system and their influence on carbonate deposition and bioerosion . Herbivory, predation , and other rates that ecologists view as critical to the healthy functioning, persistence, and resilience of reef systems (Jackson et al. 2001; Mumby et al. 2007b; Sandin et al. 2008) overprint the impacts of physical processes, driving the trajectories of benthic reef assemblages over a range of relevant time scales (Urban et al. 2012). The challenge is to determine how important those ecological interactions really are on ecological scales of decades to centuries, whether larger, millennial time scales have been more important than ecological scales, and which processes have been important on those millennial time scales.
Just because people were around when acroporid corals ceased building reef framework off present-day Fort Lauderdale 6000 years ago (Lighty et al. 1978; Toscano and Macintyre 2003; Banks et al. 2007) does not mean humans were responsible for their decline . Climatic cooling in the late Holocene was likely the primary cause of reef shutdown off the eastern coast of the Florida Peninsula, and a warming climate is now permitting the northward re-expansion of cold-sensitive coral taxa in the western Atlantic (Precht and Aronson 2004; Precht and Miller 2007; see Greenstein and Pandolfi 2008 for an example from western Australia ).
In a similar vein, human activities did not drive branching Porites corals to replace A. cervicornis ~500 years ago in the shallow zones of the uncemented, lagoonal , rhomboid shoals in Belize . As those reefs grew to sea level , the living coral assemblages relocated themselves to a new physical environment—shallower water—and their species composition changed to produce the observed shallowing-upward sequence (Aronson et al. 1998). The persistence of the coral assemblages and the geomorphology of the reefs are controlled by tectonic events, which occur every few millennia, cause catastrophic slope-failure, and wipe out a substantial proportion of the benthic communities (Aronson et al. 2012).
Some investigators have insisted on anthropogenic causes for the degradation of coral reefs, in spite of evidence that is at best equivocal. Much has been written about a connection between the advent of European agriculture in Barbados and the demise of populations of Acropora palmata there, but the supposition that the story is one of nutrient loading from agricultural runoff rests almost entirely on a mild suggestion in a paper by John Lewis (1984). Lewis attributed the late Holocene disappearance of Acropora palmata from inshore, fringing reefs along the west coast of Barbados to storm damage, successional processes, and possibly terrigenous runoff that resulted from land-clearing and sugar-cane cultivation beginning in the 1600s. Twenty-seven years later, Sala and Jackson (2011, p. 197) had this to say about Lewis’s (1984) results from Barbados:
Circumstantial evidence suggests that the problems in Barbados were due to deforestation of the island for sugarcane and the consequent runoff of sediments and human waste, as well as extreme overfishing to feed the burgeoning population.
Four years before that, however, Macintyre et al. (2007) had commented on Lewis’s (1984) interpretation of the late Holocene record of A. palmata in Barbados :
Formerly attributed to human activity, the demise of a bank–barrier reef off southeastern Barbados …is now thought to be largely the result of late Holocene , millennial-scale storm damage.
Macintyre et al. (2007) suggested that the vibrant growth of stands of A. palmata in a reef-crest habitat off the south coast had ceased long before the European colonization of Barbados . The primary cause of mortality was physical damage 3000–4500 cal BP, with agricultural runoff possibly accounting for the mortality of remnant colonies 300–400 cal BP (see also Toscano 2016). Roff et al. (2013) echoed this latter model in suggesting that terrigenous runoff combined with climatic perturbation drove a phase shift in a nearshore coral assemblage on the Great Barrier Reef. The loss of A. palmata from the south coast of Barbados , millennia before any European influence, could have been part of a regional drawdown of that species (Hubbard et al. 2005; Macintyre et al. 2007).
Lewis’s (1984) speculation about agriculture applies to inshore, fringing reefs . It is, therefore, not necessarily incompatible with the conclusions of Macintyre et al. (2007) about a bank–barrier reef further offshore , but populations of A. palmata in Barbados were not all killed by runoff . Furthermore, the idea that terrigenous input was to blame cannot fully account for the replacement of A. palmata by a vibrant assemblage of massive corals on the inshore reefs (Lewis 1960). A coral assemblage dominated by Orbicella spp. and other massive species is precisely what one would expect in a hurricane -dominated environment (Stoddart 1963; Porter et al. 1981; Woodley 1989), such as that envisioned by Macintyre et al. (2007). Overfishing , incidentally, had never been part of Lewis’s (1984) original scenario, nor did it figure in the interpretations of Macintyre et al. (2007) or Toscano (2016).
An argument for the runoff hypothesis would also have to explain why Barbados , which is a low, carbonate island, is the only known location in the Caribbean for which such a scenario has been suggested. Jamaica , for example, is a high island with a similar history of colonization and sugar-cane cultivation. The impacts of terrigenous runoff should have been accentuated compared to Barbados, yet there is no evidence for a mass mortality of A. palmata in Jamaica (or any other Caribbean island) 300–400 years ago. The best way to test the hypothesis would be through biogeochemical analysis (cf. Aronson et al. 2014).
11.6 Temporal Priority
Which regional or global driver is or was most important in the degradation of modern reefs is largely a consequence of temporal priority. Thermally induced bleaching has been secondarily important in the Caribbean only because bleaching episodes came after much of the Acropora had already been killed by white-band disease , in a regional outbreak that itself might have been thermally driven. The situation is reversed in the Indo-Pacific : bleaching has had a greater influence than coral disease because bleaching-induced mortality occurred on a large geographic scale prior to disease outbreaks (Buddemeier et al. 2004). It remains to be seen whether ocean acidification will have the opportunity to damage coral populations and coral reefs (Hoegh-Guldberg et al. 2007; Veron 2008; Hönisch et al. 2012) after the impacts of rising sea temperatures have taken their toll. Some state or rate is the primary limit to the growth of coral populations, the integrity of the assemblages those populations comprise, and the accretion of the reef frameworks on which they perch as a living veneer.
11.7 Conclusion
It should come as no surprise that coral reefs are highly sensitive to climate change. Narrow annual temperature ranges characterize shallow-benthic habitats at tropical and polar latitudes, compared to analogous habitats in the highly seasonal temperate zone (Fig. 11.2). Benthic ectotherms near their latitudinal extremes are adapted to the narrow seasonal temperature ranges to which they are normally exposed. Stenothermy limits their scope for phenotypic or evolutionary adjustment to warming temperatures. The tropical and polar benthos, therefore, are responding earlier and more strongly to warming sea temperatures than are temperate-marine biotas.
All biological interactions play out on the template of the physical environment. For every ecosystem , there is a range of larger scales at which physical drivers trump biotic interactions in determining its trajectory; it is just a matter of scaling up sufficiently to that range. Reefs through Phanerozoic time are no exception (Wood 1999, 2007; Veron 2008; Kiessling and Simpson 2011; Hönisch et al. 2012; Norris et al. 2013). The projected dynamical responses of reefs to large-scale physical drivers, both natural and anthropogenic , are the appropriate foundations of hypotheses against which to test the ecological effects of localized human activities.
Our point about modern coral reefs under human influence is that one does not have to scale up very much at all to discover the scales at which physical controls predominate, because the lower end of the range of scales at which physical drivers strongly influence the biotic milieu, or indeed overpower biological processes, is not very large. For coral populations and coral reefs, physical controls are primary at scales equal to, or only slightly larger than, the scales at which biotic interactions are measured and observed. A good example is the influence of anthropogenically warming temperatures on outbreaks of coral disease . Geologic al and ecological processes may be disjunct in some marine ecosystems, but for coral reefs they operate on scales that are very similar.
That is not to say that geology equals ecology . Some paleoecologists view the geologically rapid changes in sea level and sea temperature during the Pleistocene glaciations as disturbances in ecological time, precipitating the collapse of reef communities and requiring their subsequent reconstitution in other habitats or locations (Jackson 1992; Pandolfi 1996; Webster et al. 2004; Jackson and Erwin 2006; Pandolfi and Jackson 2006). But even when glacial/interglacial fluctuations were dramatic in geologic time, they were still slow compared to the turnover rates of the corals (see Kleypas 1997). In most cases, coral populations should have been able to alter their spatial and habitat distributions incrementally over long periods, but the endpoints of these incremental shifts displayed in the fossil record have been presumed to represent radical disassembly and reassembly (see discussion in Tager et al. 2010; see also Chap. 7).
Overfishing is a terrible problem with far-reaching consequences. There is more than enough sorrow to go around over the strangulation of marine ecosystems, and we wholeheartedly endorse efforts to protect life in the sea from the grotesque and irrational level of exploitation to which it is being subjected. Even worse for coral reefs, unfortunately, are rising temperatures, ocean acidification , and other potential impacts of climate change, such as predicted future increases in the intensity of hurricanes (see also Glynn 2011). Because these physical drivers operate at a global scale, they exert a powerful influence that is harder to control by a long shot than fishing pressure and terrigenous input, which are the feasible targets of existing MPAs and other local management strategies. Local actions to control fishing and runoff , along with a more strategic, integrative approach to the design and location of marine reserves that accounts for spatial variation in susceptibility to climate change (Riegl and Piller 2003; McClanahan et al. 2008, 2009; Mumby et al. 2011), could prolong the death-throes of coral populations; however, they will keep dying until government and society recognize climate change for the grave threat it is and address it on a geopolitical level. If and when the impacts of climate change can be mitigated or reversed, their impacts will no longer overwhelm local threats. Fishing , terrigenous input, and other localized problems will then more commonly become the limiting factors and will respond more strongly to the appropriate protective measures. Liebig’s Law of the Minimum, a nineteenth-century model from agronomy (Hooker 1917), has great value for understanding the challenges facing coral reefs and their human stewards.
The Law of the Minimum describes the serial limitation of different nutrients on crop yields. Liebig used the metaphor of a barrel with staves of different lengths, each stave representing a nutrient . The water-level in the barrel represented crop yield, and the shortest stave represented the limiting nutrient. Adapting the metaphor to coral reefs, the staves of the barrel in Fig. 11.3 represent the many factors that potentially limit the growth of coral populations and accretion of reef frameworks , both of which are represented by the water level. The shortest stave identifies the rate-limiting process. The water can be no higher than that stave, meaning that coral growth or framework accretion can be no greater than allowed by the rate-limiting process.
We have chosen rising global temperatures as the primary limitation in Fig. 11.3, based on the foregoing discussion. If the temperature rise can be slowed or reversed, then warming will cease to be the rate-limiting process. The global-warming stave of the barrel will then be lengthened and will no longer be the shortest one. The shortest stave in the figure will then be disease , which as we have said could be linked to warming seas. Synergistic or antagonistic interactions among the drivers of coral-reef degradation (Mora et al. 2007) mean that lengthening some staves will lengthen or shorten certain other staves as well. If and when regional- and global-scale limitations are adequately addressed, then local-scale factors, such as nutrient loading and overfishing , will serially become the limiting staves. The upshot is that simultaneous actions at local, regional, and global levels are our only hope for saving coral reefs (see also Hoegh-Guldberg and Bruno 2010; Kennedy et al. 2013; Rogers 2013). Clearly, global problems will be more difficult to solve and take longer than local ones, but that is very different from saying we should focus on local management now because it will buy time for us to address the impacts of climate change in the future.
Planning to have reefs around for our children and our children’s children to enjoy—meaning that we want to keep them reasonably intact for a little over a century—is just not good enough. We need to conserve reefs on a millennial time scale by fighting climate change on a global spatial scale. It may sound perverse but, considering the jeopardy in which we humans have placed coral reefs, the biosphere, and our very existence, addressing climate change and being left with an exceedingly difficult set of local-scale, ecological problems to attack would be a blessing.
References
ABRT, Acropora Biological Review Team (2005) Atlantic Acropora Status Review Document. Miami, Florida: Report to National Marine Fisheries Service, Southeast Regional Office, 152 p
Acevedo R, Morelock J, Olivieri RA (1989) Modification of coral reef zonation by terrigenous sediment stress. Palaios 4:92–100
Adam TC, Burkepile DE, Ruttenberg BI, Paddack MJ (2015) Herbivory and the resilience of Caribbean coral reefs: knowledge gaps and implications for management. Mar Ecol Prog Ser 520:1–20
Adey WH, Burke RB (1977) Holocene bioherms of Lesser Antilles: geologic control of development. In: Frost SH, Weiss MP, Saunders JB (eds) Reefs and Related Carbonates—Ecology and Sedimentology. American Association of Petroleum Geologists Studies in Geology 4:67–82
Alvarez-Filip L, Dulvy NK, Gill JA, Côté IM, Watkinson AR (2009) Flattening of Caribbeancoral reefs: region-wide declines in architectural complexity. Proc Soc B 276:3019–3025
Alvarez-Filip L, Gill JA, Dulvy NK, Perry AL, Watkinson AR, Côté IM (2011) Drivers of region-wide declines in architectural complexity on Caribbean reefs. Coral Reefs 30:1051–1060
Anthony KRN, Maynard JA, Diaz-Pulido G , Mumby PJ , Marshall PA, Cao L, Hoegh-Guldberg O (2011) Ocean acidification and warming will lower coral reef resilience. Global Change Biol 17:1798–1808
Aronson RB (1990) Onshore–offshore patterns of human fishing activity. Palaios 5:88–93
Aronson RB (2007) Geological approaches to coral reef ecology. New York: Springer-Verlag, 425 pp
Aronson RB, Bruckner A, Moore J, Precht W, Weil E (2009a) Acropora cervicornis (staghorn coral). IUCN Red List: The IUCN Red List of Threatened Species, version 2009.1. http://www.iucnredlist.org/details/133381/0.
Aronson RB, Bruckner A, Moore J, Precht W, Weil E (2009b) Acropora palmata (elkhorn coral). IUCN Red List: The IUCN Red List of Threatened Species, version 2009.1. http://www.iucnredlist.org/details/133006/0.
Aronson RB, Hilbun NL, Bianchi TS, Filley TR, McKee BA (2014) Land use, water quality, and the history of coral assemblages at Bocas del Toro, Panamá. Mar Ecol Prog Ser 504:159–170
Aronson RB, Macintyre IG, Lewis, SA, Hilbun NL (2005) Emergent zonation and geographic convergence of coral reefs. Ecology 86:2586–2600
Aronson, RB, Macintyre IG, Precht WF, Wapnick CM, Murdoch TJT (2002) The expanding scale of species turnover events on coral reefs in Belize. Ecol Monogr 72:233–249
Aronson RB, Macintyre IG, Wapnick CM, O’Neill MW (2004) Phase shifts, alternative states, and the unprecedented convergence of two reef systems. Ecology 85:1876–1891
Aronson RB, Precht WF (1997) Stasis, biological disturbance, and community structure of a Holocene coral reef. Paleobiology 23:326–346
Aronson RB, Precht WF (2000) Herbivory and algal dynamics on the coral reef at Discovery Bay, Jamaica. Limnol Oceanogr 45:251–255
Aronson RB, Precht WF (2001a) Evolutionary paleoecology of Caribbean coral reefs. In: Allmon WD, Bottjer DJ (eds) Evolutionary paleoecology: the ecological context of macroevolutionary change. New York: Columbia University Press, pp 171–233
Aronson RB, Precht WF (2001b) White-band disease and the changing face of Caribbean coral reefs. Hydrobiologia 460:25–38
Aronson RB, Precht WF (2006) Conservation, precaution, and Caribbean reefs. Coral Reefs 25:441–450
Aronson RB, Precht WF, Macintyre IG (1998) Extrinsic control of species replacement on a Holocene reef in Belize: the role of coral disease. Coral Reefs 17:223–230
Aronson RB, Precht WF, Macintyre IG, Toth LT (2012) Catastrophe and the lifespan of coral reefs. Ecology 93:303–313
Aronson RB, Thatje S, McClintock JB, Hughes KA (2011) Anthropogenic impacts on marine ecosystems in Antarctica. Ann NY Acad Sci 1223:82–107
Bak RPM, Criens SR (1981) Survival after fragmentation of colonies of Madracis mirabilis, Acropora palmata and A. cervicornis (Scleractinia) and the subsequent impact of coral disease. Proc 4th Intl Coral Reef Symp 2:221–228
Bak RPM, Nieuwland G (1995) Long-term change in coral communities along depth gradients over leeward reefs in the Netherlands Antilles. Bull Mar Sci 56:609–619
Baker AC, Glynn PW, Riegl B (2008) Climate change and coral reef bleaching: an ecological assessment of long-term impacts, recovery trends and future outlook. Estuar Coast Shelf Sci 80:435–471
Banks KW, Riegl BM, Shinn EA, Pillar WE Dodge RE (2007) Geomorphology of the Southeast Florida continental reef tract (Miami-Dade, Broward, and Palm Beach Counties, USA). Coral Reefs 26:617–633
Bégin C, Schelten CK, Nugues MM, Hawkins J, Roberts C, Côté IM (2016) Effects of protection and sediment stress on coral reefs in St. Lucia. PLoS ONE 11:e0146855, doi: 10.1371/journal.pone.0146855
Bellwood DR, Hughes TP, Folke C, Nyström M (2004) Confronting the coral reef crisis. Nature 429:827–833
Birkeland C (1997) Geographic differences in ecological processes on coral reefs. In: Birkeland C (ed) Life and death of coral reefs. New York: Chapman and Hall, pp 273–297
Boesch D, Burreson E, Dennison W, Houde E, Kemp M, Kennedy V, Newell RT, Paynter K, Orth R, Ulanowicz R (2001). Factors in the decline of coastal ecosystems. Science 293:1589–1590
Bood, N.D. (2006) Recovery and resilience of coral assemblages on managed and unmanaged reefs in Belize: a long-term study. MS Thesis. Mobile: University of South Alabama
Box SJ, Mumby PJ (2007) Effect of macroalgal competition on growth and survival of juvenile Caribbean corals. Mar Ecol Prog Ser 342:139–149
Bruno JF, Precht WF, Vroom PS, Aronson RB (2014) Coral reef baselines: how much macroalgae is natural? Mar Poll Bull 80:24–29
Bruno JF, Selig ER, Casey KS, Page CA, Willis B, Harvell CD, Sweatman H, Melendy A (2007) Thermal stress and coral cover as drivers of coral disease outbreaks. PLoS Biol 5:e124, doi:10.1371/journal.pbio.0050124
Bruno JF, Sweatman H, Precht WF, Selig ER, Schutte VGW (2009) Assessing evidence of phase shifts from coral to macroalgal dominance on coral reefs. Ecology 90:1478–1484
Buddemeier RW, Kleypas JA, Aronson RB (2004) Coral reefs and global climate change: potential contributions of climate change to stresses on coral reef ecosystems. Arlington, Virginia: Pew Center on Global Climate Change, 44 pp
Bythell J, Sheppard C (1993) Mass mortality of Caribbean shallow corals. Mar Poll Bull 26:296–297
Carpenter RC (1986) Partitioning herbivory and its effects on coral reef algal communities. Ecol Monogr 56:345–363
Carpenter KE Abra M, Aeby G, Aronso RB, Banks S, Bruckner A, Chiriboga A, Cortés J, Delbeek JC, DeVantier L, Edgar GJ, Edwards AJ, Fenner D, Guzmán HM, Hoeksema BW, Hodgson G. Johan O, Licuanan WY, Livingstone SR, Lovell ER, Moore JA, Obura DO, Ochavillo D, Polidoro BA, Precht WF, Quibilan MC, Reboton C, Richards ZT, Rogers AD, Sanciangco J, Sheppard A, Sheppard C, Smith J, Stuart S, Turak E, Veron JEN, Wallace C, Weil E, and Wood E (2008) One-third of reef-building corals face elevated extinction risk from climate change and local impacts. Science 321:560–563.
Clarke A, Crame JA (2010). Evolutionary dynamics at high latitudes: speciation and extinction in polar marine faunas. Phil Trans R Soc B 365:3655–3666
Coelho VR, Manfrino C (2007) Coral community decline at a remote Caribbean island: marine no-take reserves are not enough. Aquat Conserv Mar Freshw Ecosys 17:666–685
Condon RH, Graham WM, Duarte CM, Pitt KA, Lucas CH, Haddock SHD, Sutherland KR, Robinson KL, Dawson MN, Decke MB, Mills CE, Purcell JE, Malej A, Mianzan H, Uye S-I, Gelsich S, Madin. LP2 (2012) Questioning the rise of gelatinous zooplankton in the world’s oceans. BioScience 62:160–169
Cortés, J (1994) A reef under siltation stress: a decade of degradation. In: Ginsburg RN (ed.) Proceedings of the Colloquium on Global Aspects of Coral Reefs: Health, Hazards and History. Miami: Rosenstiel School of Marine and Atmospheric Science, University of Miami, pp 240–246
Côté IM, Darling ES (2010) Rethinking ecosystem resilience in the face of climate change. PLoS Biology 8:e1000438, doi:10.1371/journal.pbio.1000438
Côté IM, Gill JS, Gardner TA, Watkinson, AR (2005) Measuring coral reef decline through meta-analyses. Phil Trans R Soc Lond B 360:385–395
Côté IM, Precht WF, Aronson RB, Gardner TA (2013) Is Jamaica a good model for understanding Caribbean coral reef dynamics? Mar Poll Bull 76:28–31
Cramer KL, Jackson JBC, Angioletti CV, Leonard-Pingel J, Guilderson TP (2012) Anthropogenic mortality on coral reefs in Caribbean Panama predates coral disease and bleaching. Ecol Lett 15:561–567
Dana JD (1843) On the temperature limiting the distribution of corals. Am J Sci 45:130–131
Darwin CR (1842) The Structure and Distribution of Coral Reefs. Being the First Part of the Geology of the Voyage of the Beagle, under the Command of Capt. Fitzroy, R.N. during the Years 1832 to 1836. London: Smith, Elder and Company, 214 pp
Davis GE (1982) A century of natural change in coral distribution at the Dry Tortugas: a comparison of reef maps from 1881 and 1976. Bull Mar Sci 32:608–623
De’ath G, Fabricius K (2010) Water quality as a regional driver of coral biodiversity and macroalgae on the Great Barrier Reef. Ecol Appl 20:840–850
Donner SD (2009) Coping with commitment: projected thermal stress on coral reefs under different future scenarios. PLoS One 4:e5712, doi:10.1371/journal.pone.0005712
Donner SD, Skirving WJ, Little CM, Oppenheimer M, Hoegh-Guldberg O (2005) Global assessment of coral bleaching and required rates of adaptation under climate change. Global Change Biol 11:1–15
Dudgeon SR, Aronson RB, Bruno JR, Precht WF (2010) Phase shifts and stable states on coral reefs. Mar Ecol Prog Ser 413:201–216
Dulvy NK Freckleton RP, Polunin NVC (2004) Coral reef cascades and the indirect effects of predator removal by exploitation. Ecol Lett 7:410–416
Eakin CM, Morgan JA, Heron SF, Smith TB, Lui G, et al. (2010) Caribbean corals in crisis: record thermal stress, bleaching, and mortality in 2005. PloS ONE 5:e13969, doi: 10.1371/journal.pone.0013969
Edmunds PJ (2013) Decadal-scale changes in the community structure of coral reefs of St. John, US Virgin Islands. Mar Ecol Prog Ser 489:107–123
Edmunds PJ, Carpenter RC (2001) Recovery of Diadema antillarum reduces macroalgal cover and increases abundance of juvenile corals on a Caribbean reef. Proc Natl Acad Sci USA 98:5067–5071
Edwards HJ, Elliott IA, Eakin CM, Irikawa A, Madin JS, McField M, Morgan JA, van Woesik R Mumby PJ (2011) How much time can herbivore protection buy for coral reefs under realistic regimes of hurricanes and coral bleaching? Global Change Biol 17:2033–2048
Estes JA, Duggins DO (1995) Sea otters and kelp forests in Alaska: generality and variation in a community ecological paradigm. Ecol Monogr 65:75–100
Estes JA, Terborgh T, Brashares JA, Power ME, Berger J, Bond WJ, Carpenter SR, Essington TE, Hol RD, Jackson JBC, Marquis RJ, Oskanen L, Oskane T, Paine RT, Pikitch EK, Ripple WJ, Sandin SA, Scheffer M, Schoene TW, Shurin JB, Sinclair ARE, Soulé ME, Virtanen R, DA Wardle (2011) Trophic downgrading of Planet Earth. Science 333:301–306
Foster MS, Schiel DR (2010) Loss of predators and the collapse of southern California kelp forests (?): alternatives, explanations and generalizations. J Exp Mar Biol Ecol 393:59–70
Gardner TA, Côté IM, Gill JA, Grant A, Watkinson AR (2003) Long-term region-wide declines in Caribbean corals. Science 301:958–960
Gardner TA, Côté IM, Gill JA, Grant A, Watkinson AR (2005). Hurricanes and Caribbean coral reefs: impacts, recovery patterns, and role in long-term decline. Ecology 86:174–184
Geister J (1977) The influence of wave exposure on the ecological zonation of Caribbean coral reefs. Proc 3rd Intl Coral Reef Symp 1:23–29
Gignoux-Wolfsohn SA, Marks CJ, Vollmer SV (2012) White band disease transmission in the threatened coral, Acropora cervicornis. Nature Sci Rep 2:804, doi:10.1038/srep00804
Gil-Agudelo DL, Smith GW, Weil E (2006) The white band disease type II pathogen in Puerto Rico. Rev Biol Trop 54(Suppl 3):59–67
Ginsburg RN, Shinn EA (1994) Preferential distribution of reefs in the Florida reef tract: the past is the key to the present. In Proceedings of the Colloquium on Global Aspects of Coral Reefs: Health, Hazards and History, 1993, Ginsburg RN (ed.), Miami: Rosenstiel School of Marine and Atmospheric Science, University of Miami, pp 21–26
Gladfelter WB (1982) White band disease in Acropora palmata: implications for the structure and growth of shallow reefs. Bull Mar Sci 32:639–643
Glynn PW (2011) In tandem reef coral and cryptic metazoan declines and extinctions. Bull Mar Sci 87:767–794
Graham NAJ, McClanahan TR, MacNeil MA, Wilson SK, Polunin NVC, Jennings S, Chabanet P, Clark S, Spalding MD, Letourneur Y, Bigot L, Gazin R, Öhma MC, Garpe KC, Edwards AJ, Sheppard CRC (2008) Climate warming, marine protected areas and the ocean-scale integrity of coral reef ecosystems. PLoS One 3:e3039, doi:10.1371/journal.pone.0003039
Graham NAJ, Nash KL, Kool JT (2011) Coral reef recovery dynamics in a changing world. Coral Reefs 30:283–294
Graus RR, Macintyr IG (1989) The zonation of Caribbean coral reefs as controlled by wave and light energy input, bathymetric setting and reef morphology: computer simulation experiments. Coral Reefs 8:9–18
Greenstein BJ, Curran AH, Pandolfi JM (1998) Shifting ecological baselines and the demise of Acropora cervicornis in the western North Atlantic and Caribbean Province: a Pleistocene perspective. Coral Reefs 17:249–261
Greenstein BJ, Pandolfi JM (2008) Escaping the heat: range shifts of reef coral taxa in coastal western Australia. Global Change Biol 14:513–528
Greer L Jackson JE, Curran HA, Guilderson T, Teneva L (2009) How vulnerable is Acropora cervicornis to environmental change? Lessons from the early to middle Holocene. Geology 37:263–266
Grottoli AG, Warner ME, Levas SJ, Aschaffenburg MD, Schoepf V, McGinley Baumann MJ, Matsui Y (2014) The cumulative impact of annual coral bleaching can turn some coral species winners into losers. Global Change Biol, doi:10.1111/gcb.12658
Hallock P, Schlager W (1986) Nutrient excess and the demise of coral reefs and carbonate platforms. Palaios 1:389–398
Halpern BS, Walbridge S, Selkoe KA, Kappel CV, Micheli F, D’Agrosa C, Bruno JF, Casey KS, Ebert C, Fox HE, Fujita R, Heinemann D, Lenihan HS, Madin EMP, Perry MT, Selig ER, Spalding M, Steneck R, Watson R (2008) A global map of human impact on marine ecosystems. Science 319:948–952
Harvell D, Altizer S, Cattadori IM, Harrington L, Weil E (2009) Climate change and wildlife diseases: when does the host matter the most? Ecology 90:912–920
Hay ME (1984) Patterns of fish and urchin grazing on Caribbean coral reefs: are previous results typical? Ecology 65:446–454
Hoegh-Guldberg O, Bruno JF (2010) The impact of climate change on the world’s marine ecosystems. Science 328:1523–1528
Hoegh-Guldberg O, Mumby PJ, Hooten AJ, Steneck RS, Greenfield P, Gomez E, Harvell CD, Sale PF, Edwards AJ, K. Caldeira, N. Knowlton, C.M. Eakin, R. Iglesias-Prieto, N. Muthiga, R.H. Bradbury, Dubi E, Hatziolos ME (2007) Coral reefs under rapid climate change and ocean acidification. Science 318:1737–1742
Hoegh-Guldberg O, Ortiz JC, Dove S (2011) The future of coral reefs. Science 334:1494–1495
Hogarth WT (2006) Endangered and threatened species: final listing determinations for elkhorn coral and staghorn coral. Fed Reg 71:26852–26872
Hönisch B, Ridgwell A, Schmidt DN, Thomas E, Gibbs SJ, Sluijs A, Zeebe R, Kump L, Martindale RC, Greene SE, Kiessling W, Ries J, Zachos J, Royer DL, Barker S, Marchitto TM Jr, Myoer R, Pelejero C, Ziveri P, Foster GL, Williams B (2012) The geological record of ocean acidification. Science 335:1058–1063
Hooker HD (1917) Liebig’s Law of the Minimum in relation to general biological problems. Science 46:197–204
Houk P, Musburger C, Wiles P (2010) Water quality and herbivory interactively drive coral-reef recovery patterns in American Samoa. PLoS One 5:e13913, doi:10.1371/journal.pone.0013913
Hubbard DK (1988) Controls of modern and fossil reef development: common ground for biological and geological research. Proc 6th Intl Coral Reef Symp 1:243–252
Hubbard DK (1997) Reefs as dynamic systems. In: Birkeland C (ed) Life and death of coral reefs. New York: Chapman and Hall, pp 43–67
Hubbard DK, Zankl H, van Heerden I, Gill IP (2005) Holocene reef development along the northeastern St. Croix shelf, Buck Island, US Virgin Islands. J Sed Res 75:97–113
Hughes TP (1994) Catastrophes, phase shifts and large-scale degradation of a Caribbean coral reef. Science 265:1547–1551
Hughes TP, Baird AH, Bellwood DR, Card M, Connoll SR, Folke C, Grosberg R, Hoegh-Guldberg O, Jackson JBC, Kleypas J, Lough JM, Marshall P, Nyström M, Palumbi SR, Pandolfi JM, Rosen B, Roughgarden J (2003) Climate change, human impacts, and the resilience of coral reefs. Science 301:929–933
Hughes TP, Bellwood DR, Folke CS, McCook LS, Pandolfi JM (2007) No-take areas, herbivory and coral reef resilience. Trends Ecol Evol 22:1–3
Hughes TP., Graham NAJ, Jackson JBC, Mumby PJ, Steneck RS (2010) Rising to the challenge of sustaining coral reef resilience. Trends Ecol Evol 25:633–642
Hughes TP, Szmant AM, Steneck R, Carpenter R, Miller S (1999) Algal blooms on coral reefs: what are the causes? Limnol Oceanogr 44:1583–1586
Hughes TP, Tanner JE (2000) Recruitment failure, life histories, and long-term decline of Caribbean corals. Ecology 81:2250–2263
Huntington BE, Karnauskas M, Lirman D (2011) Corals fail to recover at a Caribbean marine reserve despite ten years of reserve designation. Coral Reefs 30:1077–1085
Idjadi JA, Haring RN, Precht WF (2010) Recovery of the sea urchin Diadema antillarum promotes scleractinian coral growth and survivorship on shallow Jamaican reefs. Mar Ecol Prog Ser 403:91–100
Idjadi JA, Lee SC, Bruno JF, Precht WF, Allen-Requa L, Edmunds PJ (2006) Rapid phase-shift reversal on a Jamaican coral reef. Coral Reefs 25:209–211
Jackson JBC (1992) Pleistocene perspectives on coral reef community structure. Am Zool 32:719–731
Jackson JBC (2008) Ecological extinction and evolution in the brave new ocean. Proc Natl Acad Sci 105(Suppl 1):11458–11465
Jackson JBC, Donovan MK, Cramer KL, Lam VV (eds) (2014) Status and trends of Caribbean coral reefs: 1970–2012. Gland: Global Coral Reef Monitoring Network, IUCN, 299 pp
Jackson JBC, Erwin DH (2006) What can we learn about ecology and evolution from the fossil record. Trends Ecol Evol 21:322–328
Jackson JBC, Johnson KG (2000) Life in the last few million years. Paleobiology 24 (Suppl):221–235
Jackson JBC, Kirby MX, Berger WH, Bjorndal KA, Botsford LW, Bourque BJ, Bradbury RH, Cooke R, Erlandson J, Estes JA, Hughe TP, Kidwell S, Lange CB, Lenihan HS, Pandolfi JM, Peterson CH, Steneck RS, Tegner MJ. Warner RR (2001) Historical overfishing and the recent collapse of coastal ecosystems. Science 293:629–638
Johannes RE, Wiebe WJ, Crossland CJ, Rimmer DW, Smith SV (1983) Latitudinal limits of coral reef growth. Mar Ecol Prog Ser 11:105–111
Jones GP, McCormick MI, Srinivasan M, Eagle JV (2004) Coral decline threatens fish biodiversity in marine reserves. Proc Natl Acad Sci 101:8251–8253
Kennedy EV, Perry CT, Halloran PR, Iglesias-Prieto R, Schönberg CHL, Wisshak M, Form AU, Carricart-Ganive JP, Fine M, Eakin CM, Mumby PJH (2013) Avoiding coral reef functional collapse requires local and global action. Curr Biol 23:912–918
Kidwell SM (2001) Preservation of species abundance in marine death assemblages. Science 294:1091–1094
Kidwell SM (2007) Discordance between living and death assemblages as evidence for anthropogenic ecological change. Proc Natl Acad Sci USA 104:17701–17706
Kiessling W, Simpson C (2011) On the potential for ocean acidification to be a general cause of ancient reef crises. Global Change Biol 17:56–67
Kleypas J (1997) Modeled estimates of global reef habitat and carbonate production since the last glacial maximum. Paleoceanography 12:533–545
Kleypas JA, Buddemeier RW, Archer D, Gattuso J-P, Langdon, Opdyke, B (1999) Geochemical consequences of increased atmospheric carbon dioxide on coral reefs. Science 284:118–120
Kleypas JA, Buddemeier RW, Gattuso J-P (2001) The future of coral reefs in an age of global change. Intl J Earth Sci (Geol Rdsch) 90:426–437
Kline DI, Vollmer SV (2011) White Band Disease (type I) of endangered Caribbean acroporid corals is caused by pathogenic bacteria. Nature Sci Rep 1:7, doi:10.1038/srep00007
Knowlton N, Jackson JBC (2008) Shifting baselines, local impacts, and global change on coral reefs. PLoS Biology 6:e54, doi:10.1371/journal.pbio.0060054
Knowlton N, Lang JC, Rooney MC, Clifford P (1981) Evidence for delayed mortality in hurricane-damaged Jamaican staghorn corals. Nature 294:251–252
Kramer KL, Heck KL Jr. (2007) Top-down trophic shifts in Florida Keys patch reef marine protected areas. Mar Ecol Prog Ser 349:111–123
Kuntz NM., Kline DI, Sandin SA, Rowher F (2005) Pathologies and mortality rates caused by organic carbon and nutrient stressors in three Caribbean coral species. Mar Ecol Prog Ser 294:173–180
Lafferty KD, Porter, J, Ford SE (2004) Are diseases increasing in the ocean? Ann Rev Ecol Evol Syst 35:31–54
Lescinsky H (2012) Partial mortality and re-sheeting in fossil corals: a disease proxy. Proc 12th Intl Coral Reef Symp 3:1–5
Lescinsky HB, Titus B, Hubbard D (2012) Live coral cover from the fossil record: an example from Holocene reefs of the Dominican Republic. Coral Reefs 31:335–346
Lewis JB (1960) The coral reefs and coral communities of Barbados, WI. Can J Zool 38:1133–1145
Lewis JB (1984) The Acropora inheritance: a reinterpretation of the development of ringing reefs in Barbados, West Indies. Coral Reefs 3:117–122
Lighty RG, Macintyre IG, Stuckenrath R (1978) Submerged early Holocene barrier reef south-east Florida shelf. Nature 276:59–60
Lowe PK, Bruno JF, Selig ER, Spencer M (2011) Empirical models of transitions between coral reef states: effects of region, protection, and environmental change. PLoS ONE 6:e26339, doi:10.1371/journal.pone.0026339
Macintyre IG, Aronson RB (2006) Lithified and unlithified Mg-calcite precipitates in tropical reef environments. J Sed Res 76:81–90
Macintyre IG, Glynn PW, Toscano MA (2007) The demise of a major Acropora palmata bank–barrier reef off the southeast coast of Barbados, West Indies. Coral Reefs 26:765–773
MacKenzie CL Jr (2007.) Causes underlying the historical decline in eastern oyster (Crassostrea virginica Gmelin, 1791) landings. J Shellfish Res 26:927–938
Manzello DP, Kleypas JA, Budd DA, Eakin CM, Glynn PW, Langdon C (2008) Poorly cemented coral reefs of the eastern tropical Pacific: possible insights into reef development in a high-CO2 world. Proc Natl Acad Sci 105:10450–10455
McClanahan TR (2008) Response of the coral reef benthos and herbivory to fishery closure and the 1998 ENSO disturbance. Oecologia 155:169–177
McClanahan TR, Cinner JE, Graham NAJ, Daw TM, Maina J, Stead SM, Wamukota A, Brown K, Venus V, Polunin NVC (2009) Identifying reefs of hope and hopeful actions: contextualizing environmental, ecological, and social parameters to respond effectively to climate change. Cons Biol 23:662–671
McClanahan TR, Cinner JE, MainaJ, Graham NAJ, Daw TM, Stead, SM, Wamukota A, Brown K, Ateweberhan M, Venus V, Polunin NVC (2008) Conservation action in a changing climate. Cons Lett 1:53–59
McClanahan TR Graham NAJ, McNeil MA, Muthiga NA, Cinner JA, Bruggeman JH, Wilson, SK (2011) Critical thresholds and tangible targets for ecosystem-based management of coral reef fisheries Proc Natl Acad Sci USA 108:17230–17233
McClanahan TR, Huntington BE, Cokos B, (2011) Coral responses to macroalgal reduction and fisheries closure on Caribbean patch reefs. Mar Ecol Prog Ser 437:89–102
McClanahan TR, Maina J, Starge JCr, Herron-Perez P, Dusek E (2005) Detriments to post-bleaching recovery of corals. Coral Reefs 24:230–246
McClanahan TR., Muthiga NA, Mangi S (2001) Coral and algal changes after the 1998 coral bleaching: interaction with reef management and herbivores on Kenyan reefs. Coral Reefs 19:380–391
McCook LJ, Ayling T, Cappo M, Choat JH, Evans RD, De Freitas DM, Heupel M, Hughes TP, Jones GP, Mapstone B, Marsh H, Mills M, Molloy FJ, Pitcher CR, Pressey RL, Russ GR, Sutton S, Sweatman H, Tobin R, Wachenfeld DR,, Williamson DH (2010)Adaptive management of the Great Barrier Reef: a globally significant demonstration of the benefits of networks of marine reserves. Proc Natl Acad Sci USA 107:18278–18285
McWilliams JP, Côté IM, Gill JA, Sutherland WJ, Watkinson. AR (2005) Accelerating impacts of temperature-induced coral bleaching in the Caribbean. Ecology 86:2055–2060
Miller J, Muller E, Rogers C, Waara R, Atkinson A, Whelan KRT, Patterson M, Witcher B (2009) Coral disease following massive bleaching in 2005 causes 60 % decline in coral cover on reefs in the US Virgin Islands. Coral Reefs 28:925–937
Montaggioni LF, Braithwaite CJR (2009) Quaternary Coral Reef Systems: History, Development Processes and Controlling Factors. Amsterdam: Elsevier, 532 pp
Mora C, Andréfouët S, Costello MJ, Kranenburg C, Rollo A, Veron J, Gaston JK, Myers RA (2006) Coral reefs and the global network of marine protected areas. Science 312:1750–1751
Mora C, Metzger R, Rollo A, Myers RA (2007) Experimental simulations about the effects of overexploitation and habitat fragmentation on populations facing environmental warming. Proc R Soc B 274:1023–1028
Mumby PJ. (2006) The impact of exploiting grazers (Scaridae) on the dynamics of Caribbean coral reefs. Ecol Appl 16:747–769
Mumby PJ, Elliott IA, Eakin CM, Skirving W, Paris CB, Edwards HJ, Enríquez S, Iglesias- Prieto R, Cherubin LM, Stevens JR (2011) Reserve design for uncertain responses of coral reefs to climate change. Ecol Lett 14:132–140
Mumby PJ, Harborne, Williams AJ, Cappel CV, Brumbaugh DR, Micheli F, Holmes KE, Dahlgren CP, Paris CB, Blackwell PG (2007a) Trophic cascade facilitates coral recruitment in a marine reserve. Proc Natl Acad Sci 104:8362–8367
Mumby PJ, Hastings A, Edwards HJ (2007b) Thresholds and the resilience of Caribbean coral reefs. Nature 450:98–101
Murdoch TJT (2007) A functional group approach for predicting the composition of hard coral assemblages in Florida and Bermuda. PhD Dissertation. Mobile: University of South Alabama, 299 pp
Myers MR, Ambrose RF (2009) Differences in benthic cover inside and outside marine protected areas on the Great Barrier Reef: influence of protection or disturbance history? Aquat Conserv Mar Freshw Ecosys 19:736–747
National Marine Fisheries Service (2015) Recovery Plan for Elkhorn (Acropora palmata) and Staghorn (A. cervicornis) Corals. Prepared by the Acropora Recovery Team for the National Marine Fisheries Service, Silver Spring, Maryland.
Neumann AC, Macintyre IG (1985) Reef response to sea level rise: keep-up, catch-up or give-up. Proc 5th Intl Coral Reef Congr 3:105–110
Norris RD, Turner SK, Hull PM, Ridgwell A (2013) Marine ecosystem responses to Cenozoic global change. Science 341:492–498
Paddack M, Reynolds J, Aguilar C, Appledoor R, Beets J, Burkett EW, Chittar PM, Clarke K, Esteves R, Fonseca AC, Forrester GE, Friedlander AM, García-Sais J, González-Sansón G, Jordan LKB, McClellan DB, Miller MW, Molloy PP, Mumby PJ, Nagelkerken I, Nemeth M, Navas-Camacho R, Pitt J, Polunin NVC, Reyes-Nivia MC, Robertson DR, Rodríguez-Ramírez Salas AE, Smith SR, Spieler R, Steele MA, Williams ID, Wormald CL, Watkinson AR, Côté IM (2009) Recent region-wide declines in Caribbean reef fish abundance. Curr Biol 19:590–595
Pandolfi JM (1996) Limited membership in Pleistocene reef coral assemblages from the Huon Peninsula, Papua New Guinea: constancy during global change. Paleobiology 22:152–176
Pandolfi JM, Bradbury RH, Sala E, Hughes TP, K. Bjorndal A, Cooke RG, McArdle D, McClenachan L, Newman MJH, Paredes G, Warner RR, Jackson JBC (2003) Global trajectories of the long-term decline of coral reef ecosystems. Science 301:955–958
Pandolfi JM, Connolly SR, Marshall DJ, Cohen AL (2011) Projecting coral reef futures under global warming and ocean acidification. Science 333:418–422
Pandolfi JM, Jackson JBC (2001) Community structure of Pleistocene coral reefs of Curaçao, Netherlands Antilles. Ecol Monogr 71:49–67
Pandolfi JM, Jackson JBC (2006) Ecological persistence interrupted in Caribbean coral reefs. Ecol Lett 9:818–826
Peterson C, Jackson J, Kirby M, Lenihan H, Bourque B, Bradbury R, Cooke R, Kidwell S (2001) Factors in the decline of coastal ecosystems. Science 293:1590–1591
Planes S, Galzin R, Bablet J-P, Sale PF (2005) Stability of coral reef fish assemblages impacted by nuclear tests. Ecology 86:2578–2585
Porter JW, Battey JF, Smith GI (1982) Perturbation and change in coral reef communities. Proc. Natl Acad Sci 79:1678–1681
Porter JW, Woodley JD, Smith GJ, Neigel JE, Battey JF, Dallmeyer DG (1981) Population trends among Jamaican reef corals. Nature 294:249–250
Precht WF, Aronson RB (2004) Climate flickers and range shifts of reef corals. Front Ecol Environ 2:307–31
Precht WF, Aronson RB (2006) Death and resurrection of Caribbean coral reefs: a palaeoecological approach. In: Côté IM, Reynolds JD (eds) Coral reef conservation. Cambridge: Cambridge University Press. pp 40–77
Precht WF, Miller SL (2007) Ecological shifts along the Florida Reef Tract: the past as the key to the future. In: Aronson RB (ed) Geological approaches to coral reef ecology. New York: Springer-Verlag, pp 237–312
Precht WF, Robbart ML, Aronson RB (2004) The potential listing of Acropora species under the US Endangered Species Act. Mar Poll Bull 49:534–536
Quinn JF, Dunham AE (1983) On hypothesis testing in ecology and evolution. Am Nat 122:602–617
Randall CJ, van Woesik R (2015) Contemporary white-band disease in Caribbean corals driven by climate change. Nature Clim Change 5:375–379
Riegl B, Piller WE (2003) Possible refugia for reefs in times of environmental stress. Int J Earth Sci (Geol Rdsch) 92:520–531
Roberts HH, Wilson PA, Lugo-Fernández A (1992) Biologic and geologic responses to physical processes: examples from modern reef systems of the Caribbean–Atlantic region. Cont Shelf Res 12:809–834
Roff G, Clark TR, Reymond CE, Zhao J-X, Feng Y, McCook LJ, Done TD, Pandolfi JM (2013) Palaeoecological evidence of a historical collapse of corals at Pelorus Island, inshore Great Barrier Reef, following European settlement. Proc R Soc B 280, doi: 10.1098/rspb.2012.2100
Rogers CS (1990) Responses of coral reefs and reef organisms to sedimentation. Mar Ecol Prog Ser 62:185–202
Rogers CS (2008) Coral bleaching and disease should not be underestimated as causes of Caribbean coral reef decline. Proc R Soc B 276:197–198
Rogers CS (2013) Coral reef resilience through biodiversity. ISRN Oceanography 2013:739034, doi: 10.5402/2013/739034
Rogers CS, Miller J (2013) Coral diseases cause reef decline. Science 340:1522
Rosenberg E, Ben-Haim Y (2002) Microbial diseases of corals and global warming. Env Microbiol 4:318–326
Sala E, Jackson JBC (2011) Lessons from coral reefs. In: Jackson JBC, Alexander KE, Sala E (eds) Shifting baselines: the past and future of ocean fisheries, Washington, DC: Island Press, pp 193–203
Sammarco PW (1982) Echinoid grazing as a structuring force in coral communities: whole-reef manipulations. J Exp Mar Biol Ecol 61:31–55
Sammarco PW, Strychar KB (2009) Effects of climate change/global warming on coral reefs: adaptation/exaptation in corals, evolution in zooxanthellae, and biogeographic shifts. Environ Bioindic 4:9–45
Sandin SA, McNamara DE (2012) Spatial dynamics of benthic competition on coral reefs. Oecologia 168:1079–1090
Sandin SA, Smith JE, DeMartini EE, Dinsdale EA, Donner SD, Friedlander AM, Konotchik T, Malay M, Maragos JE, Obura D, Pantos O, Paulay G, Richie M, Rohwer F, Schroeder RE, Walsh S, Jackson JBC, Knowlton N, Sala E (2008) Baselines and degradation of coral reefs in the northern Line Islands. PloS One 3(2): e1548, doi:10.1371/journal.pone.0001548
Schutte VGW, Selig ER, Bruno JF (2010) Regional spatio-temporal trends in Caribbean coral reef benthic communities. Mar Ecol Prog Ser 402:115–122
Selig ER, Bruno JF (2010) A global analysis of the effectiveness of marine protected areas in preventing coral loss. PLoS One 5:e9278, doi:10.1371/journal.pone.0009278
Selig ER, Casey KS, Bruno JF (2012) Temperature-driven coral decline: the role of marine protected areas. Global Change Biol 18:1561–1570
Selig ER, Harvell CD, Bruno JF, Willis BL, Page CA, Casey KS, Sweatman H (2006) Analyzing the relationship between ocean temperature anomalies and coral disease outbreaks at broad spatial scales. In: Phinney JT, Hoegh-Guldberg O, Kleypas J, Skirving W, Strong A (eds) Coral reefs and climate change: science and management, Washington, DC: American Geophysical Union, pp 118–128
Shears, NT, Babcock RC (2003) Continuing trophic cascade effects after 25 years of no-take marine reserve protection. Mar Ecol Prog Ser 246:1–16
Stockwell B, Jadloc CRL, Abesamis RA, Alcala AC, Russ GR (2009) Trophic and benthic responses to no-take marine reserve protection in the Philippines. Mar Ecol Prog Ser. 389:1–15
Stoddart DR (1963) Effects of Hurricane Hattie on the British Honduras reefs and cays, October 30–31, 1961. Atoll Res Bull 95:1–142
Sweet MJ, Croquer A, Bythell JC (2014) Experimental antibiotic treatment identifies potential pathogens of white band disease in the endangered Caribbean coral Acropora cervicornis. Proc R Soc B 281:20140094, doi: 10.1098/rspb.2014.0094
Tager D, Webster JM, Potts DC, Renema W, Braga JC, Pandolfi JM (2010) Community dynamics of Pleistocene coral reefs during alternative climatic regimes. Ecology 91:191–200
Thompson DM, van Woesik R (2009) Corals escape bleaching in regions that recently and historically experienced frequent thermal stress. Proc R Soc B 276:2893–2901
Toscano MA (2016) Revised paleoenvironmental analysis of the Holocene portion of the Barbados sea-level record: Cobbler's Reef revisited. Coral Reefs. doi:10.1007/s00338-016-1397-0
Toscano MA, Macintyre IG (2003) Corrected western Atlantic sea-level curve for the last 11,000 years based on calibrated 14C dates from Acropora palmata framework and intertidal mangrove peat. Coral Reefs 22:257–270
Toth LT, Aronson RB, Cobb KM, Cheng H, Edwards RL, Grothe PR, Sayani HR (2015) Climatic and biotic thresholds of coral-reef shutdown. Nature Clim Change 5:369–374
Toth LT, Aronson RB, Vollmer SB, Hobbs JW, Urrego DH, Cheng H, Enochs IC, Combosch DJ, van Woesik R, Macintyre IG (2012) ENSO drove 2500-year collapse of eastern Pacific coral reefs. Science 337:81–84
Toth LT, van Woesik R, Murdoch TJT, Smith SR, Ogden JC, Precht WF, Aronson RB (2014) Do no-take reserves benefit Florida’s corals? 14 years of change and stasis in the Florida Keys National Marine Sanctuary. Coral Reefs 33:565–577
Urban MC, Tewksbury JJ, Sheldon KS (2012) On a collision course: competition and dispersal differences create no-analogue communities and cause extinctions during climate change. Proc R Soc B 279:2072–2080
van Duyl FC (1985) Atlas of the living reefs of Curaçao and Bonaire (Netherlands Antilles). Utrecht: Natuurwetenschappelijke Studiekring voor Suriname en de Nederlandse Antillen [Foundation for Scientific Research in Surinam and the Netherlands Antilles], no. 117, 37 pp
van Woesik R, Jordán-Garza AG (2011) Coral populations in a rapidly changing environment. J Exp Mar Biol Ecol 408:11–20
Veron JEN (2008) Mass extinctions and ocean acidificaiton: geological constraints on biological dilemmas. Coral Reefs 27:459–472
Vollmer SV, Kline DI (2008) Natural disease resistance in threatened staghorn corals. PLoS One 3:e3718, doi:10.1371/journal.pone.0003718
Vroom PS, Page KN, Kenyon JC, Brainard BE (2006) Algae-dominated reefs. Am Sci 94:430–437
Wapnick CM, Prech WF, Aronson RB (2004) Millennial-scale dynamics of staghorn coral at Discovery Bay, Jamaica. Ecol Lett 7:354–361
Waycott M, Duarte CM, Carruthers TJB, Orth RJ, Dennison WC, Olyarnik S, Calladine A, Fourqurean JW, Heck KL Jr., Hughes AR, Kendrick GA, Kenworthy WJ, Short FT, Williams SL (2009) Accelerating loss of seagrasses across the globe threatens coastal ecosystems. Proc Natl Acad Sci 106:12377–12381
Webster JM, Wallace L, Silver E, Potts D, Braga JC, Renema W, Riker-Coleman K, Gallup C (2004) Coralgal composition of drowned carbonate platforms in the Huon Gulf, Papua New Guinea; implications for lowstand reef development and drowning. Mar Geol 204:59–89
Weil E, Smith G, Gil-Agudelo DL (2006) Status and progress in coral reef disease research. Dis Aquat Org 69:1–7.
Wood, R (1999) Reef evolution. Oxford: Oxford University Press, 414 pp
Wood R (2007) The changing fate of coral reefs: lessons from the deep past. In: Aronson RB (ed) Geological approaches to coral reef ecology, New York: Springer-Verlag, pp 3–27
Woodley JD (1989) The effects of Hurricane Gilbert on coral reefs at Discovery Bay. In: Bacon PR (ed.) Assessment of the Economic Impacts of Hurricane Gilbert on Coastal and Marine Resources in Jamaica, Nairobi: UNEP Regional Seas Reports and Studies 110, United Nations Environmental Programme, pp 71–73
Woodley JD (1992) The incidence of hurricanes on the north coast of Jamaica since 1870: are the classic reef descriptions atypical? Hydrobiologia 247:133–138
Woodley JD, Chornesky EA, Clifford PA, Jackson JBC, Kaufman LS, Knowlton N, Lang JC, Pearson MP, Porter JW, Rooney MC, Rylaarsdam KW, Tunnicliffe VJ, Wahle CM, Wulff JL, Curtis AGS, Dallmeyer MD, Jupp BP, Koehl MAR, Neigel J, Sides EM (1981) Hurricane Allen’s impact on Jamaican coral reefs. Science 214:749–755
Żychaluk K, Bruno JF, Clancy D, McClanahan TR, Spencer M (2012) Data-driven models for regional coral-reef dynamics. Ecol Lett 15:151–158
Acknowledgments
We thank M. E. S. Bracken, J. F. Bruno, N. L. Hilbun, D. K. Hubbard, L. Kaufman, L. T. Toth, S. Thatje, and R. van Woesik for advice and discussion. R. M. Moody and L. T. Toth drafted the figures. Our research on coral reefs has been supported over the years by the Smithsonian Institution, the National Geographic Society, Northeastern University’s Three Seas Program, and the U.S. National Science Foundation (most recently grant OCE-1535007 to R.B.A.). This is contribution number 75 from the Institute for Research on Global Climate Change at the Florida Institute of Technology.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Aronson, R.B., Precht, W.F. (2016). Physical and Biological Drivers of Coral-Reef Dynamics. In: Hubbard, D., Rogers, C., Lipps, J., Stanley, Jr., G. (eds) Coral Reefs at the Crossroads. Coral Reefs of the World, vol 6. Springer, Dordrecht. https://doi.org/10.1007/978-94-017-7567-0_11
Download citation
DOI: https://doi.org/10.1007/978-94-017-7567-0_11
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-017-7565-6
Online ISBN: 978-94-017-7567-0
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)