Abstract
Macrophages undergo fusion with other macrophages to form the hallmark multinucleated giant cells of chronic inflammation. However, neither the existence of distinct morphological types of giant cells, the signaling pathways that induce their formation, the molecular mechanism(s) of macrophage fusion, nor the significance of macrophage multinucleation at chronic inflammatory sites are well understood. Our efforts have been focused on these unknowns, particularly as they relate to the foreign body-type giant cells that form on implanted biomaterials and biomedical devices. We have pursued the discoveries of human macrophage fusion factors (interleukin-4, interleukin-13, α-tocopherol) with emphasis on foreign body giant cells, and identified adhesion receptors and signaling intermediates, as well as an adhesion protein substrate (vitronectin) that supports macrophage fusion. Studies on the molecular mechanism of macrophage fusion have revealed it to be a mannose receptor-mediated phagocytic process with participation of the endoplasmic reticulum. Further phenotypic and functional investigations will foster new perspectives on these remarkable multinucleated cells and their physiological significances in multiple inflammatory processes.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Giant Cell
- Connective Tissue Growth Factor
- Adhesion Structure
- Foreign Body Giant Cell
- Diacylglycerol Kinase
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
7.1 Introduction
Multinucleated giant cells have long been regarded as hallmark indicators of chronic inflammatory processes. As early as 1868, Langhans reported unusual giant cells containing multiple peripherally-arranged nuclei in the granulomas of tuberculosis [1]. Other than this long-standing link with chronic inflammation, however, we know relatively little about these intriguing cells and even less about why they appear where and when they do.
In many cases where giant cells are observed, there is a definable pathological agent, such as in tuberculosis, in which the causative organism is a mycoplasma. Additional examples of known causes are persistent bacterial, viral, parasitic, or fungal infections. In other cases, giant cells arise where the chronic inflammatory cause is not precisely known, for example, in sarcoidosis, rheumatoid arthritis, and certain neoplasias [2]. Of particular interest for our research, giant cells appear where there is a non-phagocytosable foreign body in the form of an implanted biomedical device or biomaterial [3]. In fact, so-called foreign body giant cells have been observed to interface with vascular, cardiovascular, orthopedic, and breast prostheses for periods extending to 15 years [4] and to occupy as much as 25% of implant surface area [5]. Therefore, they are a prominent cell type on biomaterials and have been widely linked to the biodegradation of certain biomedical polymers in vivo.
From a cell biological perspective, the most interesting and striking feature of giant cells is that they are actually multinucleated macrophages, formed by macrophage fusion with other macrophages [6, 7]. Other well known examples of cell–cell fusion, such as myoblast fusion, sperm/ovum fusion, or osteoclast formation, are clearly function-driven aspects of normal physiology. In contrast, and because macrophages in their mononuclear form appear to be effective in other immune and inflammatory scenarios, the biological basis of macrophage multinucleation at various sites of chronic inflammation remains only speculative. For example, “frustrated phagocytosis” has been suggested as a driving force for multinucleation [8], which could potentially serve to combine phagocytic forces that are otherwise ineffective. An alternative possibility is that macrophage multinucleation might function to sequester a nonphagocytosable foreign body in order to protect host tissues from the adverse consequences of an on-going chronic inflammatory response. In a polarized cell type, as has been proposed by Vignery [9], both of these situations could be the case. Nevertheless, the single common denominator in these otherwise pathologically distinguishable scenarios appears to be the persistent, i.e. unresolvable by phagocytosis, presence of foreign microorganisms or materials. Beyond this, the precise molecular mechanism of macrophage fusion has not been elucidated, and the potential physiological significance of multinucleation itself, so key to understanding these cells, is as yet unclear.
7.2 Morphological Types of Multinucleated Giant Cells
A further dimension to these unknowns stems from the existence of morphological “variants” of multinucleated giant cells, of which there are two major recognized types. These have also long been observed, and yet their potential differences have been largely, and even surprisingly, overlooked. However, in order to advance our understanding of multinucleated macrophage biology, we must begin to view distinct morphological types as more than vague “variants” of the same thing. In keeping with structure/function relationships in biological systems, our in vitro findings with human macrophage fusion support this view and raise new questions on the potential significances of types of multinucleation.
The type of giant cell originally observed by Langhans is consistently circular or ovoid in shape with a limited number of nuclei, often arranged in a characteristic circular or semi-circular “horseshoe” pattern (Fig. 7.1a). These multinucleated cells vary somewhat in diameter but seldom exceed 50 microns, nor do they normally contain more than about 10–20 nuclei, which indicates a limited potential for macrophage fusion. Langhans-type giant cells (LGC) are commonly seen in association with granulomas due to chronic microbial infections [10].
Micrographic images of (a) LGC or (b) FBGC giant cell morphological types induced with IFN-γ + IL-3 or IL-4, respectively. The inset at lower left in (a) is an enlargement of the multinucleated cell seen at the top right corner of the same image. It demonstrates a random nuclear arrangement under LGC-inducing conditions, illustrating how classification of giant cells by morphology alone has generated confusion in the literature
Quite unmistakably distinct from LGC are the foreign body-type giant cells (FBGC) which are found interfacing with foreign materials such as surgical sutures or implanted biomedical devices (Fig. 7.1b). These exhibit an irregularly-shaped cytoplasm which is highly variable in size. Numbers of nuclei in FBGC are also widely variable and range from ten to many tens to even hundreds within a cytoplasm that may exceed one millimeter in diameter [5, 11]. This suggests that, unlike LGC, a mechanism to restrict degrees of fusion does not operate in FBGC. In further contrast to LGC, FBGC exhibit no definable patterns of nuclear arrangments, with multiple nuclei randomly scattered throughout the extensive cytoplasm of these irregularly-shaped cells.
There also exists a gray area between these two readily identifiable giant cell types, which is occupied by multinucleated cells with relatively few nuclei (usually around 3–10) and no particular pattern of nuclear arrangement (for example, the multinucleated cell in the top right corner of Fig. 7.1a, which is enlarged in the inset at lower left). This is the cell type that is responsible for apparent confusion in the literature because: (1) the classification of giant cells is as yet insufficiently based almost completely on morphology, and (2) macrophage fusion leading to giant cell formation is a morphological continuum, of which we can glimpse only part. Although their random nuclear arrangements usually place these cells in the foreign body giant cell category, this may, in fact, be misleading. Obviously, they could be precursors to larger and more highly multinucleated FBGC. However, they may actually be LGC precursors, i.e in the early stages of formation, post several fusion events but prior to the circular or semi-circular arrangement of nuclei. Finally, they could represent a non-Langhans-, non-foreign body-type giant cell. Further studies focused on the phenotypes and functions of morphologic types of giant cells are rquired to illuminate these unknowns.
7.3 Differential Signaling Pathways for Macrophage Multinucleation
In our in vitro studies with human monocyte-derived macrophages, we were able to demonstrate that the FBGC and LGC morphologies clearly arise under the influences of very different cytokines. As depicted in Fig. 7.2, FBGC can be induced by interleukin (IL)-4 [11] or IL-13 [12], or, as we later discovered, by a non-cytokine moiety, α-tocopherol [13]. Alternatively, LGC generation is mediated by interferon (IFN)-γ plus a macrophage maturation factor [11, 14]. The FBGC or LGC generated in our parallel in vitro systems were morphologically indistinguishable from those observed adherent to implanted biomaterials or in association with infectious granulomas, respectively. Both IL-4 and IFN-γ had been previously linked to macrophage fusion [14–17]. It was difficult to compare these investigations, however, because of considerable variations in cell sources and culture conditions. We demonstrated that, under otherwise identical culture conditions, these two distinct cytokines induced two morphologically distinguishable types of giant cells from human blood monocyte-derived macrophages [11]. This indicated, for the first time, that the occurrence of distinct types of giant cells at chronic inflammatory sites may represent different host responses to diverse inflammatory stimuli.
Morphologically distinct types of giant cells are induced by differential cytokine signals from human blood monocyte-derived macrophages in vitro. IL-4, IL-13, or α-tocopherol promotes the formation of foreign body-type giant cells (FBGC) on implanted biomaterials. FBGC vary greatly in cytoplasmic areas and numbers of randomly-arranged nuclei. In contrast, Langhans-type giant cells (LGC) are induced by IFN-γ in the presence of a macrophage maturation factor such as GM-CSF, M-CSF, or IL-3. LGC are relatively much smaller and are characterized by a circular or semi-circular arrangement of nuclei within an ovoid cytoplasm. At longer culture times, LGC develop pyknotic nuclei, indicating apoptosis, whereas pyknotic nuclei are not observed in FBGC
7.3.1 Interleukin-4 and Interleukin-13
Our early efforts to establish an in vitro system of human macrophage fusion that would duplicate FBGC morphology were aided by a study from McInnis and Rennick [15], who, working with IL-4-treated mouse bone marrow macrophages, were able to achieve a fusion rate of approximately 10%. However few, the FBGC that they generated were morphologically very similar to the FBGC observed on retrieved biomedical materials. Other investigators had, up to that point, also reported varying degrees of human macrophage fusion using INF-γ [14, 16] or supernatant from lectin-stimulated mononuclear leukocytes [17–19], but these cultures mainly appeared to produce “polykaryons” which bore no resemblance to giant cells observed in vivo [16] or Langhans-type multinucleated cells [14, 17], which are not oberved interfacing with implanted biomaterials [3]. In two of these studies, each attempted to extend the finding of McInnis and Rennick from mouse to human monocytes/macrophages. They were each unsuccessful, concluding that IL-4 was not a human macrophage fusion factor and that the effect observed by McInnis and Rennick was a species-specific one [14, 17]. However, these investigators had added IL-4 directly to human monocytes in culture, which actually inhibits monocyte adhesion [20]. Instead, our approach provided a period of monocyte-to-macrophage development prior to the addition of IL-4. Thus, we were able to demonstrate that IL-4 is indeed a potent human macrophage fusion factor [11], and that the process of IL-4-induced FBGC formation requires a degree of macrophage development prior to the induction of fusion. We initially found that this could be achieved by the inclusion of macrophage maturation factors, such as granulocyte-macrophage colony-stimulating factor (GM-CSF), macrophage (M)-CSF, or IL-3 in the cultures [11]. However, mauration factors do not, by themselves, induce fusion and are not required for IL-4-induced fusion per se. We later learned that if monocytes/macrophages were cultured on chemically supportive surfaces, such as arginine-glycine-aspartate (RGD)-modified cell culture polystyrene, the addition of IL-4 only was sufficient to induce FBGC formation [21]. At the time, IL-13 was also emerging as alternative macrophage activation cytokine with multiple activities that were similar to IL-4. Therefore, we tested IL-13 under the same culture conditions that had been established for IL-4 and discovered that it, too, was a potent human macrophage fusion factor that, by itself, could induce the formation of FBGC in a manner indistinguishable from that of IL-4 [22]. In vivo studies confirmed a role for IL-4 in FBGC formation on biomaterials [23]. Our more recent in vivo efforts with athymic (nude) mice suggest that Th2 lymphocytes are not the source of IL-4/IL-13 [24]. Other possibilities include mast cells, eosinophils, basophils, natural killer (NK) lymphocytes, and NKT lymphocytes [25, 26]. These avenues remain to be addressed.
7.3.2 Interferon-γ
In parallel cultures and under identical conditions, we tested IFN-γ as a fusion factor compared to IL-4. We found that IFN-γ induced fusion leading to the formation of LGC only [11]. Unlike FBGC formation, IFN-γ-mediated fusion does require the cocommitant presence of a macrophage maturation factor, which can be either granulocyte-macrophage colony-stimulating factor (GM-CSF), macrophage-CSF, or IL-3 [11]. As illustrated in Fig. 7.2, LGC appear to have a limited lifespan, as nuclear pyknosis occurs with continued culture times, whereas FBGC cultures do not develop pyknotic nuclei (our unpublished observations). In the presence of IL-4, apoptosis does not occur [27], and the resultant FBGC exhibit morphologically normal nuclei.
7.3.3 α-Tocopherol
It was also intriguing that vitamin E incorporated in a poly(etherurethane) biomaterial induced increases in FBGC formation yet decreases in biomaterial degradation in vivo ( [28] and unpublished data). This was the first evidence to disconnect IL-4-induced macrophage fusion from biomedical polymer degradation. In vitro studies to directly pursue this revealed that vitamin E (90% α-tocopherol) moderately induced macrophage fusion and increased IL-4-induced FBGC formation. However, the purified α-tocopherol isomer alone most remarkably induced macrophage fusion, leading to cultures of confluent FBGC below normal plasma tocopherol concentrations [13]. This was not the case with the structurally similar antioxidants probucol or Trolox, suggesting that the α-tocopherol effects on FBGC formation were independent of its antioxidant activity. In this regard, multiple activities of α-tocopherol have been described that are independent of its antioxidant properties, including effects on phospholipase A2, protein kinase C (PKC), adhesion, and diacylglycerol kinase activity ([29] and see below). This study revealed that α-tocopherol is yet a third as well as the most potent human macrophage fusion factor, inducing striking multinucleated giant cells of the foreign body-type.
7.4 Mechanisms of Adhesion that Support FBCG Formation
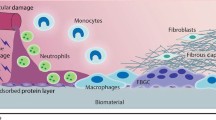
Based on our collective experiences, monocyte-derived macrophage fusion leading to FBGC or LGC formation requires adhesion success. This is highly dependent on culture material surface chemistry and/or adsorbed blood proteins [21, 30]. Culture materials that do not support initial monocyte adhesion and its ensuing macrophage morphological development (cytoplasmic expansion) beyond monocyte adhesion cannot support giant cell formation. Apparently, this requires the engagement of select integrins with appropriate adsorbed protein ligands to initiate activation of specific adhesion signals. Our investigations on adhesion mechanisms that support FBGC formation have focused on specific components of adhesion success. These include the adsorption of appropriate blood proteins or the provision of a supportive culture material surface, the identification of relevant adhesion receptors (integrins), integrin-mediated signaling (adhesion kinases), cytoskeletal responses (microfilaments and microtubules), and the assembly of focal adhesion structures (podosomes). In addition, we have pursued mechanisms of material-dependent adhesion failure, or anoikis (apoptosis) as a means of intervention in the process of FBGC formation on biomaterials.
7.4.1 Adhesion Receptors (Integrins)
Initial efforts to identify adhesion receptors that support FBGC formation revealed that both β1 and β2 integrins mediated adhesion during IL-4-induced FBGC formation [31]. Initial monocyte adhesion required functional β2 integrins, whereas, during the IL-4 induction of macrophage fusion, an additional dependence on functional β1 integrins was acquired. Of note, we did not find a functional role for β3 integrins in FBGC formation, nor did we detect β3 integrin in our culture system. This indicated that FBGC adhesion differs from that of osteoclasts, which utilize αVβ3 integrins for adhesion to bone [32]. Subsequent studies focused on the identities of the α integrin partners to these heterodimeric adhesion receptors [33]. Immunoprecipitation of fusing macrophage/FBGC lysates with anti-β1 integrin and immunoblotting revealed the presence of α5 and αV, as well as α2 and α3. As expected, αM and αX immunoprecipitated with β2 but not with β1. We did not detect α4 or several other β integrin partners. Immunocytochemistry coupled with confocal microscopy indicated that α5 and αX are poorly expressed on day 0. However, following the induction of macrophage fusion by IL-4 on day 3, they were each readily detectable in fusing macrophages/FBGC on day 7. In contrast, αM and αV were present throughout the culture period, with very strong αM and αX expression on day 7. We also demonstrated expression and co-localization of α3, α5, or αV with β1 on fusing macrophages/FBGC at this time point as well as strong co-localization of αM and αX with β2 at cell–cell fusion interfaces. Therefore, IL-4-induced FBGC are characterized by the expression of αMβ2 and αXβ2 > α5β1 and αVβ1 > α3β1 and α2β1. Thus, monocytes/macrophages and FBGC express a select group of adhesion receptors with potential for binding to specific blood proteins that may adsorb to biomaterials and to extracellular matrix proteins, including complement C3, fibrin(ogen), fibronectin, Factor X, vitronectin, certain collagens, laminin, and perhaps others as new ligands for these receptors become known.
7.4.2 RGD and Vitronectin
Initially, we discovered that arginine-glycine-aspartate (RGD)-modified culture polystyrene supported optimal monocyte-to-macrophage development in the absence of any other macrophage maturation factors [21], suggesting that an adhesion protein(s) containing this prototypical cell attachment sequence [34] is critical for this morphological progression. Combined with our later integrin receptor findings, we directly addressed the identification of relevant ligand(s) for these adhesion receptors. The approach was to adsorb potential integrin protein ligands to polystyrene culture surfaces. We thereby found that IL-4-induced FBGC formation did not proceed in vitro on material adsorbed with complement C3bi, fibrinogen, plasma fibronectin, cell-derived (fibroblast) fibronectin, collagen types I or IV, or laminin [30]. Surprisingly, these proteins also completely restricted macrophage adhesion and development and FBGC formation on RGD-modified surfaces, our optimal FBGC substrate material. In striking contrast, FBGC formation readily occurred on adsorbed vitronectin, which contains the RGD cell attachment sequence, and this effect of vitronectin was comparable to FBGC formation on adsorbed RGD peptide. Therefore, although fusing macrophages/FBGC express several β1 and β2 integrins and thereby would appear to possess broad ligand binding potential, they selectively utilize specific adhesive proteins to support macrophage adhesion and fusion leading to FBGC formation. Although fibrinogen, fibronectin, collagens, and laminin are well known to support the adhesion of multiple other cell types, they do not support macrophage development leading to FBGC formation. These findings indicate that the optimal cell binding RGD adhesive sequence for FBGC formation is presented in the blood-derived protein vitronectin, and that the propensity for vitronectin adsorption may be one mechanism for the material surface chemistry dependency of FBGC formation on biomaterials.
7.4.3 Adhesion Kinases
Integrin activation is a process in which conformational changes that increase integrin ligand binding affinity occur [35]. In phagocytic cells, activation leads to integrin clustering, which promotes intracellular signaling pathways that collectively control cytoskeletal rearrangements and formation of adhesion structures. These, in turn, support cell mobility, survival, and synthetic abilities. The integrin signaling proline-rich tyrosine kinase-2 (PYK2) is a member of the focal adhesion kinase (FAK) family and is highly expressed in macrophages [36]. PYK2 is co-localized with paxillin, talin, vinculin, and αMβ2 integrin in cell adhesion structures termed podosomes (see below). PYK2 is tyrosine phosphorylated upon macrophage adhesion and has been functionally linked to integrin-mediated regulation of cell spreading and migration [36]. We have demonstrated strong signals for FAK and PYK2 in whole cell lysates of IL-4-induced fusing macrophages/FBGC, the expression of which each increases during macrophage development and FBGC formation [37]. Further, FBGC adhesion was abrogated by the tyrosine kinase inhibitor genistein and by the phosphatidylinositol-3-kinase inhibitors wortmannin and LY294002 [31].
7.4.4 Microfilaments and Microtubules
Our early inhibitor studies demonstrated that F-actin was necessary for IL-4- or IL-13-induced FBGC formation because cytochalasins, which disrupt actin microfilaments, inhibited macrophage fusion in a concentration-dependent manner [38]. Importantly, the concentrations of cytochalasins that interfered with fusion did not decrease macrophage adhesion, cytoplasmic spreading, or motility but did prevent internalization of yeast via mannose receptor-mediated phagocytosis. This indicated that the mechanism of fusion is related to phagocytosis (see below, Molecular Mechanism of Macrophage Fusion). Furthermore, nocodazole restricted macrophage fusion in a concentration-dependent manner, pointing to an additional role for microtubules in this phenomenon [39].
7.4.5 Podosomes
Podosomes are specialized macrophage adhesion structures located at the cell/substrate interface where actin microfilaments terminate at the cell membrane [40]. We have noted that podosomes are a striking feature of FBGC, which display dense peripheral rings of these adhesion structures [12]. Surrounding the actin microfilament core, podosomes are specifically associated with β2 integrins in the membrane and are composed of gelsolin, paxillin, talin, vinculin, and other proteins, such as the actin-bundling protein, fascin-1 [41]. Beta integrin subunit binding by talin is the last common step in integrin activation and is believed to represent the center of converging integrin activation signals leading to formation of adhesion structures [35]. Adhesive structural proteins (gelsolin, paxillin, talin, fascin-1) are detectable during macrophage development and strongly up-regulated in fusing macrophages/FBGC on RGD-modified surfaces in vitro [37].
7.4.6 Adhesion Failure (Anoikis)
The alternative to adhesion success is anoikis, which is defined as apoptosis specifically due to adhesion failure [42]. Anoikis is a normal biological mechanism for the control and regulation of cell proliferation and tissue development. Cell death signaling by this mechanism is believed to be regulated by the cytoskeleton. Our in vitro observations have been that monocytes initially adhere quite well to most surfaces. However, those that fail to maintain adhesion and undergo macrophage morphological development, i.e. cytoplasmic spreading, apparently do not survive and/or cannot form FBGC. Thus, whether adhesion signals that promote integrin clustering, adhesion kinase activation, cytoplasmic spreading, and adhesion structure (podosome) formation are initiated and maintained is evidently determined by the material adherence substrate. Exploiting this natural cellular phenomenon to better understand the material surface chemistry dependence of macrophage survival and FBGC formation on biomaterials, we have evaluated apoptosis on several types of modified materials by measuring early and late events in apoptosis. As initial studies with differentially-modified polystyrenes had indicated, macrophage development and FBGC formation are highly material surface-dependent [43]. We found that a mechanism for this phenomenon is the biomaterial surface chemistry-dependent induction of apoptosis [44–46]. In addition, we found that activation of caspase-3, an intermediate indicator of apoptosis signaling, in inflammatory cells under shear stress leads to cell detachment [47]. It is of particular interest that caspases are known to cleave gelsolin, an important component of podosomes, and thereby disrupt adhesive interactions, leading to apoptosis/anoikis [42].
7.5 Molecular Mechanism of Macrophage Fusion
7.5.1 A Role for Mannose Receptors (MR)
Upon the establishment of our in vitro system of IL-4-induced FBGC formation, we could begin to address the moleular mechanism of macrophage-macrophage fusion. Inasmuch as IL-4 was reported to most stongly induce MR on macrophages, a finding which fostered the concept of “alternative activation” [48], we tested the effects of previously described inhibitors of MR activity on IL-4-induced FBGC formation [49]. Patterns of inhibition were consistent with participation of MR in the mechanism of fusion, and MR were found to be specifically up-regulated by IL-4 in our culture system and concentrated at fusion interfaces [49]. The formation of LGC, induced by a cytokine-enriched supernatant from lectin-stimulated mononuclear leukocytes, was also inhibited by α-mannan [43], indicating that similar mechanisms of fusion may operate to induce both types of giant cells. At that time, the MR was already well known for its key role in innate immunity via the clearance of microorganisms bearing terminal mannose oligosaccharides from mammalian tissues. Our data revealed a novel function for this endocytic/phagocytic receptor in the formation of multinucleated macrophages.
7.5.2 A Phagocytic Mechanism for Fusion with Participation of the Endoplasmic Reticulum
We extended these findings as well as our previously demonstrated requirements for F-actin in macrophage fusion with similarities to phagocytosis [38] and found that macrophage fusion leading to FBGC formation exhibits multiple additional features of phagocytosis [39]. Exploiting multiple pharmacological inhibitors, we discovered critical roles for vacuolar-type ATPase, microtubules (see above) and the endoplasmic reticulum (ER) in macrophage fusion. Further, we found a specific requirement for the calcium-independent phospholipase A2 (iPLA2), but not calcium-dependent PLA2 (cPLA2), secretory PLA2 (sPLA2), cyclooxygenase, or lipoxygenase in the mechanism of fusion. Immunocytochemistry confirmed iPLA2 expression and absence of cPLA2 or sPLA2 expression in macrophages/FBGCs. As markers of ER-mediated phagocytosis, calnexin and calregulin were detectable on non-permeabilized fusing macrophages and also concentrated at fusion interfaces where they co-localized with actin in permeabilized macrophages/FBGCs. Furthermore, ER markers co-localized with concanavalin A reactivity, which is a marker of potential MR ligand, on non-permeabilized fusing macrophages, suggesting that the ER may present relevant MR ligand at fusion interfaces. These data demonstrated for the first time that the mechanism of macrophage fusion leading to formation of multinucleated giant cells occurs by ER-mediated phagocytosis. We believe that these findings reveal a mechanism by which such extensive degrees of cell–cell fusion and cytoplasmic spreading, characteristic features of these remarkable multinucleated cells, can take place.
7.5.3 Diacyl Glycerol (DG)-Dependent and -Independent PKCs
To further investigate the macrophage fusion signaling pathways that promote and support FBGC formation, we investigated the participation of PKC in the IL-4-induced fusion of human monocyte-derived macrophage in vitro [50]. The PKC inhibitors H-7, calphostin C, and GF109203X attenuated macrophage fusion, whereas H-8, which is more selective for PKA and PKG, did not. Macrophage fusion was also prevented by the phospholipase C inhibitor, Et-18-OCH3, the PKC isoform inhibitors GO6983 or rottlerin and by peptide inhibitors for PKC (20–28), PKCβ, or PKCζ but not by HBDDE or peptide inhibitors for PKCε or PKA. In cultures of fusing macrophages/FBGCs, we detected only PKCα, β, δ, and ζ by immunoprecipitation and immunoblotting, and we also observed strong expression of these isoforms by immunocytochemistry. Our collective results suggest that the γ, ε, η, μ, θ, or ι PKC isoforms are not required in the mechanism of IL-4-induced macrophage fusion; whether PKCα is required is unclear. However, new evidence is provided that FBGC formation is specifically supported by PKCβ, PKCδ, and PKCζ in combined diacylglycerol-dependent (PKCβ and PKCδ) and -independent (PKCζ) signaling pathways. Importantly, inhibition of PKCβ and PKCδ do not affect macrophage development or cytoplasmic spreading during monocyte-to-macrophage differentiation but interfere with FBGC formation at the point of macrophage fusion. In contrast, inhibition of PKCζ has more drastic effects on the morphological progression by restricting early events that must support macrophage development and cytoplasmic spreading. Whether PKCζ is also required for macrophage fusion itself cannot be determined from the present studies. The collective data from studies of PKCβ, δ, and ζ suggest that these isoforms play prominent roles in phagocytosis signaling as well as in migration and macrophage differentiation [51–54].
7.5.4 Diacylglycerol Kinase
Consistent with the reported activation of diacylglycerol kinase by α-tocopherol, we found that the diacylglycerol kinase inhibitor R59022 completely abrogates FBGC formation [13]. R59022 inhibition of IL-4-induced FBGC formation is reversed by α-tocopherol, suggesting that FBGC formation induced by both fusion factors requires diacylglycerol kinase activation. This study suggests a novel role for diacylglycerol kinase in the mechanism of macrophage fusion/FBGC formation at sites of chronic inflammation. Diacylglycerol kinase converts diacylglycerol to phosphatidic acid, which may act as a fusogenic lipid in membranes [55]. Consistent with this, we discovered that propranolol, which also promotes phosphatidate formation, greatly enhances IL-4-induced FBGC formation (unpublished data).
7.5.5 Matrix Metalloproteinase (MMP)-13
A role for MMPs in IL-4-induced macrophage fusion was indicated by pharmacological inhibition with actinonin, which inhibits aminopeptidases such as collagenase MMPs and blocks approximately 60% of macrophage fusion at 50 μM [56]. In addition, CL-82198, reported to be a selective inhibitor of MMP-13 that does not affect MMP-1 or MMP-9, strongly inhibits fusion by approximately 60% between 1 and 5 μM. Epigallocatechin gallate, which inhibits MMP-2, MMP-9, and MMP-12, and NNGH, which inhibits MMP-3 and MMP-12 do not restrict macrophage fusion at the relevant concentrations [56]. Finally, we confirmed that MMP-13 is detectable in lysates of fusing macrophages/FBGC (unpublished finding).
7.6 Phenotypic and Functional Profiles of Multinucleated Giant Cells
New perspectives continue to emerge on “classical” versus “alternative” macrophage activation, phenotype acquisition, and switching [57–60]. The concept of alternative macrophage activation was introduced to distinguish Th2 lymphokine (IL-4)-activated from Th1 lymphokine (IFN-γ)- or classically activated macrophages [48, 61]. IL-4 (as well as its relative IL-13) has evolved as a prototypical alternative macrophage activation signal, as its effects on monocytes/macrophages are largely antagonistic to those of IFN-γ. Therefore, IL-4 promotes a macrophage phenotypic profile that is distinct from the classically activated macrophage generated by the pro-inflammatory cytokine IFN-γ.
For example, MR-mediated phagocytosis is strongly induced by IL-4 and inhibited by IFN-γ, which instead supports IgG-mediated phagocytosis. The production of pro-inflammatory cytokines (IL-1, IL-6, tumor necrosis factor-α) is induced by IFN-γ but inhibited by IL-4, which instead induces anti-inflammatory cytokines (IL-1 receptor antagonist and IL-10), thereby promoting wound healing and matrix deposition. Reactive oxygen and nitrogen species are induced by IFN-γ, but IL-4 inhibits these activities and instead induces arginase-1 (in the mouse), which mediates collagen deposition and tissue repair.
Collectively, the classically activated macrophage exhibits capacities for cellular immunity, microbicide, and tissue damage, whereas alternatively activated macrophages exhibit enhanced capacities for humoral immunity, allergic responses, and repair processes such as fibrosis, matrix remodeling, phagocytosis, and, notably, down-modulation of inflammation [62–65]. Thus, it is reasonable to predict, in keeping with current views of structure/function relationships in biological systems, that morphologically distinct types of giant cells induced by IL-4 (or IL-13) and IFN-γ will prove to exhibit distinguishable phenotypes and possess distinct functional capacities.
The FBGC that form on implanted materials have been widely believed to directly bring about the degradation of certain implanted biomaterials, negatively impacting their efficacy and biocompatibility. This perspective stems from early scanning electron micrographic analyses, which revealed pitting and cracking on poly(etherurethane) biomaterial surfaces in FBGC “footprints”, i.e. areas from which adherent FBGC had been removed [66]. This seemed to indicate, and it was thus was inferred, that polymer degradation resulted from concentration of phagocytic oxidative and microbicidal activities within an acidic closed compartment or microenvironment at the FBGC/biomaterial interface.
Paradoxically, this perspective appears to be incompatible with our subsequent findings on FBGC formation. Of three discovered human macrophage fusion factors, IL-4, IL-13, and α-tocopherol [11, 13, 22], each is well and widely known, not to promote, but to down-modulate so-called “pro-inflammatory” and oxidative activities of macrophages. IL-4 and IL-13 are cytokines which each exert inhibitory effects on respiratory burst activity, the expression and secretion of pro-inflammatory cytokines, monocyte adhesion to endothelium, cytotoxic activities, and chronic destructive experimental arthritis [62, 67]. Instead, they induce mannose receptor expression, wound healing, angiogenesis, and tissue remodeling [61, 63, 65, 68].
Our findings with α-tocopherol, which is not a cytokine but the major component of vitamin E, further oppose the view that FBGC are the perpetraters of oxidative damage to biomedical polymers. In addition to its well known antioxidant properties and host tissue protective effects [69], α-tocopherol exerts non-antioxidant effects on macrophages, including the activation of diacylglycerol kinase related to production of phosphatidic and lysophosphatidic acids, actin polymerization, chemotaxis, cellular migration, and cell survival [29, 70–73]. Interestingly, α-tocopherol induces connective tissue growth factor (CTGF), which may promote connective tissue fibrosis at sites of chronic tissue injury and wound healing [74]. Accordingly, we have identified CTGF in cultures of fusing macrophages/FBGC (unpublished data). This may indicate that CTGF is synthesized and secreted by macrophages/FBGC at sites of biomaterial implantation.
Resolution of this apparent paradox is possible, however, if one supposes that biomaterial-adherent monocyte-derived macrophages initially exhibit a pro-inflammatory and oxidative phenotype with capacities for biomaterial surface degradation. Subsequently, this phenotype could undergo down-modulation by IL-4- and/or IL-13 signaling in a process of alternative macrophage activation that is accompanied by fusion of the adherent macrophages. The resultant biomaterial-adherent FBGC would exhibit capacities for wound healing, angiogenesis, and/or tissue remodeling, and yet the “footprints” of pro-inflammatory and oxidative activities would remain on the biomaterial surface and appear to have been mediated by the FBGC. If so, and if one could accelerate IL-4-induced alternative macrophage activation and FBGC formation, biomaterial degradation should be reduced. This is consistent with our observed decreased biomaterial degradation coupled with increased FBGC formation on vitamin E-modified poly(etherurethane) in vivo (unpublished data). Whether decreased biomaterial degradation resulted from the antioxidant activity of vitamin E and/or to vitamin E (α-tocopherol)-mediated increases in FBGC formation remains an intriguing question.
Accordingly, our in vitro studies on cytokine production by biomaterial-adherent monocytes, macrophages, and IL-4-induced FBGC support the concept of a time-dependent phenotypic switch. Initially, we found a cytokine switch from a pro-inflammatory to an alternative macrophage activation phenotype and a dissociation between pro-inflammatory cytokine production and FBGC formation [75]. In additional cytokine array studies aimed at determining the direct influences of lymphocytes in co-culture with monocytes/macrophages, we discovered that temporal patterns of cytokine production switch from an initial pro-inflammatory phenotype with IFN-γ production [76]. Further, when acute inflammatory cells (polymorphonuclear leukocytes) are included in initial co-culture with monocytes/macrophages (from day 0 to day 3), they exert a negative influence on subsequent IL-4-induced FBGC formation [77].
7.6.1 FBGC Versus Osteoclasts
Regarding the above-mentioned functional differences between giant cell types, it is now clear that osteoclasts differ from FBGC by several significant criteria. Most obviously, although both cell types are multinucleated and of monocyte-derived macrophage origin, osteoclasts are a feature of normal, non-inflammatory physiology [32]. FBGC, on the other hand, arise only under conditions of chronic inflammation [2, 3]. In addition and as discussed, FBGC are induced by IL-4 both in vitro and in vivo, but osteoclast formation and mature osteoclast function are inhibited by IL-4 [78]. Conversely, osteoclasts are formed under the influences of receptor activator of nuclear factor-κB ligand (RANKL) and tumor necrosis factor-α, the latter of which does not support FBGC formation [11]. In terms of adhesion, osteoclasts adhere to bone via αVβ3 and αVβ5 integrin interactions with osteopontin and bone sialoprotein [32]. Adhesion mechanisms that operate in FBGC do not include β3 integrins but, as outlined above, appear to involve αMβ2, αXβ2, αVβ1, perhaps additional β1 integrins, and adsorbed vitronectin [30, 31, 33]. The role of β5 integrins in FBGC adhesion has not yet been addressed. Phenotypic studies to probe for the expression of recognized osteoclast markers in IL-4-induced FBGC indicate that these multinucleated giant cells do not express tartrate-resistant acid phosphatase or calcitonin receptors (manuscript in preparation), further differentiating FBGC from osteoclasts.
7.7 Summary
Our perspectives on giant multinucleated cells of inflammation have evolved considerably in the last two decades. Significant progress has been made in determining macrophage fusion factors, signaling pathways, adhesion receptors/proteins, and fusion mechanisms for FBGC formation. Importantly and based on their generation from clearly opposing signaling pathways, the long-observed morphological distinctions between FBGC and LGC now extend to potential phenotype and function. Macrophage multinucleation is apparently a much more complex phenomenon than the coincidental existence of giant cell “variants”. Further studies on these intriguing cells, directed at establishing new phenotypic classifications between types of giant cells, will broaden our perspectives of chronic inflammatory processes and perhaps reveal additional complexities therein. Ongoing investigations will ultimately reveal the physiological significances and roles played by these remarkable cells in inflammation.
References
Langhans T (1868) Über Riesenzellen mit wandständigen Kernen in Tuberkeln und die fibröse Form des Tuberkels. Arch Pathol Anat 42:382–404
Chambers TJ, Spector WG (1982) Inflammatory giant cells. Immunobiology 161:283–289
Anderson JM (2000) Multinucleated giant cells. Curr Opin Hematol 7:40–47
Anderson JM (1988) Inflammatory response to implants. ASAIO Trans 34:101–107
Zhao OH, Anderson JM, Hiltner A et al (1992) Theoretical analysis on cell size distribution and kinetics of foreign-body giant cell formation in vivo on polyurethane elastomers. J Biomed Mat Res 26:1019–1038
Murch AR, Grounds MD, Marshall CA et al (1982) Direct evidence that inflammatory multinucleate giant cells form by fusion. J Pathol 137:177–180
Sutton JS, Weiss L (1966) Transformation of monocytes in tissue culture into macrophages, epithelioid cells, and multinucleated giant cells. An electron microscope study. J Cell Biol 28:303–332
Henson PM, Henson JE, Fittschen C et al (1988) Phagocytic cells: Degranulation and secretion. In: Gallin JI, Snyderman R (eds) Inflammation: Basic principles and clinical correlates, edn, Raven, New York, NY
Vignery A, Niven-Fairchild T, Ingbar DH et al (1989) Polarized distribution of Na+,K+-ATPase in giant cells elicited in vivo and in vitro. J Histochem Cytochem 37:1265–1271
Brodbeck WG, Anderson JM (2009) Giant cell formation and function. Curr Opin Hematol 16:53–57
McNally AK, Anderson JM (1995) Interleukin-4 induces foreign body giant cells from human monocytes/macrophages. Differential lymphokine regulation of macrophage fusion leads to morphological variants of multinucleated giant cells. Am J Pathol 147:1487–1499
DeFife KM, Jenney CR, Colton E et al (1999) Cytoskeletal and adhesive structural polarizations accompany IL-13-induced human macrophage fusion. J Histochem Cytochem 47:65–74
McNally AK, Anderson JM (2003) Foreign body-type multinucleated giant cell formation is potently induced by alpha-tocopherol and prevented by the diacylglycerol kinase inhibitor R59022. Am J Pathol 163:1147–1156
Enelow RI, Sullivan GW, Carper HT et al (1992) Induction of multinucleated giant cell formation from in vitro culture of human monocytes with interleukin-3 and interferon-gamma: comparison with other stimulating factors. Am J Respir Cell Mol Biol 6:57–62
McInnes A, Rennick DM (1988) Interleukin 4 induces cultured monocytes/macrophages to form giant multinucleated cells. J Exp Med 167:598–611
Weinberg JB, Hobbs MM, Misukonis MA (1984) Recombinant human gamma-interferon induces human monocyte polykaryon formation. Proc Natl Acad Sci USA 81:4554–4557
Takashima T, Ohnishi K, Tsuyuguchi I et al (1993) Differential regulation of formation of multinucleated giant cells from concanavalin A-stimulated human blood monocytes by IFN-gamma and IL-4. J Immunol 150:3002–3010
Most J, Neumayer HP, Dierich MP (1990) Cytokine-induced generation of multinucleated giant cells in vitro requires interferon-gamma and expression of LFA-1. Eur J Immunol 20:1661–1667
Orentas RJ, Reinlib L, Hildreth JE (1992) Anti-class II MHC antibody induces multinucleated giant cell formation from peripheral blood monocytes. J Leukoc Biol 51:199–209
Elliott MJ, Gamble JR, Park LS et al (1991) Inhibition of human monocyte adhesion by interleukin-4. Blood 77:2739–2745
Anderson JM, Defife K, McNally A et al (1999) Monocyte, macrophage and foreign body giant cell interactions with molecularly engineered surfaces. J Mat Sci 10:579–588
DeFife KM, Jenney CR, McNally AK et al (1997) Interleukin-13 induces human monocyte/macrophage fusion and macrophage mannose receptor expression. J Immunol 158:3385–3390
Kao WJ, McNally AK, Hiltner A et al (1995) Role for interleukin-4 in foreign-body giant cell formation on a poly(etherurethane urea) in vivo. J Biomed Mat Res 29:1267–1275
Rodriguez A, Macewan SR, Meyerson H et al (2009) The foreign body reaction in T-cell-deficient mice. J Biomed Mat Res 90:106–113
Gessner A, Mohrs K, Mohrs M (2005) Mast cells, basophils, and eosinophils acquire constitutive IL-4 and IL-13 transcripts during lineage differentiation that are sufficient for rapid cytokine production. J Immunol 174:1063–1072
O‘Connor GM, Hart OM, Gardiner CM (2006) Putting the natural killer cell in its place. Immunology 117:1–10
Brodbeck WG, Shive MS, Colton E et al (2002) Interleukin-4 inhibits tumor necrosis factor-alpha-induced and spontaneous apoptosis of biomaterial-adherent macrophages. J Lab Clin Med 139:90–100
Schubert MA, Wiggins MJ, DeFife KM et al (1996) Vitamin E as an antioxidant for poly(etherurethane urea): in vivo studies. Student Research Award in the Doctoral Degree Candidate Category, Fifth World Biomaterials Congress (22nd Annual Meeting of the Society for Biomaterials), Toronto, Canada, May 29-June 2, 1996. J Biomed Mat Res 32:493–504
Azzi A, Stocker A (2000) Vitamin E: non-antioxidant roles. Progress Lipid Res 39:231–255
McNally AK, Jones JA, Macewan SR et al (2008) Vitronectin is a critical protein adhesion substrate for IL-4-induced foreign body giant cell formation. J Biomed Mat Res 86:535–543
McNally AK, Anderson JM (2002) Beta1 and beta2 integrins mediate adhesion during macrophage fusion and multinucleated foreign body giant cell formation. Am J Pathol 160:621–630
Teitelbaum SL (2007) Osteoclasts: what do they do and how do they do it? Am J Pathol 170:427–435
McNally AK, Macewan SR, Anderson JM (2007) alpha subunit partners to beta1 and beta2 integrins during IL-4-induced foreign body giant cell formation. J Biomed Mat Res 82:568–574
Ruoslahti E (1996) RGD and other recognition sequences for integrins. Annu Rev Cell Dev Biol 12:697–715
Ratnikov BI, Partridge AW, Ginsberg MH (2005) Integrin activation by talin. J Thromb Haemost 3:1783–1790
Duong LT, Rodan GA (2000) PYK2 is an adhesion kinase in macrophages, localized in podosomes and activated by beta(2)-integrin ligation. Cell Motil Cytoskeleton 47:174–188
MacEwan SR, McNally A, Anderson JM (2007) Focal adhesion kinase, proline-rich tyrosine kinase-2, thrombospondin, and fascin-1 expression in adherent macrophages and foreign body giant cells. Society for Biomaterials Annual Meeting, Chicago, IL, April 18–21,703
DeFife KM, Jenney CR, Colton E et al (1999) Disruption of filamentous actin inhibits human macrophage fusion. Faseb J 13:823–832
McNally AK, Anderson JM (2005) Multinucleated giant cell formation exhibits features of phagocytosis with participation of the endoplasmic reticulum. Exp Mol Pathol 79:126–135
Marx J (2006) Cell biology. Podosomes and invadopodia help mobile cells step lively. Science 312:1868–1869
Calle Y, Burns S, Thrasher AJ et al (2006) The leukocyte podosome. Eur J Cell Biol 85:151–157
Frisch SM, Screaton RA (2001) Anoikis mechanisms. Curr Opin Cell Biol 13:555–562
McNally A (1994) Mechanisms of monocyte/macrophage adhesion and fusion on different surfaces. PhD Thesis, Case Western Reserve University, Cleveland, OH
Brodbeck WG, Colton E, Anderson JM (2003) Effects of adsorbed heat labile serum proteins and fibrinogen on adhesion and apoptosis of monocytes/macrophages on biomaterials. J Mat Sci 14:671–675
Brodbeck WG, Patel J, Voskerician G et al (2002) Biomaterial adherent macrophage apoptosis is increased by hydrophilic and anionic substrates in vivo. Proc Natl Acad Sci USA 99:10287–10292
Jones JA, Dadsetan M, Collier TO et al (2004) Macrophage behavior on surface-modified polyurethanes. J Biomat Sci 15:567–584
Shive MS, Brodbeck WG, Anderson JM (2002) Activation of caspase 3 during shear stress-induced neutrophil apoptosis on biomaterials. J Biomed Mat Res 62:163–168
Stein M, Keshav S, Harris N et al (1992) Interleukin 4 potently enhances murine macrophage mannose receptor activity: a marker of alternative immunologic macrophage activation. J Exp Med 176:287–292
McNally AK, DeFife KM, Anderson JM (1996) Interleukin-4-induced macrophage fusion is prevented by inhibitors of mannose receptor activity. Am J Pathol 149:975–985
McNally AK, Macewan SR, Anderson JM (2008) Foreign body-type multinucleated giant cell formation requires protein kinase C beta, delta, and zeta. Exp Mol Pathol 84:37–45
Carnevale KA, Cathcart MK (2003) Protein kinase C beta is required for human monocyte chemotaxis to MCP-1. J Biol Chem 278:25317–25322
Larsen EC, DiGennaro JA, Saito N et al (2000) Differential requirement for classic and novel PKC isoforms in respiratory burst and phagocytosis in RAW 264.7 cells. J Immunol 165:2809–2817
Larsson C (2006) Protein kinase C and the regulation of the actin cytoskeleton. Cell Signal 18:276–284
Liu Q, Ning W, Dantzer R et al (1998) Activation of protein kinase C-zeta and phosphatidylinositol 3′-kinase and promotion of macrophage differentiation by insulin-like growth factor-I. J Immunol 160:1393–1401
Harsh DM, Blackwood RA (2001) Phospholipase A(2)-mediated fusion of neutrophil-derived membranes is augmented by phosphatidic acid. Biochem Biophys Res Commun 282:480–486
Jones JA, McNally AK, Chang DT et al (2008) Matrix metalloproteinases and their inhibitors in the foreign body reaction on biomaterials. J Biomed Mat Res 84:158–166
Mantovani A, Sica A, Locati M (2007) New vistas on macrophage differentiation and activation. Eur J Immunol 37:14–16
Mantovani A, Sica A, Sozzani S et al (2004) The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol 25:677–686
Porcheray F, Viaud S, Rimaniol AC et al (2005) Macrophage activation switching: an asset for the resolution of inflammation. Clin Exp Immunol 142:481–489
Taylor PR, Martinez-Pomares L, Stacey M et al (2005) Macrophage receptors and immune recognition. Annu Rev Immunol 23:901–944
Goerdt S, Politz O, Schledzewski K et al (1999) Alternative versus classical activation of macrophages. Pathobiology 67:222–226
Gordon S (2003) Alternative activation of macrophages. Nat Rev Immunol 3:23–35
Gratchev A, Kzhyshkowska J, Utikal J et al (2005) Interleukin-4 and dexamethasone counterregulate extracellular matrix remodelling and phagocytosis in type-2 macrophages. Scand J Immunol 61:10–17
Moestrup SK, Moller HJ (2004) CD163: a regulated hemoglobin scavenger receptor with a role in the anti-inflammatory response. Ann Med 36:347–354
Mosser DM (2003) The many faces of macrophage activation. J Leukoc Biol 73:209–212
Zhao Q, Topham N, Anderson JM et al (1991) Foreign-body giant cells and polyurethane biostability: in vivo correlation of cell adhesion and surface cracking. J Biomed Mat Res 25:177–183
Malefyt RW (1999) Role of interleukin-10, interleukin-4, and interleukin-13 in resolving inflammatory responses. In: Gallin JL, Snyderman R (eds) Inflammation: Basic principles and clinical correlates, edn, Lippincott Williams & Wilkins, Philadelphia, PA
Kodelja V, Muller C, Tenorio S et al (1997) Differences in angiogenic potential of classically vs alternatively activated macrophages. Immunobiology 197:478–493
Brigelius-Flohe R, Traber MG (1999) Vitamin E: function and metabolism. Faseb J 13:1145–1155
Chan SS, Monteiro HP, Schindler F et al (2001) Alpha-tocopherol modulates tyrosine phosphorylation in human neutrophils by inhibition of protein kinase C activity and activation of tyrosine phosphatases. Free Rad Res 35:843–856
Koh JS, Lieberthal W, Heydrick S et al (1998) Lysophosphatidic acid is a major serum noncytokine survival factor for murine macrophages which acts via the phosphatidylinositol 3-kinase signaling pathway. J Clin Invest 102:716–727
Lee IK, Koya D, Ishi H et al (1999) d-Alpha-tocopherol prevents the hyperglycemia induced activation of diacylglycerol (DAG)-protein kinase C (PKC) pathway in vascular smooth muscle cell by an increase of DAG kinase activity. Diabetes Res Clin Pract 45:183–190
Topham MK, Prescott SM (1999) Mammalian diacylglycerol kinases, a family of lipid kinases with signaling functions. J Biol Chem 274:11447–11450
Leask A, Holmes A, Abraham DJ (2002) Connective tissue growth factor: a new and important player in the pathogenesis of fibrosis. Curr Rheumatol Rep 4:136–142
Jones JA, Chang DT, Meyerson H et al (2007) Proteomic analysis and quantification of cytokines and chemokines from biomaterial surface-adherent macrophages and foreign body giant cells. J Biomed Mat Res 83:585–596
Chang DT, Colton E, Matsuda T et al (2009) Lymphocyte adhesion and interactions with biomaterial adherent macrophages and foreign body giant cells. J Biomed Mat Res 91:1210–1220
Kirk JT, McNally AK, Anderson JM (2010) Polymorphonuclear leukocyte inhibition of monocytes/macrophages in the foreign body reaction. J Biomed Mat Res 94:683–687
Moreno JL, Kaczmarek M, Keegan AD et al (2003) IL-4 suppresses osteoclast development and mature osteoclast function by a STAT6-dependent mechanism: irreversible inhibition of the differentiation program activated by RANKL. Blood 102:1078–1086
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Springer Science+Business Media B.V.
About this chapter
Cite this chapter
McNally, A.K., Anderson, J.M. (2011). Macrophage Fusion and Multinucleated Giant Cells of Inflammation. In: Dittmar, T., Zänker, K.S. (eds) Cell Fusion in Health and Disease. Advances in Experimental Medicine and Biology, vol 713. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-0763-4_7
Download citation
DOI: https://doi.org/10.1007/978-94-007-0763-4_7
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-0762-7
Online ISBN: 978-94-007-0763-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)